Chapter 6 Electron structure of atoms 1 6













































![Answer Se = 34 e[Ar] 4 s 2 3 d 10 4 p 4 Answer Se = 34 e[Ar] 4 s 2 3 d 10 4 p 4](https://slidetodoc.com/presentation_image_h/869e67c52487ae9f38862f431c8489db/image-66.jpg)


- Slides: 69

Chapter 6 Electron structure of atoms 1

6. 1 Electromagnetic Radiation l Light that we can see is visible light which is a type of electromagnetic radiation. l Radiant energy is energy that carries energy that acts like a wave and travels through space at the speed of light. Earth’s Radiant Energy 2

c = speed of light 3. 0 x 108 m/s 3

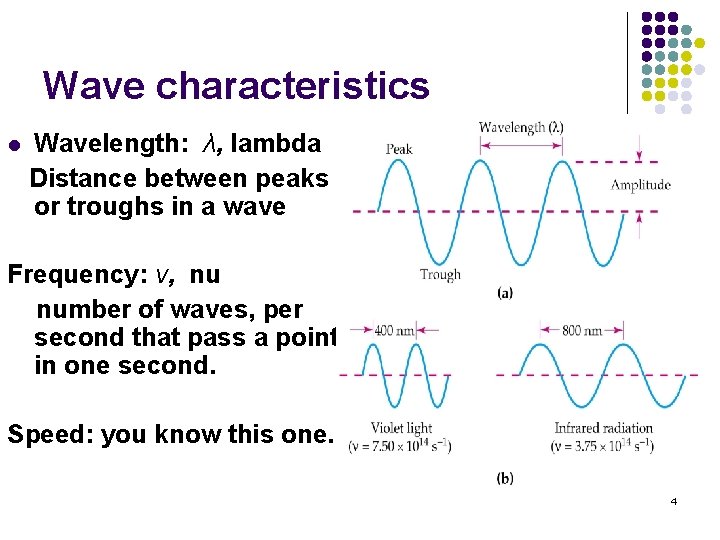
Wave characteristics l Wavelength: λ, lambda Distance between peaks or troughs in a wave Frequency: ν, nu number of waves, per second that pass a point in one second. Speed: you know this one. 4

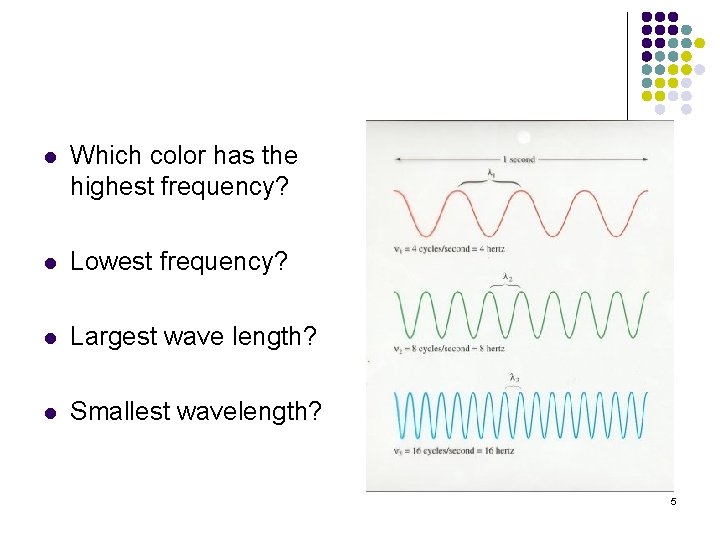
l Which color has the highest frequency? l Lowest frequency? l Largest wave length? l Smallest wavelength? 5

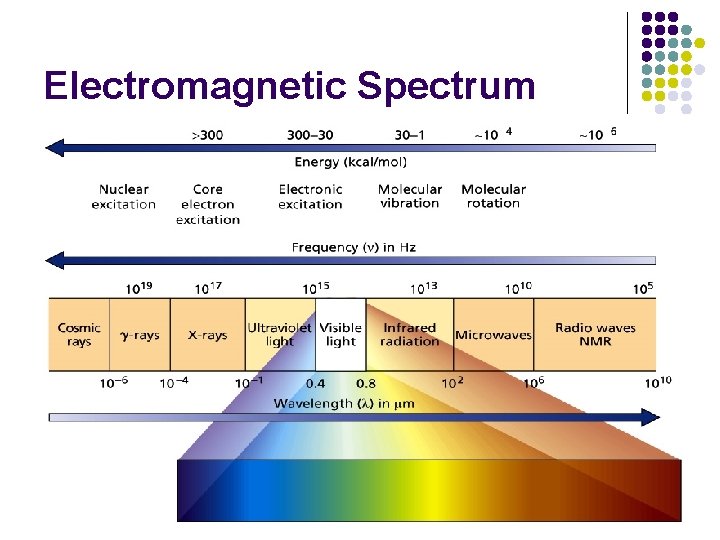
Electromagnetic Spectrum 6

Flame testing l http: //www. sciencefriday. com/videos/watch/1 0227 7

Relationship between λ and ν l Wavelength and frequency are inverses of each other. l λv = c l λ = wavelength in meters (m) ν = frequency in cycles per second (1/s or s-1 or Hertz) c = speed of light 3. 0 x 108 m/s l l 8

Try one! l The red wavelength emitted from red fireworks is around 650 nm and results when strontium salts are heated. Calculate the frequency of the red light of this wavlength. l λv=c λ = (6. 50 x 102 nm) = 6. 50 x 10 -7 m v = 4. 61 x 1014 s-1 or Hz l l 9

6. 2 Planck’s Constant Max Planck discovered that energy could be gained or lost in multiples of a constant (h) times its frequency (ν). h = 6. 626 x 10 -34 J *s 10

Quantized Energy Thus energy is quantized or in steps or packages. Energy can only be transferred as a whole package or quantum. 11

Solving equations with Planck’s E = change in energy, in J h = Planck’s constant, 6. 626 10 34 J s = frequency, in s 1 = wavelength, in m 12

Calculating energy lost l The blue color in fireworks is the result of heated Cu. Cl at 1200 °C. Then the compound emits blue light with a wavelength of 450 nm. What is the increment of energy (quantum) that is emitted at 4. 50 x 102 nm by Cu. Cl? 13

Answer ΔE = hν l v = 3. 0 x 108 m/s 4. 50 x 10 -7 m = 6. 66 x 1014 s-1 v = c/λ (6. 626 x 10 -34 J *s) x (6. 66 x 1014 s-1) = 4. 41 x 10 -19 J (quantum energy lost in this increment) 14

photons l Einstein took Planck’s idea a step further and proposed that electromagnetic radiation was quantized into particles called photons (light). l The energy of each photon is given by the expression: Ephoton = hν = hc/λ 15

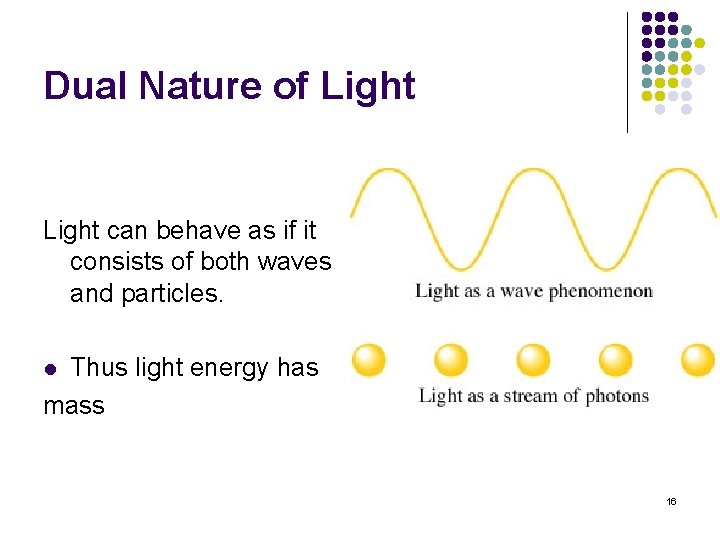
Dual Nature of Light can behave as if it consists of both waves and particles. Thus light energy has mass l 16

Old-ie but good-ie l Energy has mass l. E l l l = mc 2 E = energy m = mass c = speed of light 17

6. 4 The Behavior of the wave De Broglie We can calculate the wavelength of an e-. = wavelength, in m h = Planck’s constant, 6. 626 10 34 J s v = velocity m = mass in kg 18

Question l Compare the wavelength for an electron (mass = 9. 11 x 10 -31 kg) traveling at a speed of 1. 0 x 107 m/s with that of a ball (mass = 0. 10 kg) traveling at 35 m/s 19

Answer Electron wavelength = 7. 27 x 10 -11 m ball wavelength = 1. 9 x 10 -34 m 20

ν = nu = frequency v = velocity your book uses μ for velocity 21

Homework Chang: pg 303 #’s 1, 2, 7, 9, 15, 20 BL: Pg 230 l 1, 3, 5, 7, 10, 13, 15, 19 22

Waves = Electrons l Planck and Einstein proved that electrons in atoms act like waves of light l By understanding waves we can learn about the properties of electrons l The study of the properties of electrons is Quantum Mechanics 25

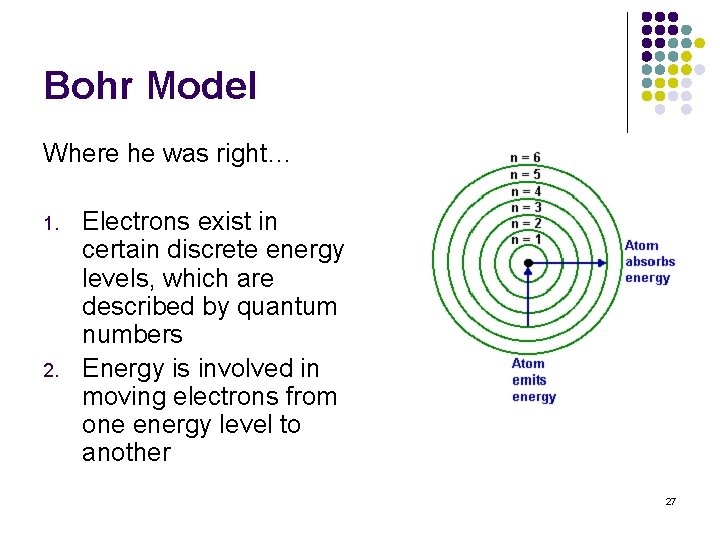
Bohr Model l Proposed the first theory on atom location and movement l His proposal was a little bit right and a lot wrong…BUT we give him props just the same Nelis Bohr 26

Bohr Model Where he was right… 1. 2. Electrons exist in certain discrete energy levels, which are described by quantum numbers Energy is involved in moving electrons from one energy level to another 27

Heisenberg Uncertainty Principle l Blew the Bohr model out of the water. l It states that we can only know so much about the exact position and momentum of an electron. …And the electron cloud is born Werner Heisenberger 28

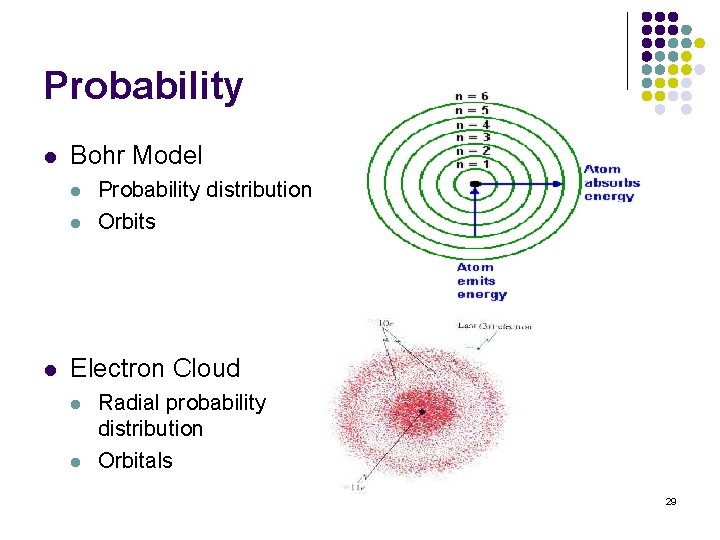
Probability l Bohr Model l Probability distribution Orbits Electron Cloud l l Radial probability distribution Orbitals 29

6. 5 Quantum model of an atom l Compared the relationship between the electron and the nucleus of an atom to that of a standing or stationary wave. The functions of these waves tell us about the electrons location and energy. Erwin SchrÖdinger 30

Schrödinger's Cat He proposed a scenario with a cat in a sealed box, where the cat's life or death was dependent on the state of a subatomic particle. According to Schrödinger, the Copenhagen interpretation implies that the cat remains both alive and dead until the box is opened. 31

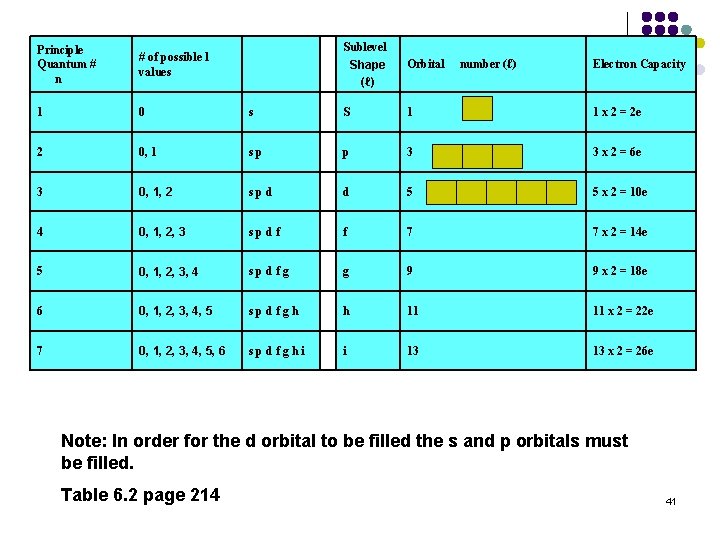
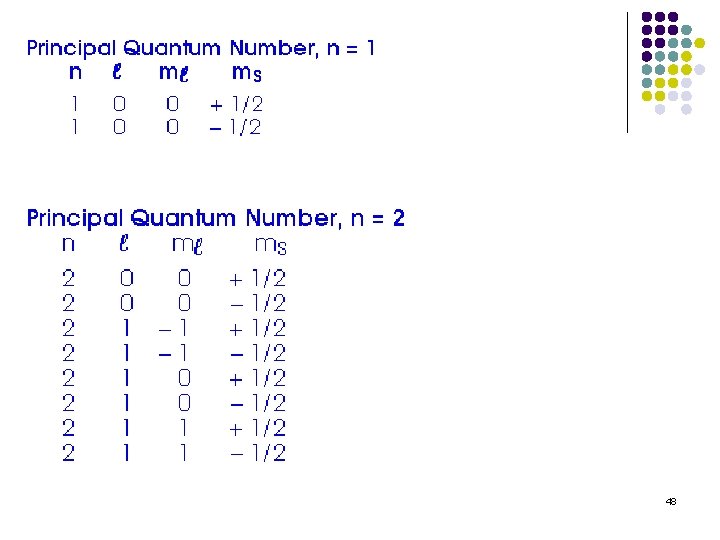
Quantum numbers!!!!! Quantum numbers describe various properties of the electrons in an atom. There are 4 quantum numbers Principal quantum number (n) Azimuthual quantum number (angular momentum) (ℓ) Magnetic quantum number (mℓ) Electron spin quantum number (ms) 33

Principal quantum number (n) l Integral values 1, 2, 3, 4, 5, 6, 7 Related to the size and energy of the orbital l Referred to as the shell or energy level l 34

Principal quantum number (n) l As n increases energy increases and orbital size increases because the electrons are farther away from the nucleus and less tightly bound to the positive protons. n=1 n=4 35

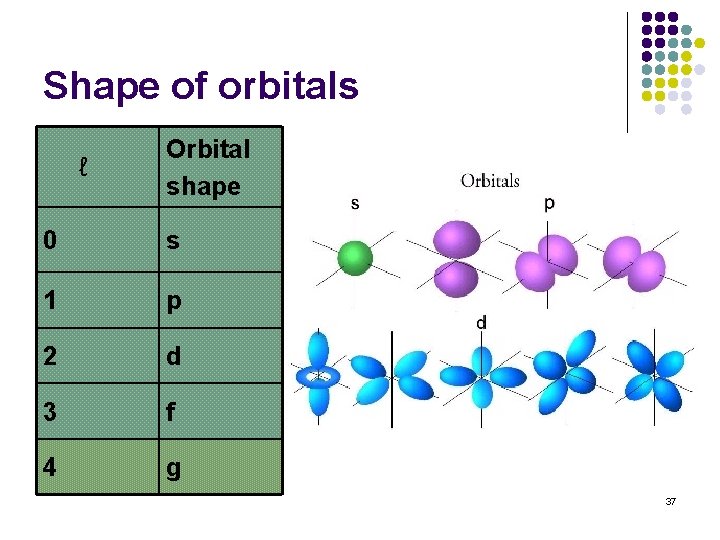
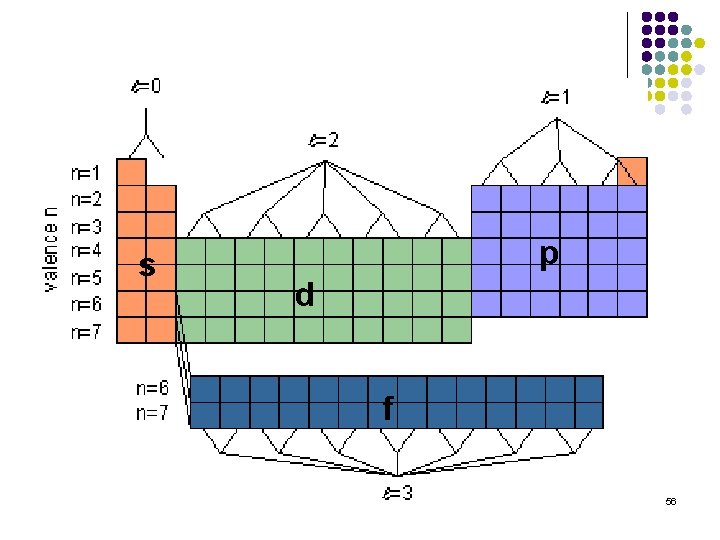
Angular momentum quantum number (ℓ) Integral numbers with values from 0 to n-1 if n = 3 possible ℓ values are 0, 1, 2 Sometimes referred to as the “sub shell” number Defines the shape of the orbital. ℓ Orbital shape 0 s 1 p 2 d 3 f 4 g 36

Shape of orbitals ℓ Orbital shape 0 s 1 p 2 d 3 f 4 g 37

Magnetic quantum number (mℓ) Integral values from ℓ to -ℓ including zero If ℓ = 2 Then mℓ = 2, 1, 0, -1, -2 Relates to the orientation of the orbital in the atom. 38

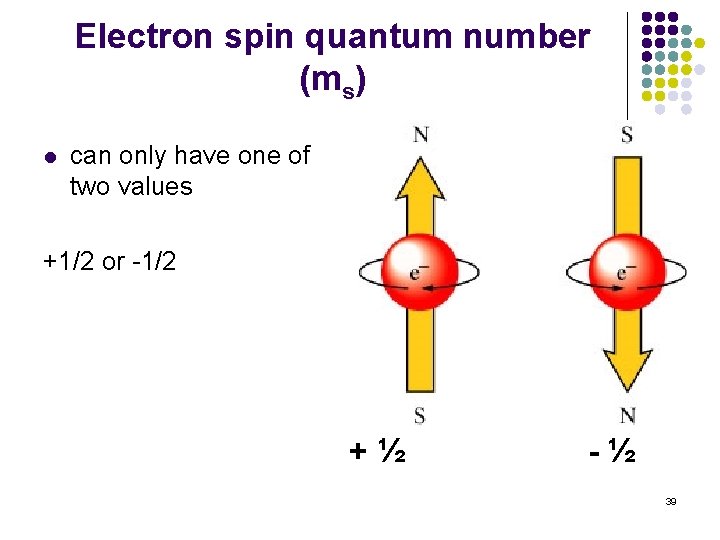
Electron spin quantum number (ms) l can only have one of two values +1/2 or -1/2 +½ -½ 39

Re cap l A collection of orbitals with the same n value is called an electron shell. EX: all orbitals that have n =3 are in the third shell. A collection of orbitals with the same n and ℓ values are in the same sub shell EX: 2 s, 2 p 40

Sublevel Shape (ℓ) Orbital number (ℓ) Electron Capacity s S 1 1 x 2 = 2 e 0, 1 s p p 3 3 x 2 = 6 e 3 0, 1, 2 s p d d 5 5 x 2 = 10 e 4 0, 1, 2, 3 s p d f f 7 7 x 2 = 14 e 5 0, 1, 2, 3, 4 s p d f g g 9 9 x 2 = 18 e 6 0, 1, 2, 3, 4, 5 s p d f g h h 11 11 x 2 = 22 e 7 0, 1, 2, 3, 4, 5, 6 s p d f g h i i 13 13 x 2 = 26 e Principle Quantum # n # of possible l values 1 0 2 Note: In order for the d orbital to be filled the s and p orbitals must be filled. Table 6. 2 page 214 41

question For the principle quantum level n = 5 Determine the number of allowed sub shells (ℓ) and give the number and letter designation of each l 42

Answer l Recall: Angular momentum quantum Integral numbers with values from 0 to n-1 n=5 ℓ = 0 or s, 1 or p, 2 or d, 3 or f, 4 or g 43

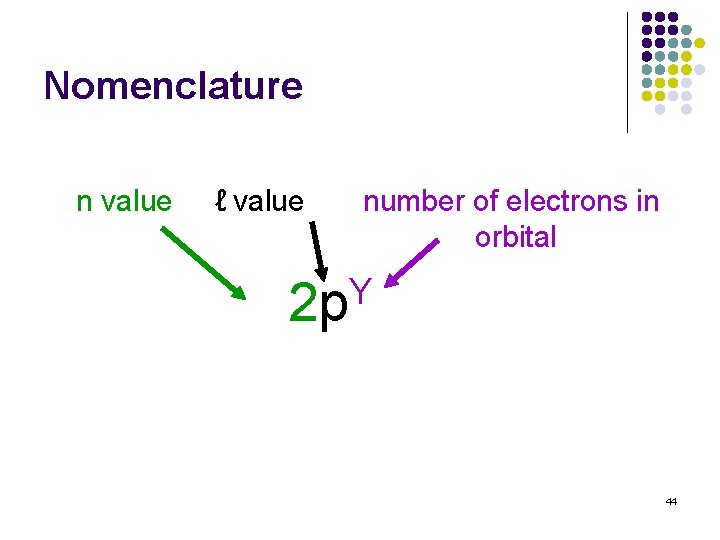
Nomenclature n value ℓ value number of electrons in orbital Y 2 p 44

Sorting our the numbers l Orbitals with the same n value are in the same shell. Ex: n = 3 is the third shell One or more orbitals with the same set of n and ℓ values are in the same sub shell Ex: n = 3 ℓ= 2 3 d sub shell n=3ℓ=1 3 p sub shell 45

Homework l Chang pg 305 #’s 43, 44. 46, 47, 48, 52, 53, 56, 57, 63, l BL: Pg 232 41, 43, 45, 46 46

Pauli exclusion principle l In a given atom no electrons can have the same 4 quantum number l So when we put more than one electron in an orbital we must alternate the spin. Thus ms = +1/2 -1/2 47

48

l Example of Pauli Exclusion Principal Quantum numbers for 2 s 2 2 s n 2 l 0 ms +1/2 2 s 2 0 0 -1/2 When ever possible electrons will prefer to have a positive spin. In this case this orbital will only hold 2 e- so one must be negative 49

Question ? What would the 4 quantum numbers be for 3 p 3? Note: all electrons have positive spin We will get to why in a minute 50

Answer n l ml ms 3 p 3 1 0 +1/2* 3 p 3 1 1 +1/2* 3 p 3 1 -1 +1/2* 51

Homework Page 232 -33 #’s 52, 53, 54, 56 l 52

Electron configuration l The order in which electrons are distributed to orbitals l We need to have rules for how we distribute electrons. Other wise all the electrons would be in the 1 s orbital because it has the lowest energy (e- ♥ ground state) l 53

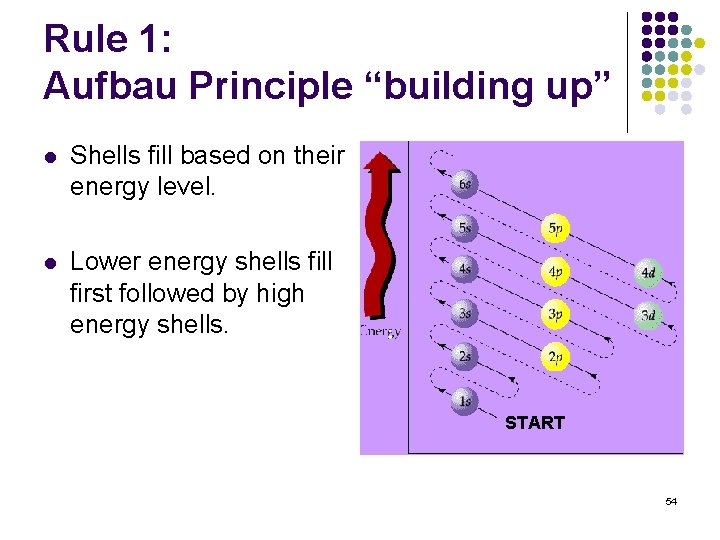
Rule 1: Aufbau Principle “building up” l Shells fill based on their energy level. l Lower energy shells fill first followed by high energy shells. START 54

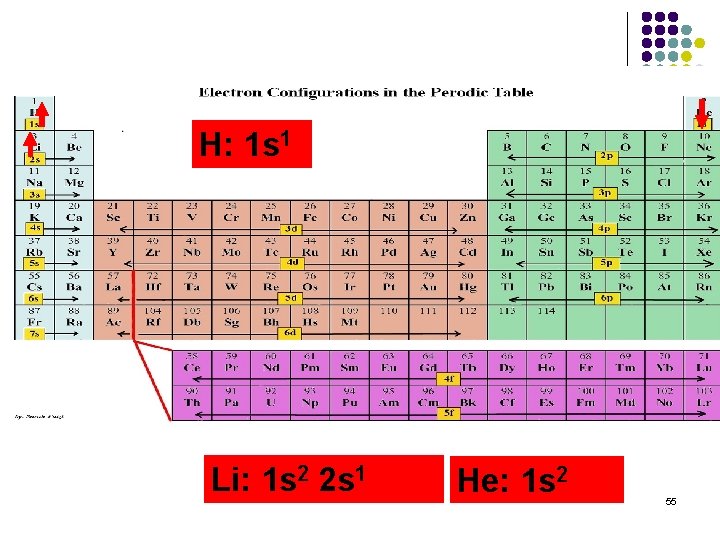
H: 1 s 1 Li: 1 s 2 2 s 1 He: 1 s 2 55

s p d f 56

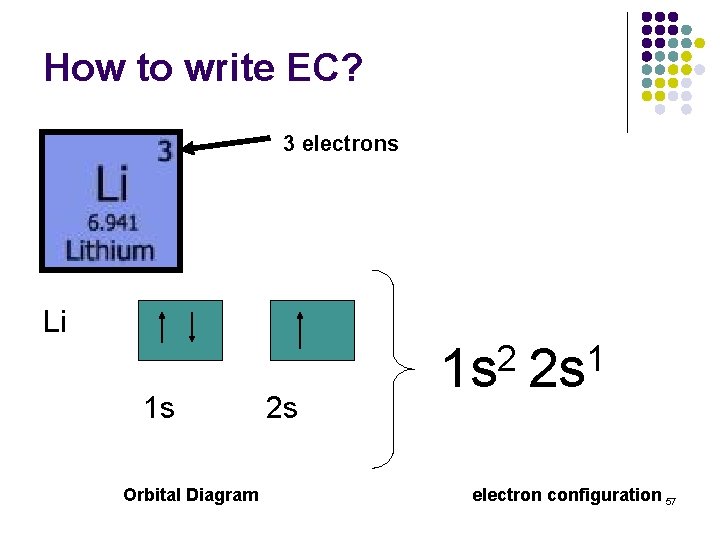
How to write EC? 3 electrons Li 1 s Orbital Diagram 2 s 2 1 1 s 2 s electron configuration 57

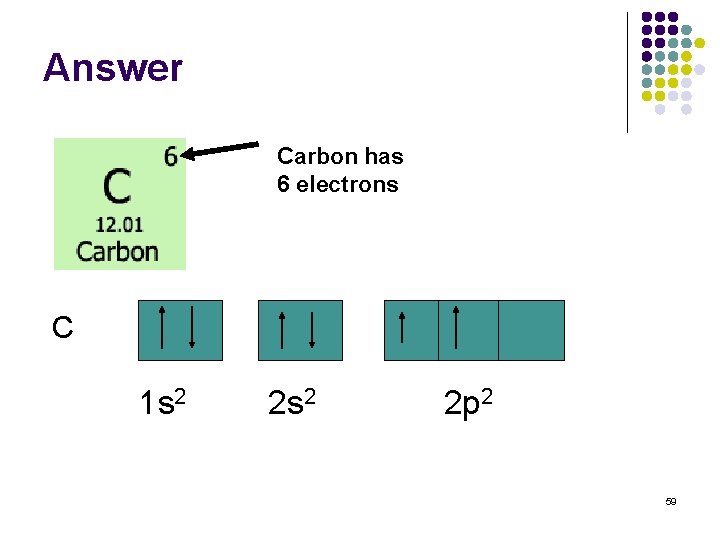
Question ? l What is the electron configuration for Carbon? 58

Answer Carbon has 6 electrons C 1 s 2 2 p 2 59

Hund’s Rule: “the grocery line rule” l Electrons are distributed among the orbitals or a sub shell in a way that gives the maximum number of unpaired electrons. C 1 s 2 2 p 2 60

Question l Write the orbital diagrams and electron configurations for the electron configurations of each element. l Nitrogen Oxygen Fluorine Potassium l l l 61

Answer 62

Valence Electrons The electrons in the outermost principle quantum level of an atom. Ve- = to group # Atom Ca N Br Ve 2 5 7 Location 4 s 2 s 2 p 4 p 3 d Inner electrons are called core electrons. 65

Short and Sweet! Writing the EC for Carbon is one thing but Xenon (54 e-), Argon (18 e-)? To write the condensed EC look to the noble gas BEFORE your element. 66

67

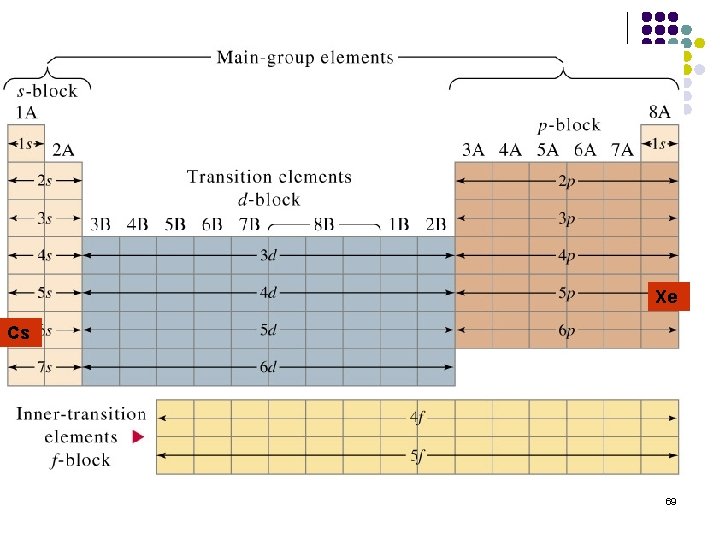
Condensed Form Example l l l Cs = 55 e. Noble gas before it is Xenon Xe= 54 e[Xe] We still need 1 more e- so we write it in [Xe] 6 s 1 68

Xe Cs 69

Question? l What is the condensed electron configuration for Selenium? 70
![Answer Se 34 eAr 4 s 2 3 d 10 4 p 4 Answer Se = 34 e[Ar] 4 s 2 3 d 10 4 p 4](https://slidetodoc.com/presentation_image_h/869e67c52487ae9f38862f431c8489db/image-66.jpg)
Answer Se = 34 e[Ar] 4 s 2 3 d 10 4 p 4 71

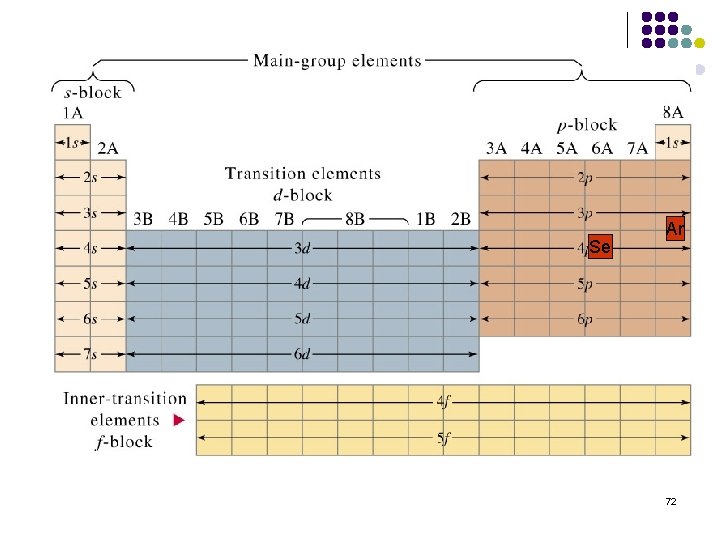
Se Ar 72

EXCEPTION ALERT!!! l l l Memorize the EC of Copper and Chromium. They are exceptions to our rules due to stability Chromium [Ar] 4 s 13 d 5 Copper [Ar] 4 s 13 d 10 73

Homework l Pg 233 l #’s 59, 60, 61, 62, 63, 65 76