Chapter 6 Chemistry in Biology 6 1 Atoms


Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Atoms § Chemistry is the study of matter. § Atoms are the building blocks of matter.

§ Neutrons and protons are located at the center of the atom. § Protons are positively charged particles. § Neutrons are particles that have no charge.

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds § Electrons are negatively charged particles that are located outside the nucleus.

Electron Arrangement • Electrons are organized in different energy levels or shells • The inner most level can hold 2 electrons • 2 e, 8 e, 18 e, 32 e

• Valence Electrons: the electrons in the outermost level • These are responsible for atoms bonding together

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Elements § Element: is a pure substance that cannot be broken down into other substances by physical or chemical means. § Each element has a unique name and symbol.

Element Examples

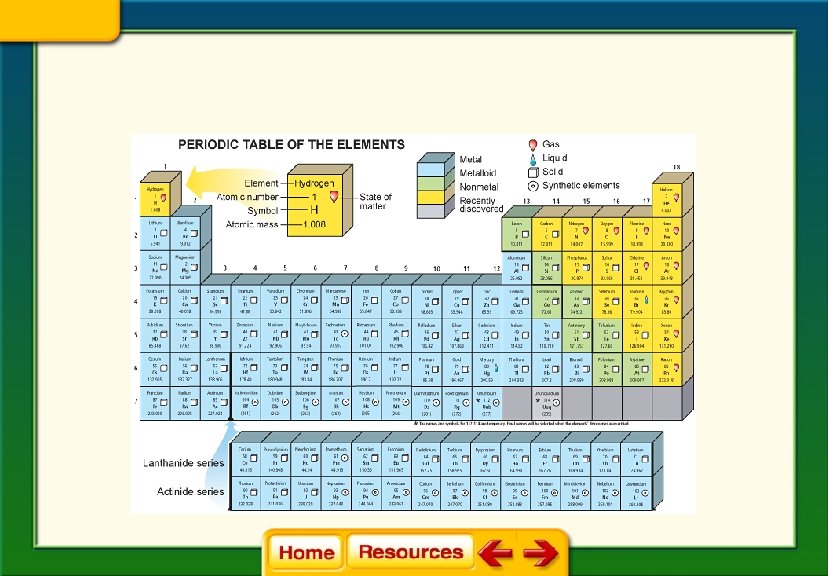
Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds The Periodic Table of Elements § Horizontal rows are called periods. §Tells the number of energy levels in each row § Vertical columns are called groups or families. §Tells the number of valence electrons in each family

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds: § A pure substance formed when two or more elements combine § Compounds are always formed from a specific combination of elements in a fixed ratio. § Compounds cannot be broken down into simpler compounds or elements by physical means.

Compound Examples • Na. Cl • 5 H 2 O • 7 Ca. Cl 2
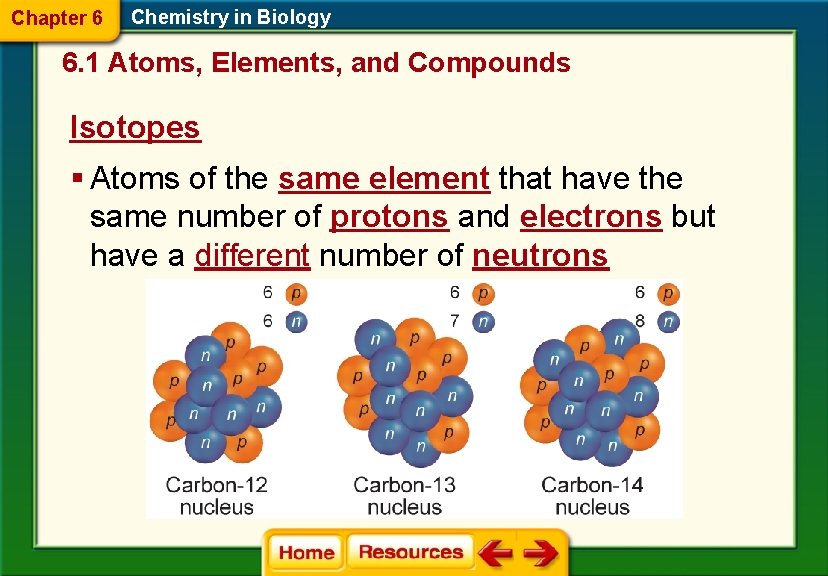
Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Isotopes § Atoms of the same element that have the same number of protons and electrons but have a different number of neutrons

• Elements are held together in compounds by bonds. • There are 2 main types of bonds: • Covalent • Ionic

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Chemical Bonds § Covalent bonds § Chemical bond that forms when electrons are shared § Occurs between nonmetals

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds Ionic Bonds § Electrical attraction between two oppositely charged atoms (ions) or groups of atoms
Chapter 6 Chemistry in Biology

• Atoms are donated or gained • Occurs between metals and non-metals

Chapter 6 Chemistry in Biology 6. 1 Atoms, Elements, and Compounds § The elements identified as metals tend to donate electrons. § The elements identified as nonmetals tend to accept electrons.
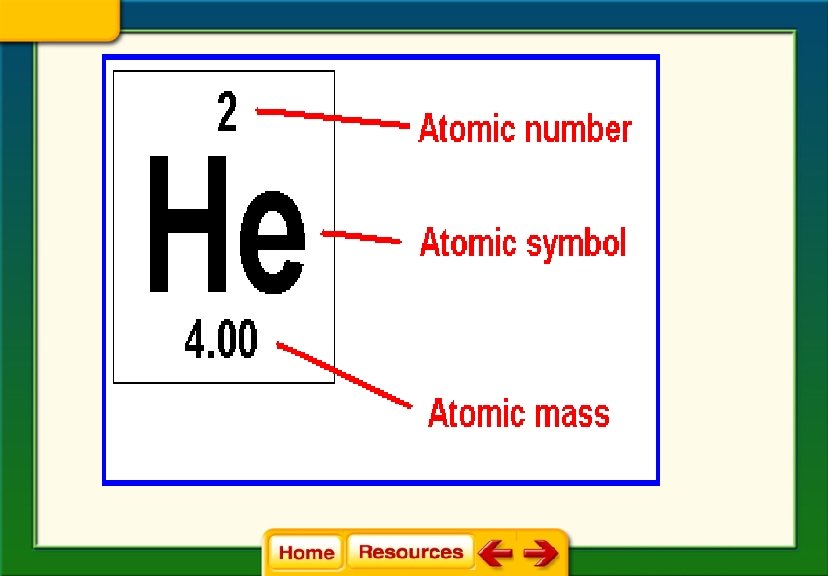

• Atomic number = number of protons • Atomic Mass = average mass of all the isotopes of an element – You can round this to find the mass number for the most common isotope of the element • Mass number = protons + neutrons

6. 2 Chemical Reactions • 2 H + O → H 2 O • Which side contains the reactant(s)? • The product(s)?
• What makes this reaction occur?
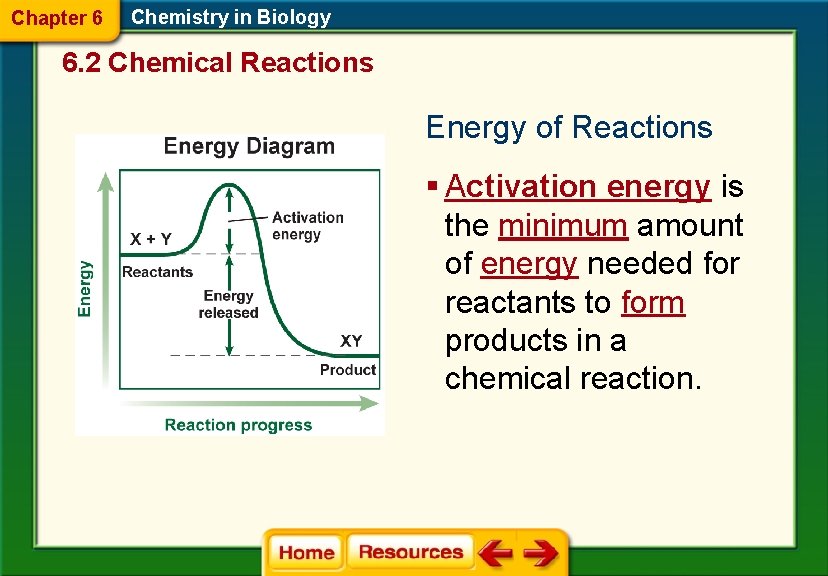
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Energy of Reactions § Activation energy is the minimum amount of energy needed for reactants to form products in a chemical reaction.
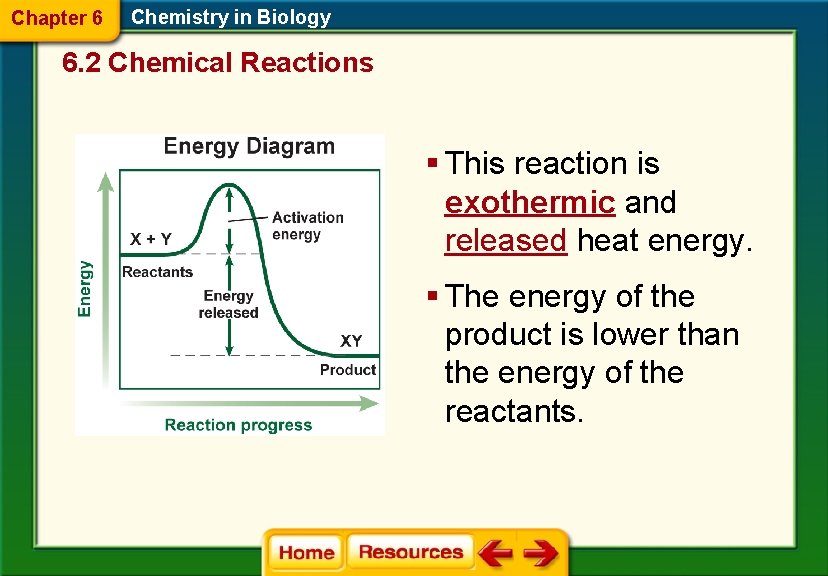
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § This reaction is exothermic and released heat energy. § The energy of the product is lower than the energy of the reactants.
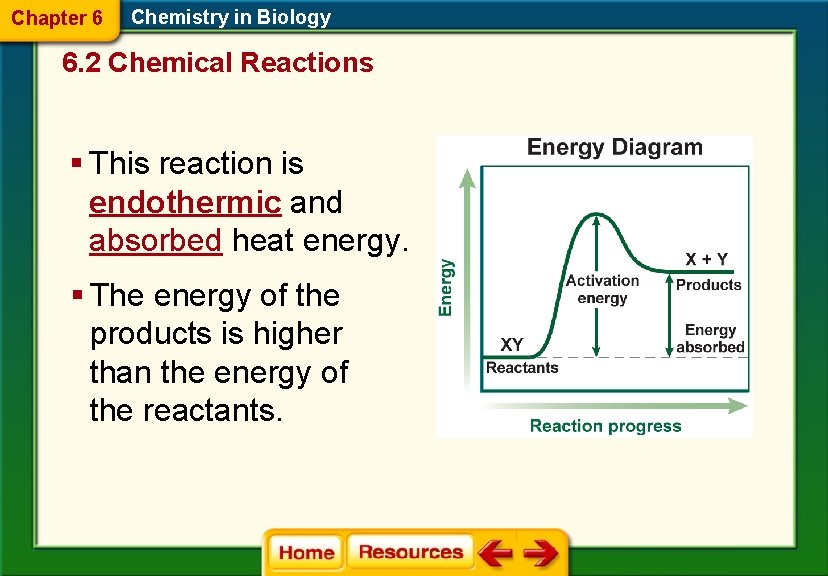
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § This reaction is endothermic and absorbed heat energy. § The energy of the products is higher than the energy of the reactants.

• We will learn about biological catalysts in the near future!

6. 4 The Building Blocks of Life • All living things are compose of organic molecules • The six most common elements found in organic molecules include: C, H, N, O, P, S.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Organic Chemistry § The element carbon is a component of almost all biological molecules.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Macromolecules § Carbon atoms can be joined to form carbon molecules. § Macromolecules are large molecules formed by joining smaller organic molecules together. § Monomers: small, repeating organic molecules that usually form larger molecules
§ They are also called biological polymers

• There are four major macromolecules associated with biology: • Carbohydrates • Lipids • Proteins • Nucleic acids
Chapter 6 Chemistry in Biology

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Carbohydrates § Compounds composed of carbon, hydrogen, and oxygen in a ratio of 1 C: 2 H: 1 O § General formula: (CH 2 O)n

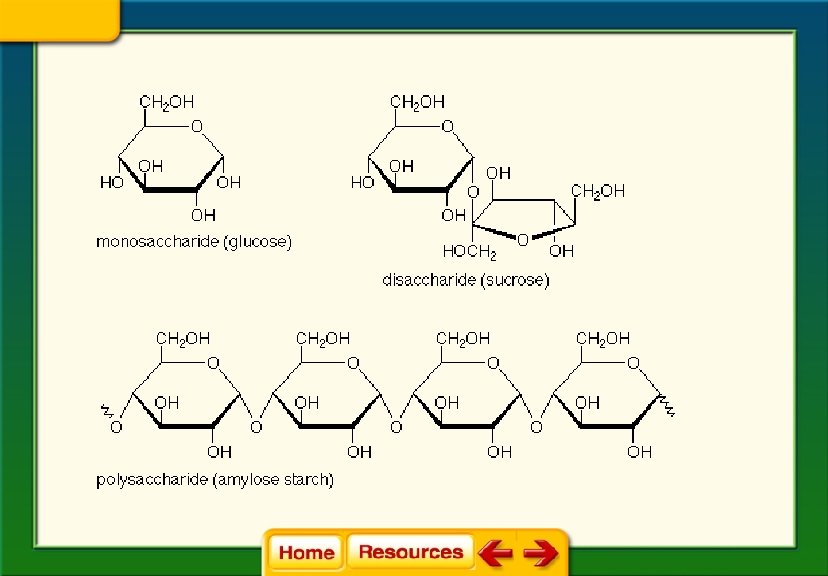
Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § If n is between three and seven the compound is called a simple sugar or monosaccharide. § Ex. C 6 H 12 O 6

§ Glucose is one very important monosaccharide used by cells for energy • It is found in the human bloodstream
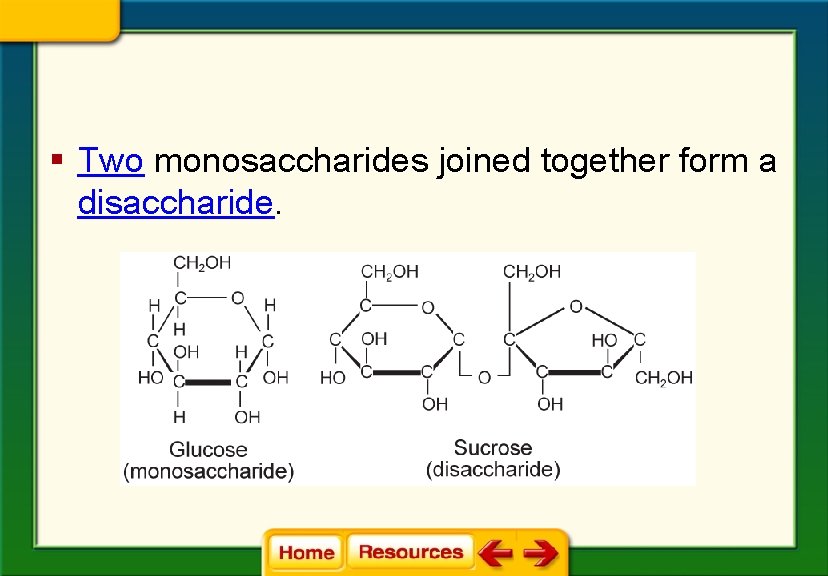
§ Two monosaccharides joined together form a disaccharide.

§ Sucrose and lactose are two examples of disaccharides. § Sucrose is table sugar § Lactose is milk sugar


How are disaccharides formed? • Dehydration synthesis: formation of disaccharide by removing water from the monosaccharides

§ Longer carbohydrate molecules are called polysaccharides.

§ Glycogen is an example of a polysaccharide. § Animals store it in the liver § It is a short term energy storage molecule

How do organisms get energy from these? ? ? • The bonds between them are broken, releasing energy
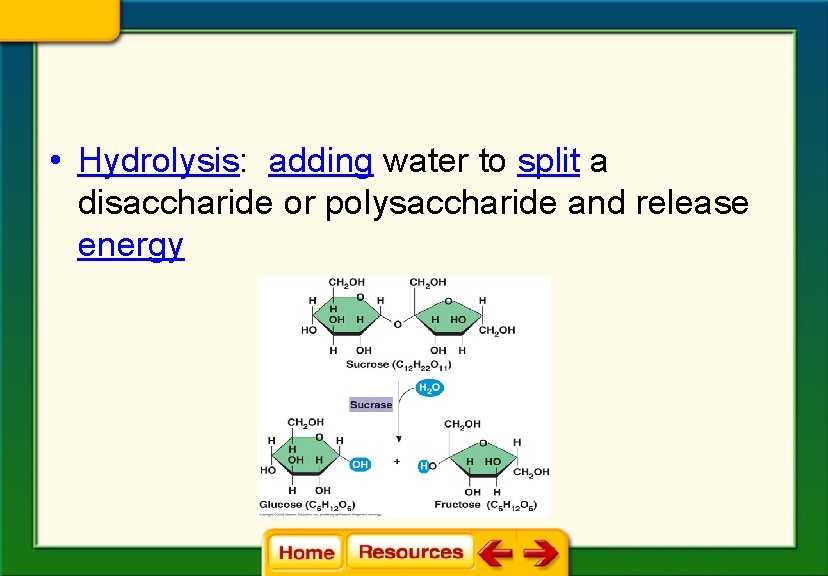
• Hydrolysis: adding water to split a disaccharide or polysaccharide and release energy

Functions of Carbohydrates: • Primary source of energy for organisms

• Structural support in plants (cellulose) • Main component in shells (chitin)

Carbohydrates Are: • Manufactured by green plants only: • Occurs during photosynthesis • 6 CO 2 + 6 H 2 O → C 6 H 12 O 6 + 6 O 2

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Lipids § Molecules made mostly of carbon and hydrogen § Make Up: §Fats §Oils §Waxes

Functions of Lipids § Primary function is to store energy § Make up cell structures § Form chemical messages between cells § Protection (wax on plant leaves)
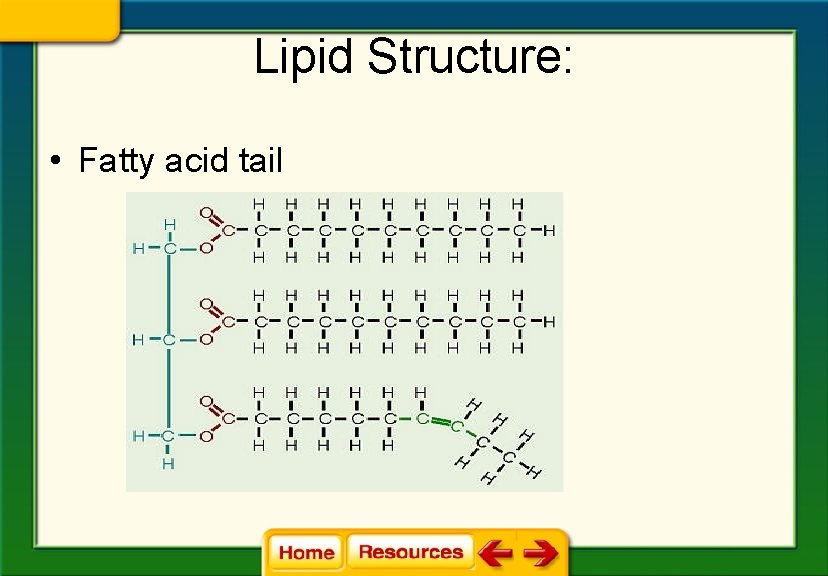
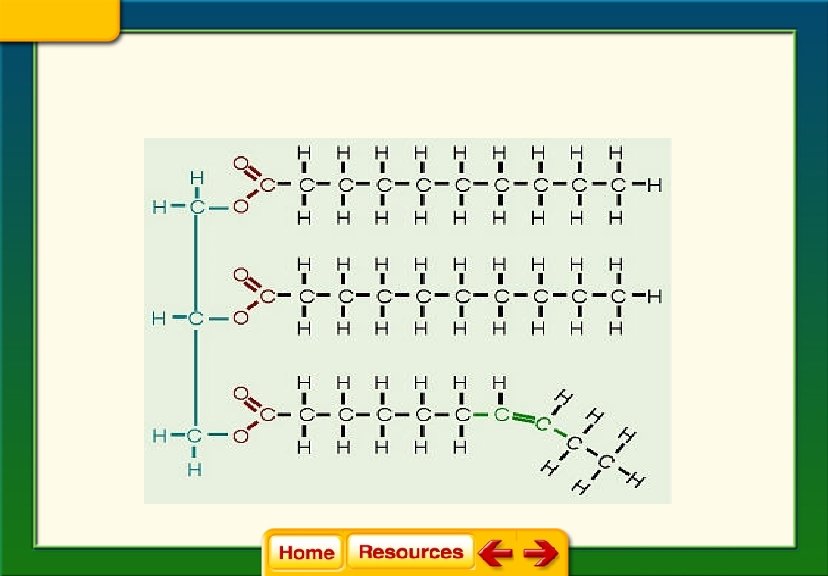
Lipid Structure: • Fatty acid tail

• Fatty acid tail is a chain of carbon atoms bonded to hydrogen atoms and other carbon atoms by single or double bonds • The bonds determine if the lipid is classified as saturated, unsaturated or polyunsaturated
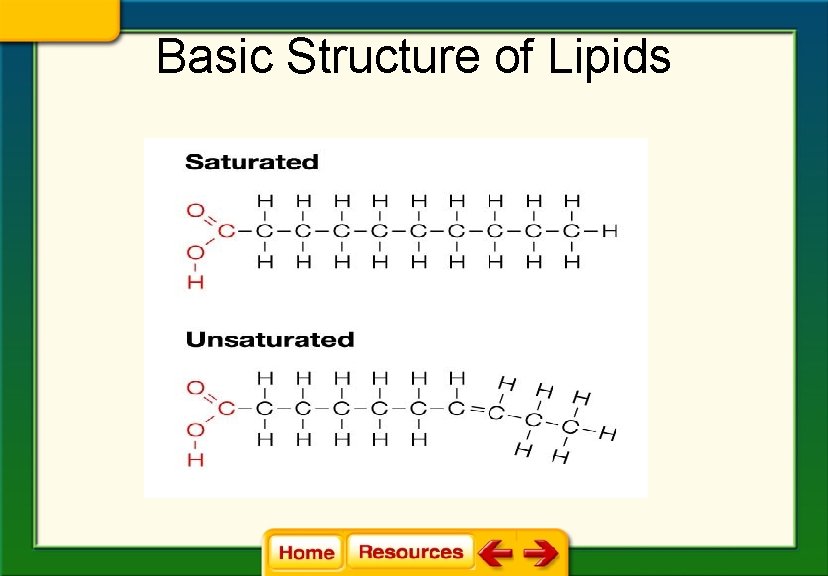
Basic Structure of Lipids
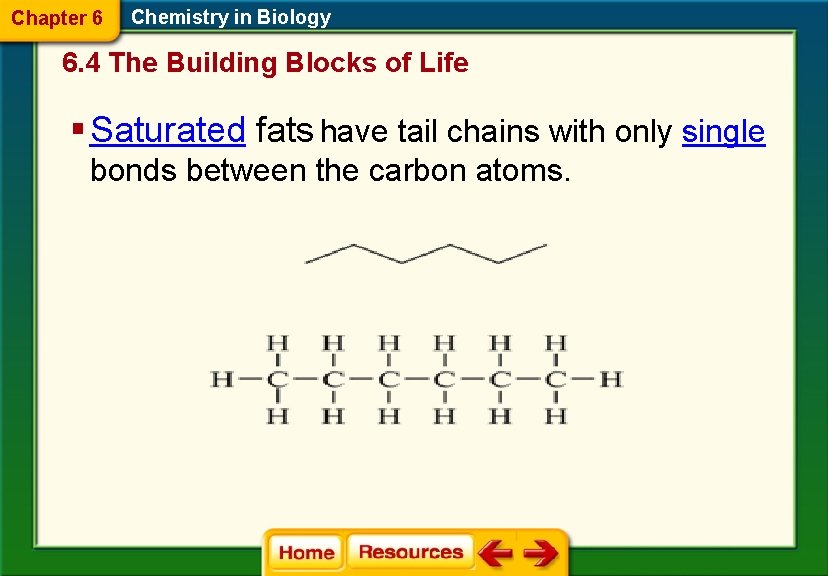
Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Saturated fats have tail chains with only single bonds between the carbon atoms.
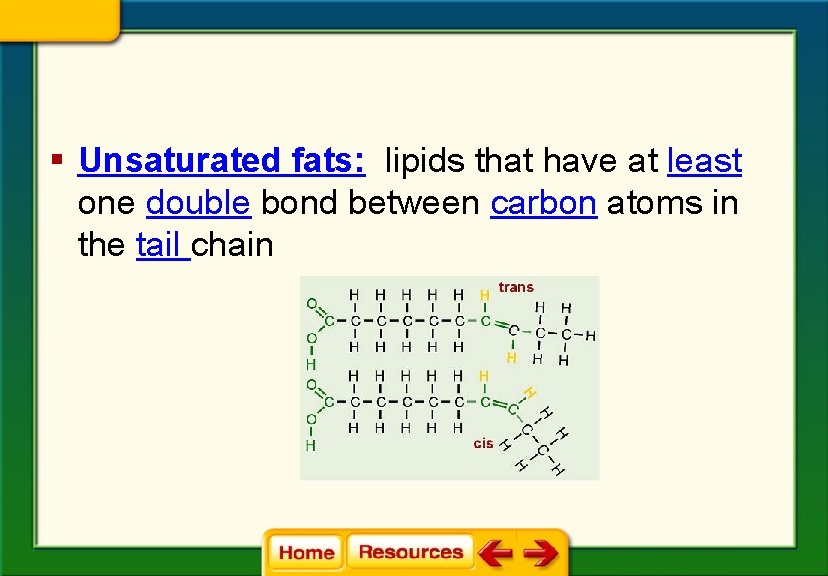
§ Unsaturated fats: lipids that have at least one double bond between carbon atoms in the tail chain

§ Fats with more than one double bond in the tail are called polyunsaturated fats.
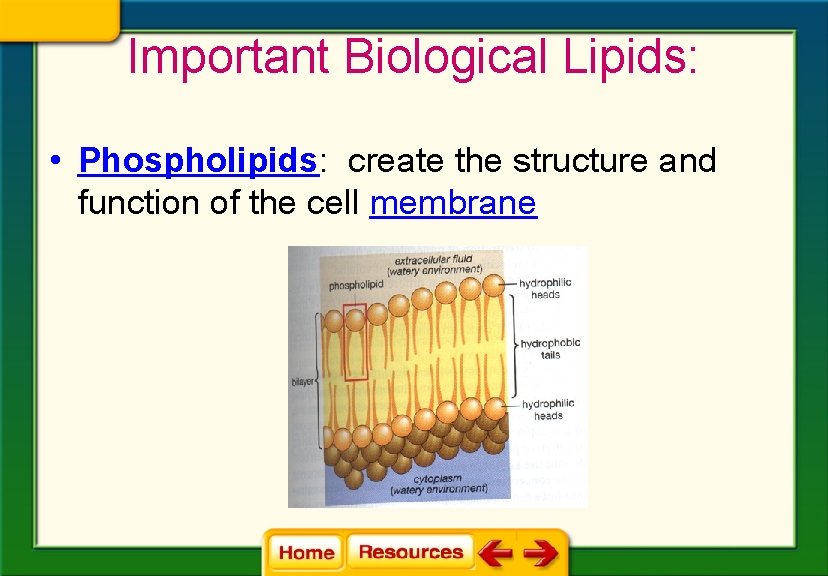
Important Biological Lipids: • Phospholipids: create the structure and function of the cell membrane

• Hydrophilic head: dissolves in water • Hydrophobic tail: does not dissolve in water • Why is this important?
Lipids • Also, responsible for the formation of important steroids including hormones and cholesterol

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life Protein § A compound made of small carbon compounds called amino acids § Amino acids are small compounds that are made of carbon, nitrogen, oxygen, hydrogen, and sometimes sulfur.

Chapter 6 Chemistry in Biology 6. 4 The Building Blocks of Life § Amino acids have a central carbon atom. § One of the four carbon bonds is with hydrogen. § The other three bonds are with an amino group (–NH 2), a carboxyl group (–COOH), and a variable group (–R).
Chapter 6 Chemistry in Biology
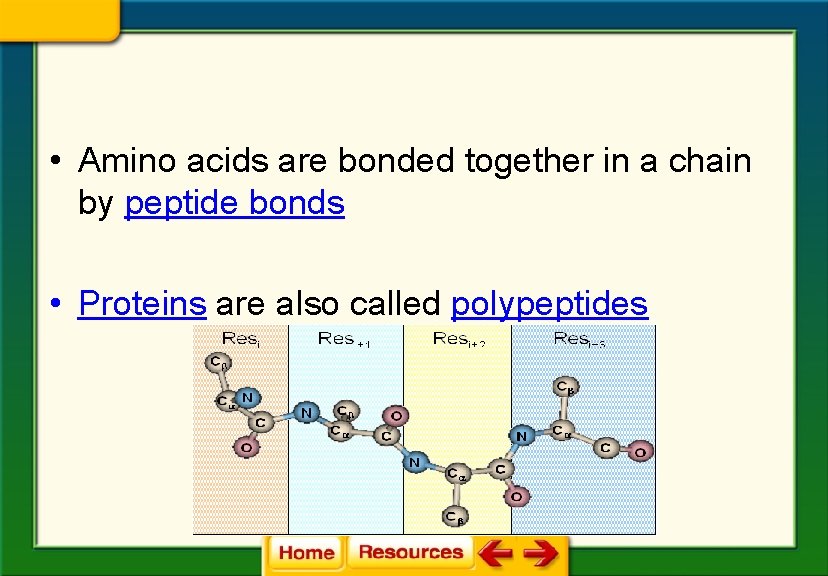
• Amino acids are bonded together in a chain by peptide bonds • Proteins are also called polypeptides

Protein Functions: • Make up mass • Involved in almost all functions – Structure – Chemical signals between cells – Control cell growth and repair – Enzymes (biological catalysts)

Enzymes • Enzymes: biological catalysts • Made of proteins
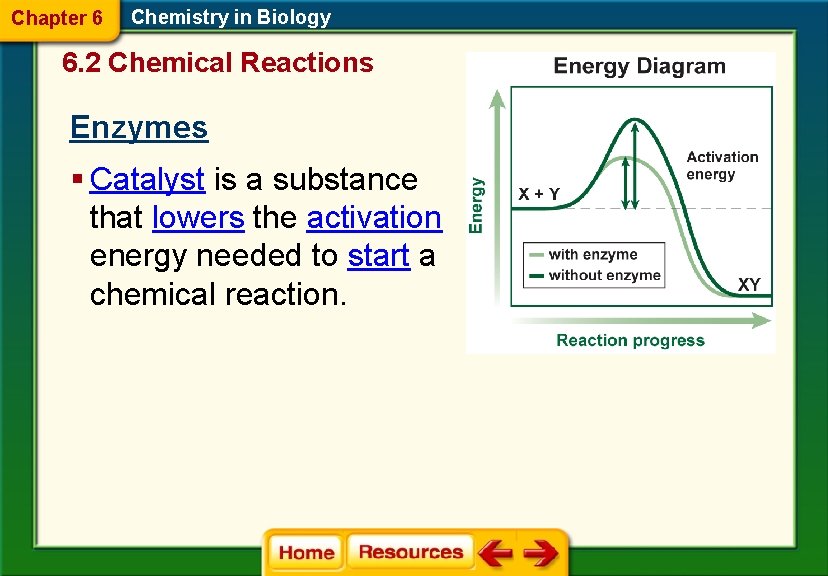
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions Enzymes § Catalyst is a substance that lowers the activation energy needed to start a chemical reaction.
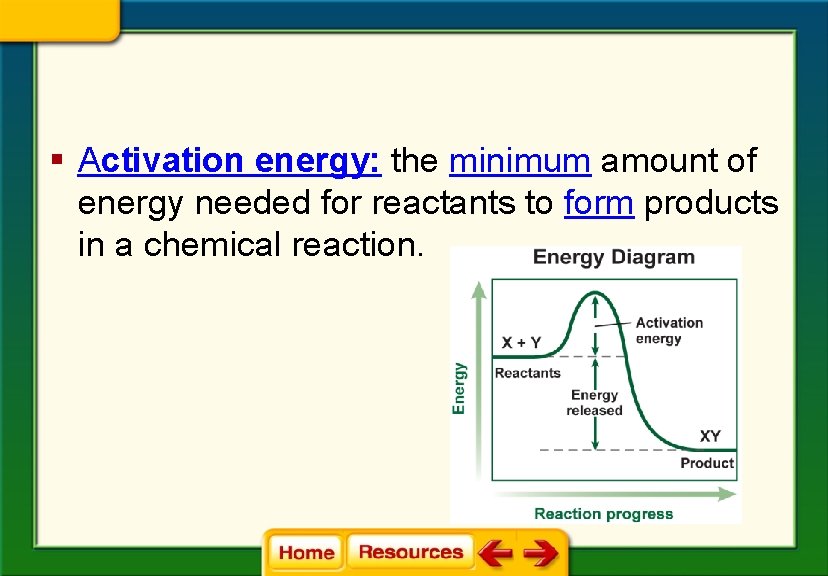
§ Activation energy: the minimum amount of energy needed for reactants to form products in a chemical reaction.

• It does not increase how much product is made. • It does not get used up in the reaction.
• How do enzymes work?
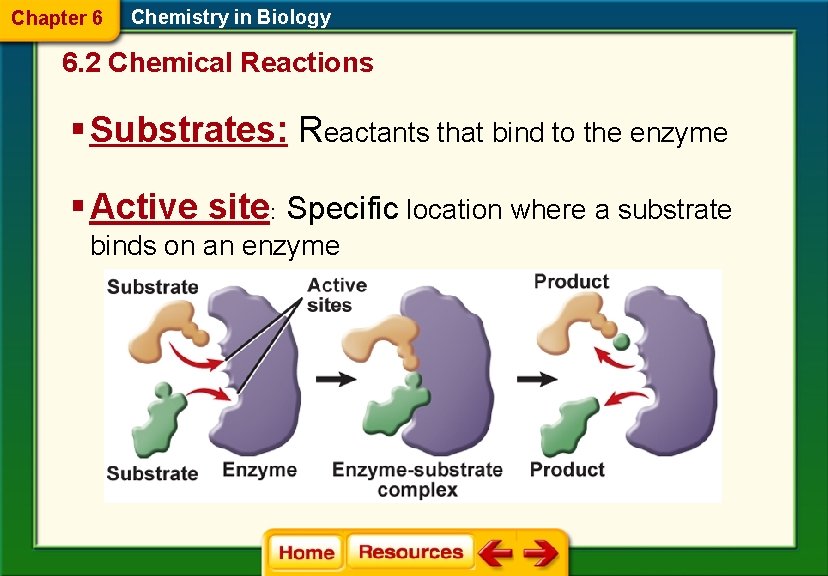
Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § Substrates: Reactants that bind to the enzyme § Active site: Specific location where a substrate binds on an enzyme
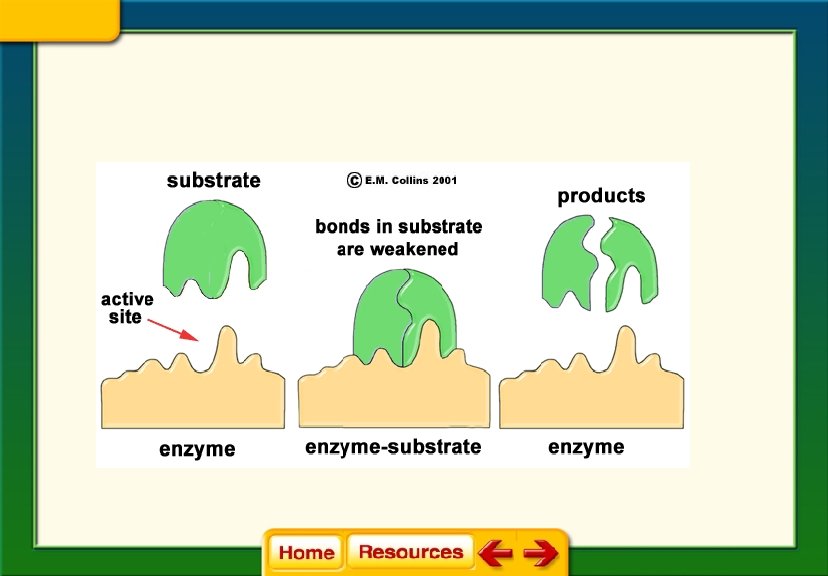

Chapter 6 Chemistry in Biology 6. 2 Chemical Reactions § The active site changes shape and forms the enzyme-substrate complex, which helps chemical bonds in the reactants to be broken and new bonds to form. § Factors such as p. H, temperature, and other substances affect enzyme activity.
Chapter 6 Chemistry in Biology

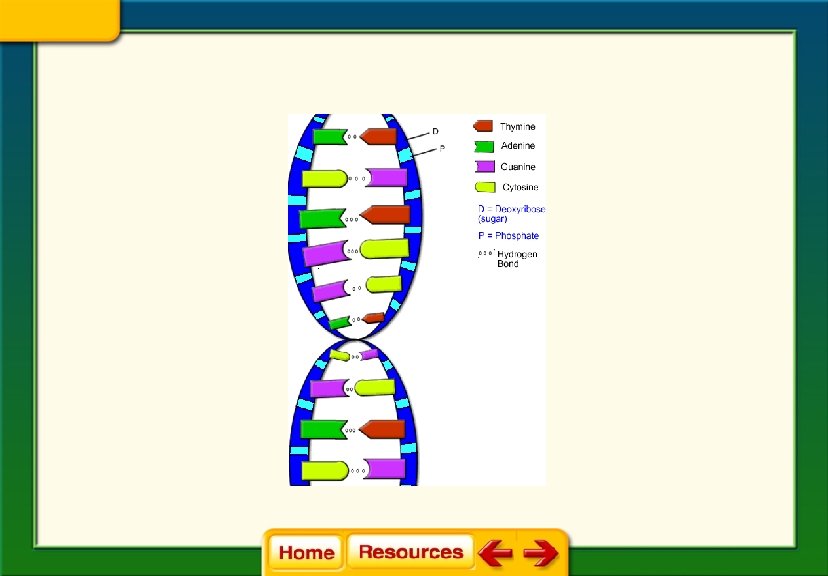
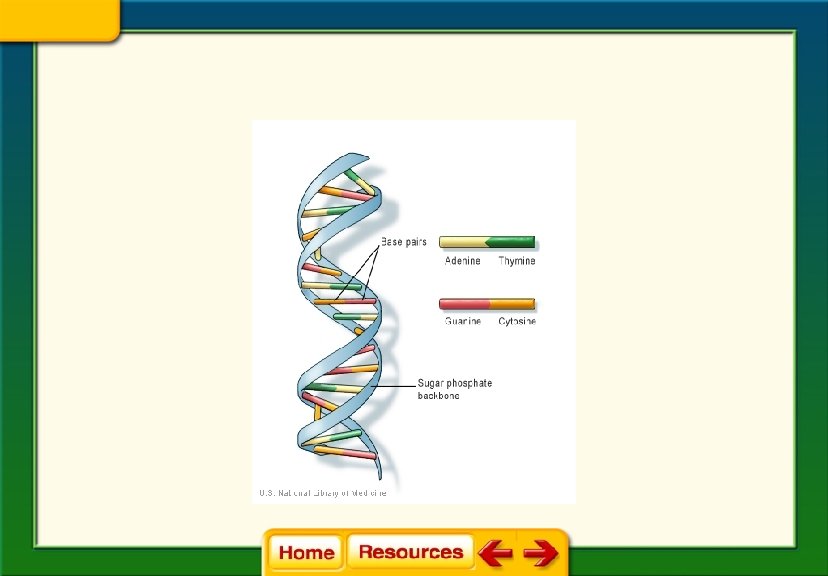
• Nucleic Acids: • Made up of smaller subunits called nucleotides • Nucleotides consist of carbon, nitrogen, oxygen, phosphorus, and hydrogen atoms

Nucleic Acid Functions: • Store and transmit genetic information • Two types of nucleic acids: – DNA – RNA
- Slides: 78