Vocabulary ION 1 atom monatomic Ion ex Cl
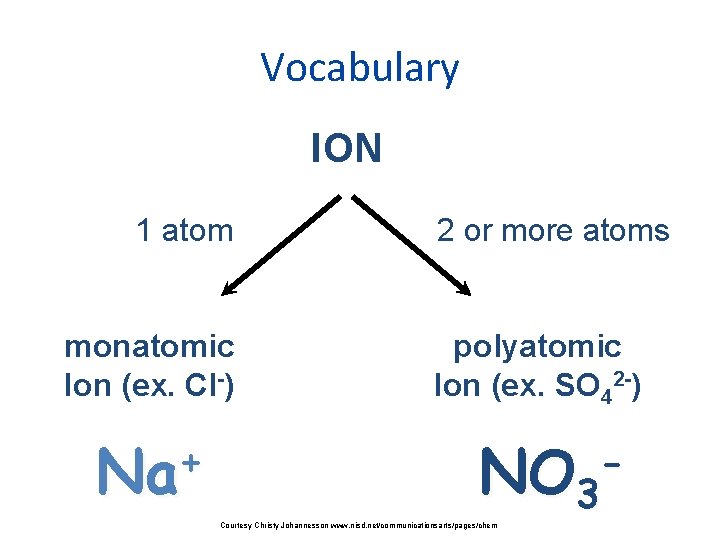
Vocabulary ION 1 atom monatomic Ion (ex. Cl-) + Na 2 or more atoms polyatomic Ion (ex. SO 42 -) NO 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem
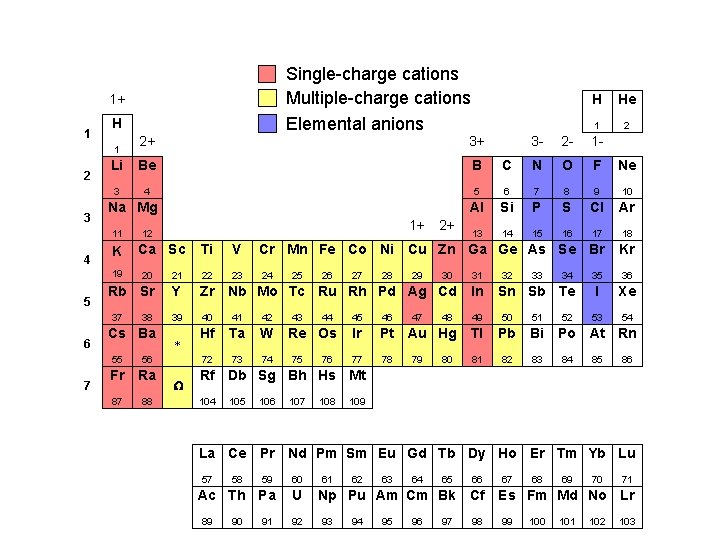
Single-charge cations Multiple-charge cations Elemental anions 1+ 1 H 2+ 3+ Li Be B 3 4 1 2 3 Na Mg 11 4 K 19 5 7 Ca Sc 2+ C N O F Ne 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 55 56 Fr Ra 87 88 * W 2 1 - 22 Cs Ba 1 2 - 21 38 He 3 - 20 37 6 1+ 12 H 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103

Cations and Anions Common Simple Cations and Anions Cation H 1+ Li 1+ Na 1+ K 1+ Cs 1+ Be 2+ Mg 2+ Al 3+ Ag 1+ Name hydrogen lithium sodium potassium cesium beryllium magnesium aluminum silver Anion H 1 F 1 Cl 1 Br 1 I 1 O 2 S 2 - Name* hydride fluoride chloride bromide iodide oxide sulfide *The root is given in color. Zumdahl, De. Coste, World of Chemistry 2002, page 86
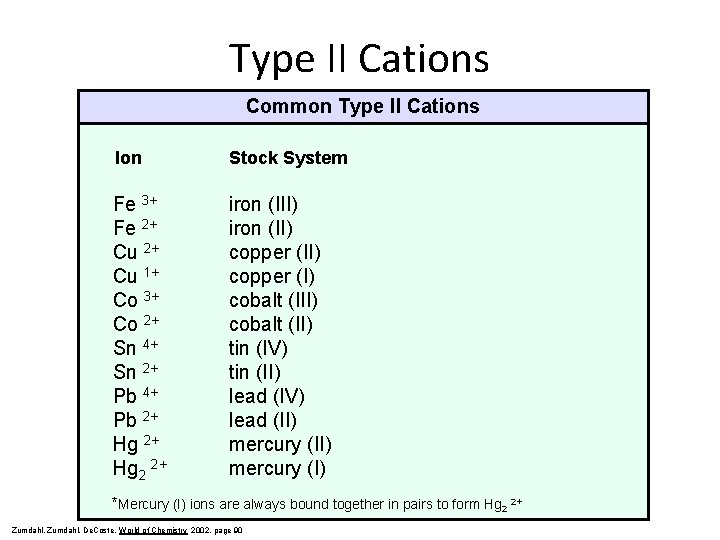
Type II Cations Common Type II Cations Ion Stock System Fe 3+ Fe 2+ Cu 1+ Co 3+ Co 2+ Sn 4+ Sn 2+ Pb 4+ Pb 2+ Hg 2 2+ iron (III) iron (II) copper (I) cobalt (II) tin (IV) tin (II) lead (IV) lead (II) mercury (I) *Mercury (I) ions are always bound together in pairs to form Hg 2 2+ Zumdahl, De. Coste, World of Chemistry 2002, page 90
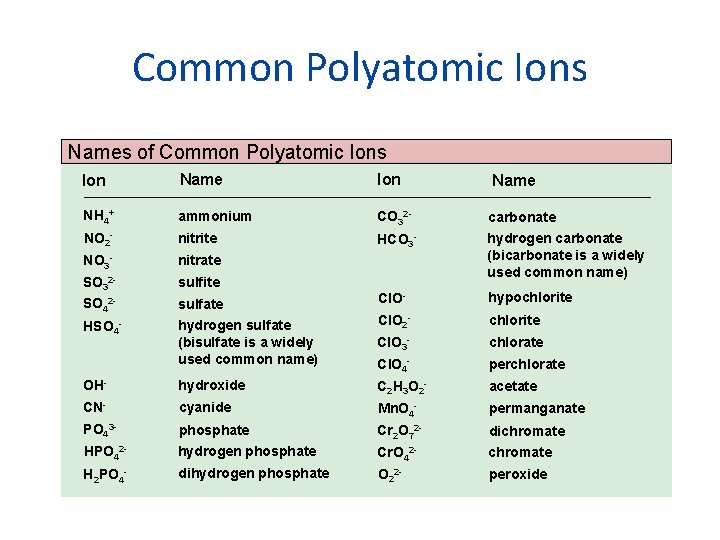
Common Polyatomic Ions Names of Common Polyatomic Ions Ion Name NH 4+ ammonium CO 32 - carbonate NO 2 - nitrite HCO 3 - NO 3 - nitrate SO 32 - sulfite hydrogen carbonate (bicarbonate is a widely used common name) SO 42 - sulfate Cl. O- hypochlorite HSO 4 - hydrogen sulfate (bisulfate is a widely used common name) Cl. O 2 - chlorite Cl. O 3 - chlorate Cl. O 4 - perchlorate OH- hydroxide C 2 H 3 O 2 - acetate CN- cyanide Mn. O 4 - permanganate PO 43 - phosphate Cr 2 O 72 - dichromate HPO 42 - hydrogen phosphate Cr. O 42 - chromate H 2 PO 4 - dihydrogen phosphate O 22 - peroxide
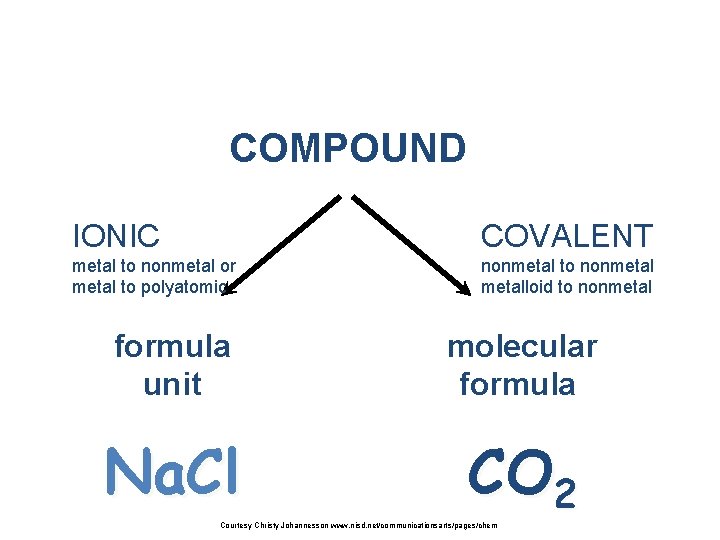
COMPOUND IONIC COVALENT metal to nonmetal or metal to polyatomic nonmetal to nonmetalloid to nonmetal formula unit molecular formula Na. Cl CO 2 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

IONIC COMPOUND ions of 2 elements (metal + non metal) more than 2 elements (metal +polyatomic) Binary (ionic) compound Na. Cl Ternary (ionic) compound Na. NO 3 Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem

Binary Ionic Compounds Binary compounds contain a metal ion (group 1, group 2, Al, Zn, Ag, etc. ), and a non-metal. To name these compounds, give the name of metal followed by the name of the non-metal, with the ending replaced by the suffix –ide. Examples: Na. Cl sodium chloride (Na 1+ Cl 1 -) Ca. S calcium sulfide (Ca 2+ Al. I 3 aluminum iodide (Al 3+ 3 I 1 -) S 2 -)

Writing Binary Ionic Compounds chemical formula (metal/non-metal): has neutral charge shows types of atoms/ions subscript shows how many of each atom/ion To write an ionic compound’s formula, we need: 1. the two types of ions 2. the charge on each ion Na 1+ and F 1– Na. F Ba 2+ and O 2– Ba. O Na 1+ and O 2– Na 2 O Ba 2+ and F 1– Ba. F 2

Naming Binary Ionic Compounds Formula Name Ba. O barium oxide __________ Na. Br 2 ________ sodium bromide 1 3 Mg. I 2 magnesium iodide __________ 4 KCl potassium chloride __________ Sr. F 2 5 ________ strontium fluoride Cs. F 6 ________ cesium fluoride

Naming Binary Ionic Compounds Formula Name 1 Hg 2 O mercury (I) oxide __________ 2 Hg. O mercury (II) oxide __________ Cu. F 2 3 ________ copper (II) fluoride Cu 2 S 4 ________ copper (I) sulfide 5 Cr 2 O 3 Pb. O 2 6 ________ chromium (III) oxide __________ lead (IV) oxide

Ternary Ionic Compounds Ternary compounds contain a metal ion (group 1, group 2, Al, Zn, Ag, etc. ), and a polyatomic ion. To name these compounds, give the name of metal followed by the name of the polyatomic ion. Examples: Na. NO 3 sodium nitrate K 2 SO 4 potassium sulfate Ca 3(PO 4)2 calcium phosphate Fe(OH)3 iron (III) hydroxide Na 2 CO 3 sodium carbonate

Naming Ternary Ionic Compounds Ca 3(PO 4) 2 1. ________ calcium phosphate (NH 4)2 CO 3 2. ________ ammonium carbonate Al 2(SO 4)3 3. ________ aluminum sulfate 4. Na 2 SO 4 sodium sulfate __________ 5. Li. CN lithium cyanide __________ 6. Ba 2 CO 3 barium carbonate __________ Cu(OH)2 7. ________ copper (II) hydroxide

Writing Ternary Ionic Compounds Parentheses are required only when you need more than one “bunch” of a particular polyatomic ion. Ba 2+ and SO 42– Ba. SO 4 barium sulfate Mg 2+ and NO 31– Mg(NO 3)2 magnesium nitrate NH 41+ and PO 43– (NH 4)3 PO 3 ammonium phosphate Sn 4+ and SO 42– Sn(SO 4)2 tin (IV) sulfate Fe 3+ and CO 32– Fe 2(CO 3)3 iron (III) carbonate NH 41+ and OH 1– NH 4 OH ammonium hydroxide
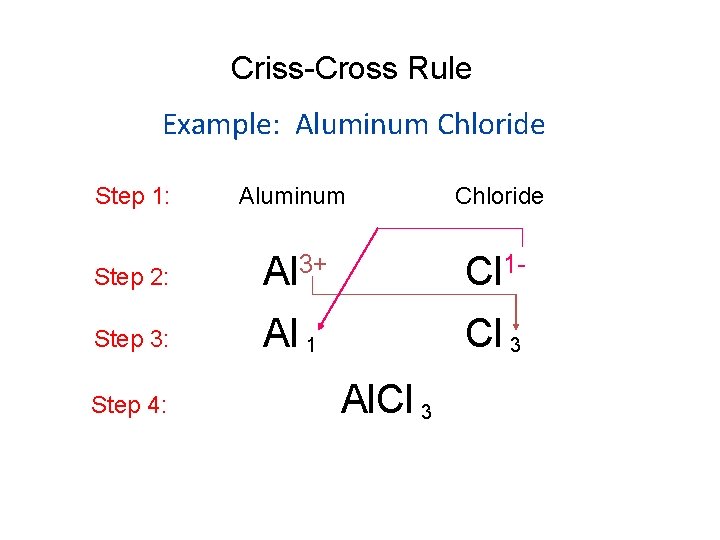
Criss-Cross Rule Example: Aluminum Chloride Step 1: Aluminum Chloride Step 2: Al 3+ Cl 1 - Step 3: Al 1 Cl 3 Step 4: Al. Cl 3
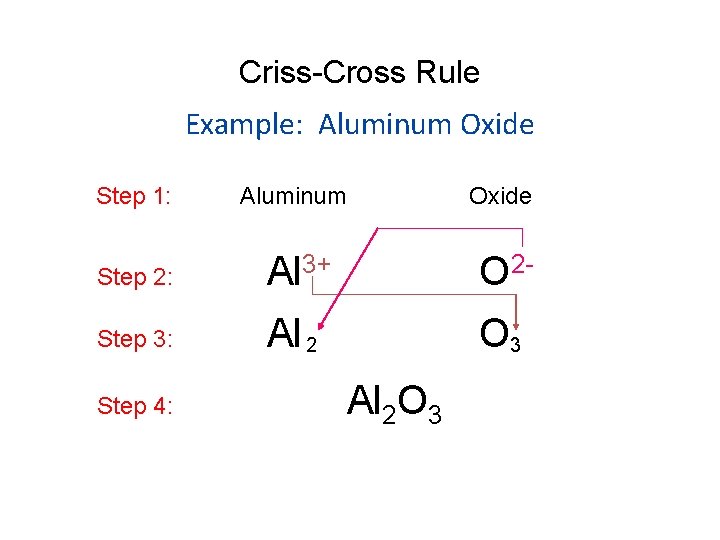
Criss-Cross Rule Example: Aluminum Oxide Step 1: Aluminum Oxide Step 2: Al 3+ O 2 - Step 3: Al 2 O 3 Step 4: Al 2 O 3
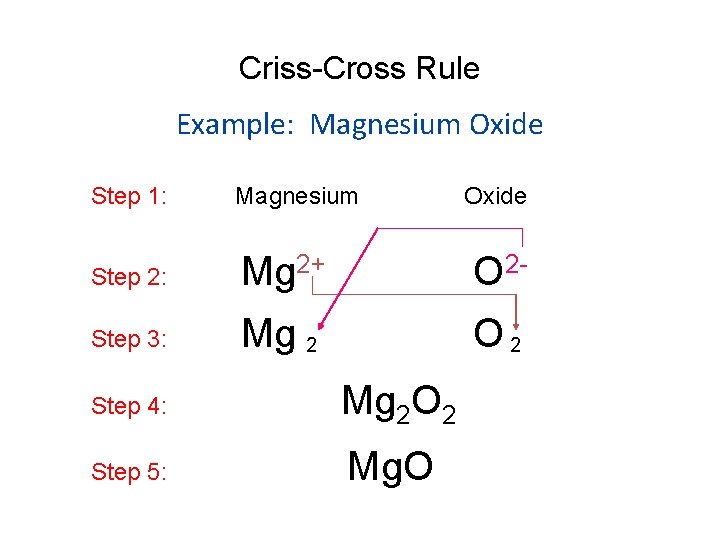
Criss-Cross Rule Example: Magnesium Oxide Step 1: Magnesium Step 2: Mg 2+ O 2 - Step 3: Mg 2 O 2 Step 4: Mg 2 O 2 Step 5: Mg. O Oxide

Writing Formulas/Names of Covalent Compounds contain two types of nonmetals, or metalloid and nonmetal (NEVER metals) Key: FORGET CHARGES What to do: Use Greek prefixes to indicate how many atoms of each element, but don’t use “mono” on first element. 1 – mono 2 – di 3 – tri 4 – tetra 5 – penta 6 – hexa 7 – hepta 8 – octa 9 – nona 10 – deca

Binary Covalent Compounds N 2 O 3 N 2 O 5 dinitrogen monoxide dinitrogen trioxide dinitrogen pentoxide ICl 3 iodine monochloride iodine trichloride SO 2 SO 3 sulfur dioxide sulfur trioxide

Writing Formulas/Names of Covalent Compounds EXAMPLES: carbon dioxide CO dinitrogen trioxide N 2 O 5 carbon tetrachloride NI 3 CO 2 carbon monoxide N 2 O 3 dinitrogen pentoxide CCl 4 nitrogen triiodide
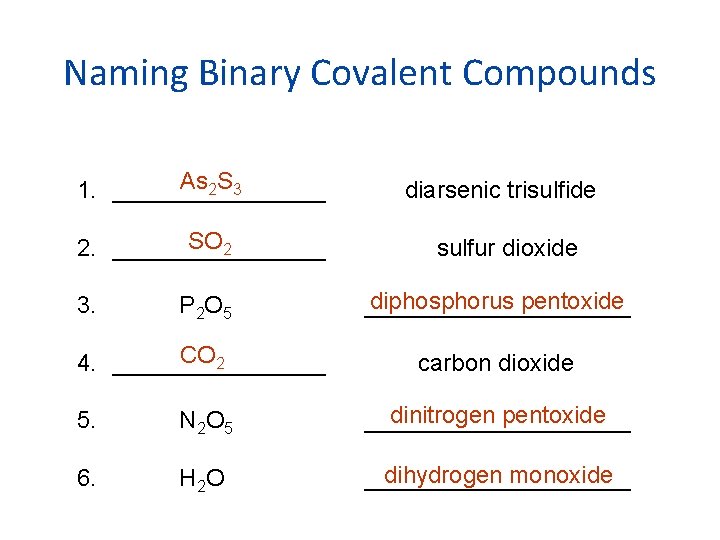
Naming Binary Covalent Compounds As 2 S 3 1. ________ diarsenic trisulfide SO 2 2. ________ sulfur dioxide P 2 O 5 diphosphorus pentoxide __________ CO 2 4. ________ carbon dioxide 3. 5. N 2 O 5 dinitrogen pentoxide __________ 6. H 2 O dihydrogen monoxide __________

Exceptions! Two exceptions to the simple –ide ending are the diatomic oxide ions, O 22 - and O 21 -. O 22 - is called peroxide Note the differences. O 21 - is called superoxide. barium oxide barium peroxide Ba. O _____ Ba. O 2 _____ sodium oxide sodium peroxide Na 2 O _____ Na 2 O 2 _____ potassium oxide potassium superoxide K 2 O _____ KO 2 _____ Ba 2+ Na 1+ Do Not Reduce to lowest terms! K 1+

Binary Hydrogen Compounds of Nonmetals When Dissolved in Water (symbol “aq”) (These compounds are commonly called acids. ) The prefix hydro- is used to represent hydrogen, followed by the name of the nonmetal with its ending replaced by the suffix –ic and the word Acid added. Examples: *HCl Hydrochloric acid HBr Hydrobromic acid *The name of this compound would be hydrogen monochloride if it was NOT dissolved in water.
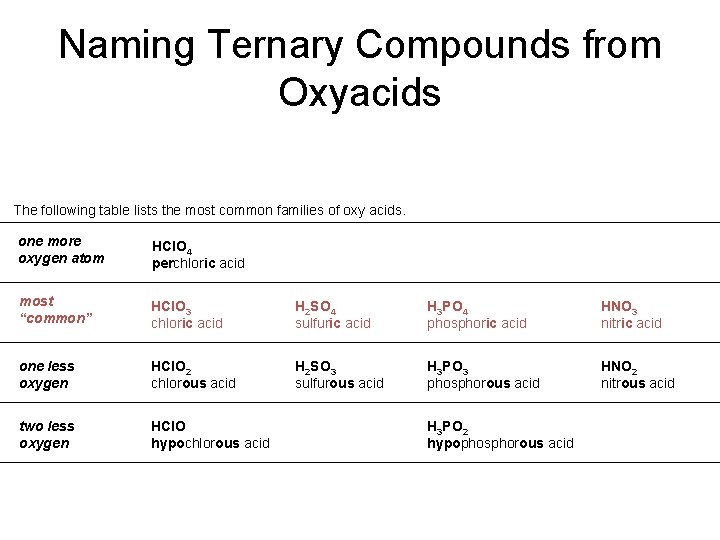
Naming Ternary Compounds from Oxyacids The following table lists the most common families of oxy acids. one more oxygen atom HCl. O 4 perchloric acid most “common” HCl. O 3 chloric acid H 2 SO 4 sulfuric acid H 3 PO 4 phosphoric acid HNO 3 nitric acid one less oxygen HCl. O 2 chlorous acid H 2 SO 3 sulfurous acid H 3 PO 3 phosphorous acid HNO 2 nitrous acid two less oxygen HCl. O hypochlorous acid H 3 PO 2 hypophosphorous acid
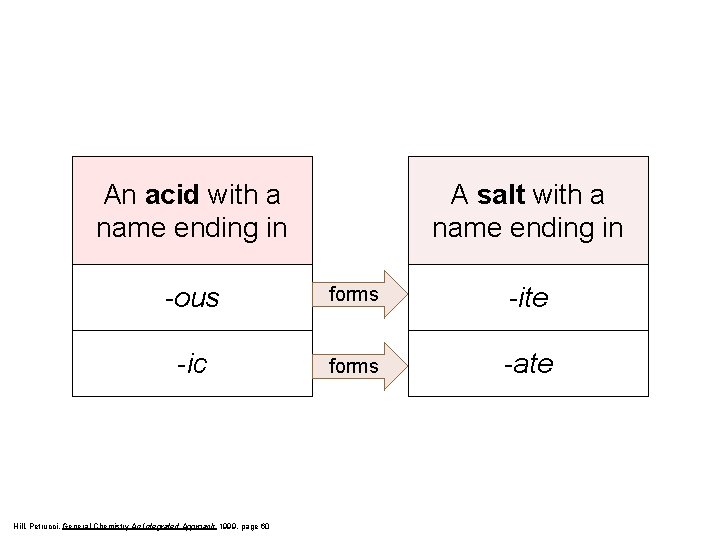
An acid with a name ending in A salt with a name ending in -ous forms -ite -ic forms -ate Hill, Petrucci, General Chemistry An Integrated Approach 1999, page 60

Oxyacids Oxysalts If you replace hydrogen with a metal (in a ternary acid), you have formed an oxysalt. A salt is an ionic compound consisting of a metal and a nonmetal. If the salt consists of a metal, a nonmetal, and oxygen it is called an oxysalt. Na. Cl. O 4, sodium perchlorate, is an oxysalt. OXYACID OXYSALT HCl. O 4 perchloric acid Na. Cl. O 4 sodium perchlorate HCl. O 3 chloric acid Na. Cl. O 3 sodium chlorate HCl. O 2 chlorous acid Na. Cl. O 2 sodium chlorite HCl. O hypochlorous acid Na. Cl. O sodium hypochlorite
- Slides: 26