Bonding Chemical Formulas Valence Electrons Dot Structures Ions

Bonding & Chemical Formulas

Valence Electrons, Dot Structures & Ions

I. Valence electrons • Valence e- are located in the highest (outermost) energy level • Valence e- are responsible for the chemical & physical properties of an element • Valence e- are used in forming chemical bonds which form compounds • Valence e- are responsible for the charge and type of ion formed

Examples 1. Group 1 elements 1 valence e 2. Group 2 elements 2 valence e 3. Group 17 elements 7 valence e 4. Group 18 elements 8 valence e 5. In general all transition elements (d & f) will have 2 valence e-. . . Why?

II. Electron (Lewis) Dot Structures are a way of representing valence electrons Grp 1 Grp 2 Grp 13 Grp 14 Li Be B C Grp 15 Grp 16 Grp 17 Grp 18 N O F Ne

The noble gas configuration with 8 valence eis a very stable condition that all atoms try to attain in reactions Example Na Cl + Na Cl - Octet - the condition with 8 valence e-

III. Formation of ions • Ions are charged particles, either (+) positive or (-) negative • Ions are formed when electrons are either lost or gained during bonding 1. Anions - are negative ions formed when atoms gain electrons 2. Cations - are positive ions formed when atoms lose electrons + +

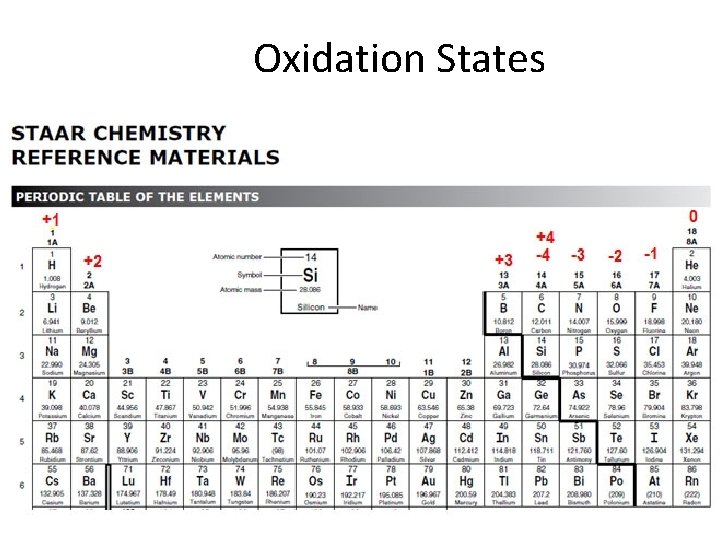
• Atoms tend to form octets in their outer shells by gaining or losing electrons • Generalizations about groups & ions 1. Group 1 atoms lose 1 e- & form +1 ions 2. Group 2 atoms lose 2 e- & form +2 ions 3. Group 13 atoms lose 3 e- & form +3 ions 4. Group 14 atoms lose 4 e- & form +4 ions gain 4 e- & form -4 ions 4. Group 15 atoms gain 3 e- & form -3 ions 5. Group 16 atoms gain 2 e- & form -2 ions 6. Group 17 atoms gain 1 e- & form -1 ions ** These groups always form these ions, and you must know this from this point on!!!!!
Oxidation States
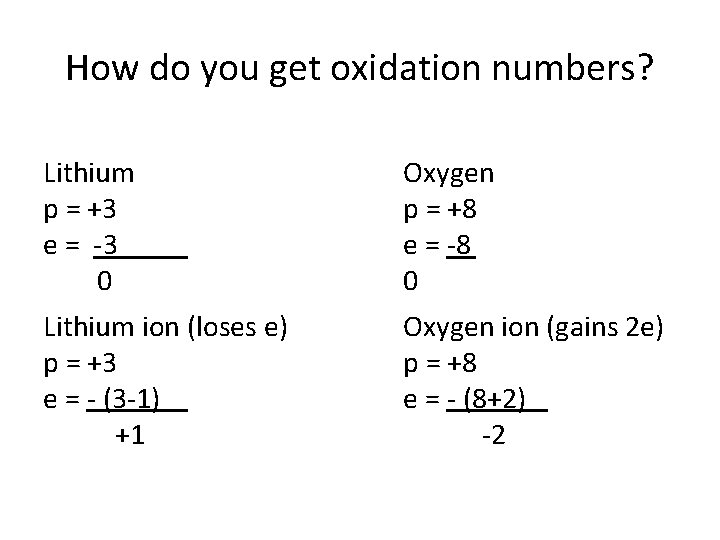
How do you get oxidation numbers? Lithium p = +3 e = -3 0 Oxygen p = +8 e = -8 0 Lithium ion (loses e) p = +3 e = - (3 -1) +1 Oxygen ion (gains 2 e) p = +8 e = - (8+2) -2

• Ions must be written with their charge when written by themselves, and may or may not show charges when in a compound formula Mg+2 Cl 1 Mg+2 Cl-1 Mg. Cl 2

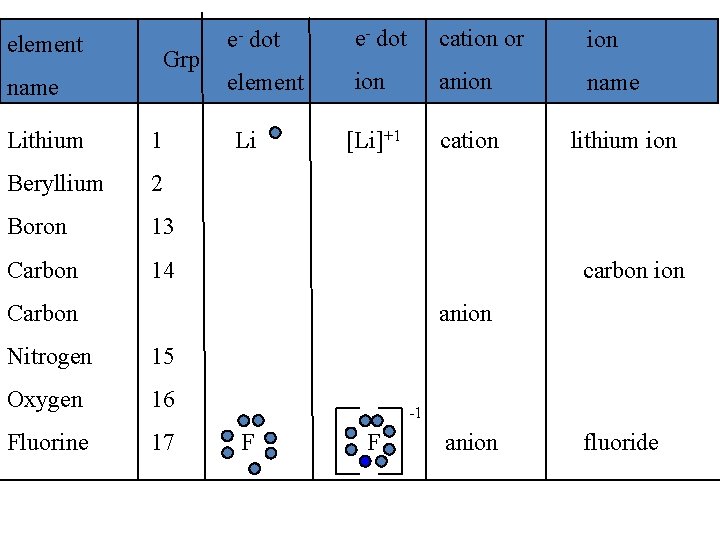
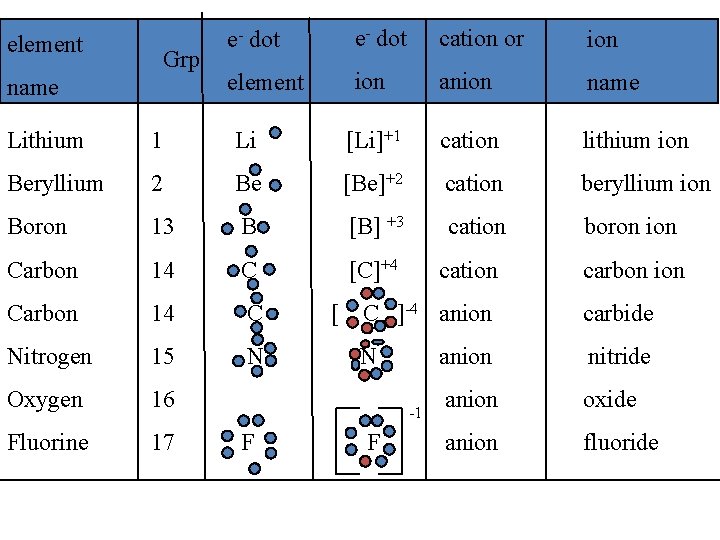
IV. Types of ions A. Monatomic ions - ions that consist of only one atom 1. Naming positive monatomic ions: most metals &/or positive ion names are the same as the element name w/ ion Na element sodium Na+ ion sodium ion

• Naming negative monatomic ions: most non-metals &/or negative ions replace the ending of element name with -ide Cl element chlorine Cl- ion chloride • Hydrogen can form either a + or a – ion H+1 = Hydrogen ion H-1 = Hydride

element Grp name Lithium 1 Beryllium 2 Boron 13 Carbon 14 e- dot cation or ion element ion anion name [Li]+1 cation Li lithium ion carbon ion Carbon anion Nitrogen 15 Oxygen 16 Fluorine 17 -1 F F anion fluoride
element Grp name e- dot cation or ion element ion anion name Lithium 1 Li [Li]+1 cation lithium ion Beryllium 2 Be [Be]+2 cation beryllium ion Boron 13 B [B] +3 cation boron ion Carbon 14 C [C]+4 Carbon 14 C Nitrogen 15 N Oxygen 16 Fluorine 17 [ cation C ]-4 anion carbide N anion nitride anion oxide anion fluoride -1 F carbon ion F

V. Intramolecular Forces (“intra” - within) A. Intramolecular forces hold atoms together to form a compound Also known as Chemical Bonds
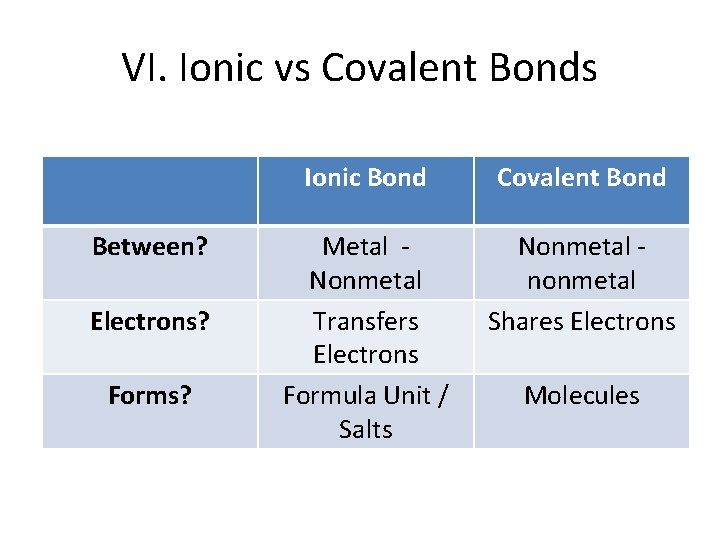
VI. Ionic vs Covalent Bonds Between? Electrons? Forms? Ionic Bond Covalent Bond Metal - Nonmetal Transfers Electrons Formula Unit / Salts Nonmetal - nonmetal Shares Electrons Molecules

Characteristics Covalent Compounds Ionic Compounds 1. Gases, liquids, or solids 1. Crystalline solids (made of molecules) of ions) 2. Low melting and boiling 2. High melting and boiling points 3. Poor electrical 3. Conduct electricity when conductors in all phases melted 4. Many soluble in non 4. Many soluble in water polar liquids but not in non-polar liquid water

• On worksheet question #1 • A, C, L, Q, U, V, Z, AA, BB, GG • On worksheet question #2 • A, C, F, I, J, S, GG

VII. Ionic Compounds • Ionic compounds – between a metal (cation) and a non-metal (anion) – or between a metal (cation) and a polyatomic ion (made of more than one atom but acts together). • If your compound contains a metal it's ionic. metal or H + non-metal (cation) (anion) (+) (-)
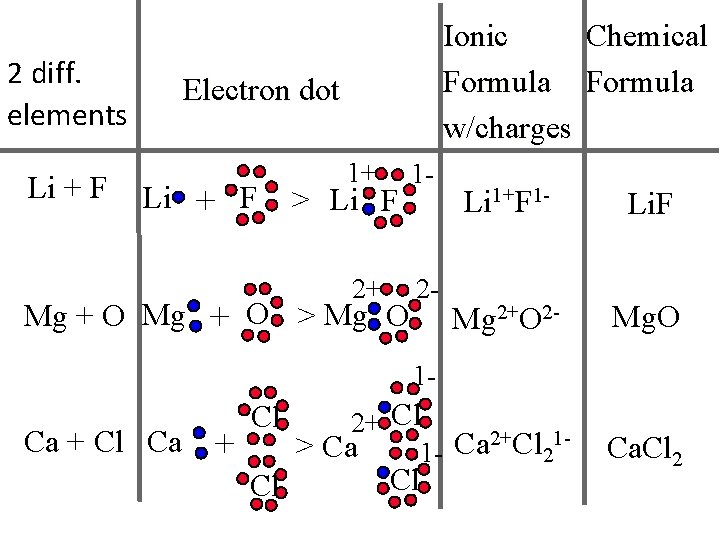
2 diff. elements Li + F Chemical Ionic Formula w/charges Electron dot Li + F 1+ 1 - 2+ 2 - > Li F Mg + O > Mg O Li 1+F 1 - Li. F Mg 2+O 2 - Mg. O 1 - Ca + Cl Cl 2+Cl 1 Ca > Ca 12 Cl Ca. Cl 2

“Criss-Cross” Method When writing formulas, the charges need to equal ZERO! 3+ 2 Al S Al 2 S 3

• Lithium's ion Li 1+bonds with oxygen's ion O 2 • • • What’s the lowest common denominator? 2 Li (+1) x 2 = 2 and Oxygen (2 -) x 1 = -2 So Li 2 O has a net charge of 0

Nomenclature of Ionic Compounds (naming) A. Binary Ionic Compounds Name consists of: 1. Cation-The metal keeps its name as listed in the Periodic Table 2. Anion- The non-metal ends in -ide (oxygen to oxide, sulfur to sulfide, chlorine to chloride, etc. ) Ex: Na. Cl sodium chloride Mg. Br 2 magnesium bromide

WORKSHEET # 2 • On worksheet questions PART 1 • 1, 2, 3, 4, 5, 6, 7, and 8 both right and left – Silver always +1 – Zinc is +2 • And the big square on worksheet 2 – Skip Au on chart– just draw a line through it.

Polyatomic Ions • Polyatomic ions are made up of more than one element BUT they behave/act as one element. • polyatomic ion as a whole has a charge • Examples – NO 31 - (the nitrate ion) – CO 32 - (the carbonate ion)

Page 215 and 220
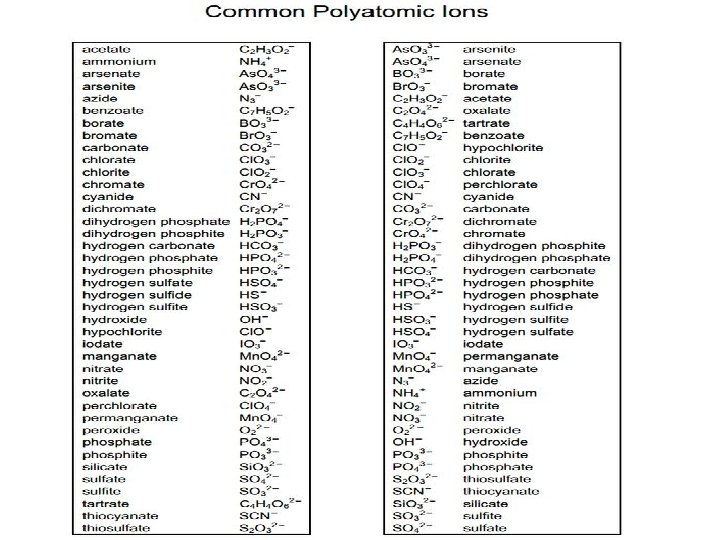

Naming Polyatomic Ions 1. The metal keeps its name as listed in the Periodic Table. 2. The polyatomic ion also keeps its name "as is. " • Mg(NO 3)2 magnesium nitrate • Li. OH lithium hydroxide **** IF you see more than 2 elements, it’s POLYATOMIC.

Worksheet side 1 • • Question 1 B, E, F, G, H, I, J, K, L, N, O, P, Q, R, T, W, X, Y BB, CC, DD, EE • Question 2 • B, D, E, G, H, K, L, M, O, P, Q, R, • T, U, V, W, AA, BB, CC, EE

Writing Formulas for Transition Elements 1. Find the charges. • The Roman Numeral tells you the charge of the cation/transition metal. • The polyatomic sheet or periodic table tells you the charge of the anion. 2. “Crisscross” charges OR find lowest common denominator. 3. Use ( ) when polyatomic ions need subscripts.

Transition Metals • REMEMBER: Because their charges never vary, silver and zinc do not require roman numerals in their names. • Ag 1+ Zn 2+ • The silver ion (Ag 1+) and chloride ion (Cl 1 -) form silver chloride, Ag. Cl. • The zinc ion (Zn 2+) and oxide ion (O 2 -) form zinc oxide, Zn. O.

Examples: Cu. Cl 2 • copper(I) chloride • copper(II) chloride Pb. O • lead(II) oxide Pb. O 2 • lead(IV) Oxide

Variable Charged Ions • Stock Naming System uses Roman numerals - I, III, IV (just learned) • Classical System uses ‘ous & ‘ic suffixes: ‘ous represents the lower charge ‘ic represents the higher charge • I/II ions with 1+ or 2+ charges – Cu 1+ (copper I, or cuprous) – Cu 2+ (copper II, or cupric) – Hg 1+ (mercury I, or mercurous) – Hg 2+ (mercury II, or mercuric)

• II/III ions with 2+ or 3+ charges – Cr 2+ – Mn 2+ – Fe 2+ – Co 2+ – Ni 2+ Cr 3+ Mn 3+ Fe 3+ Co 3+ Ni 3+ chromous/chromic manganous/manganic ferrous / ferric cobaltous/cobaltic nickelous/nickelic • II/IV ions with 2+ or 4+ charges – Sn 2+ – Pb 2+ Sn 4+ stannous / stannic Pb 4+ plumbous / plumbic • I/III ions with 1+ or 3+ charges – Au 1+ Au 3+ (aurous/auric)


WORKSHEET 2 • PART 2 • 1 -8 ON RIGHT AND LEFT

VII. Covalent Bonds

Types of Covalent Bonds • Polar – uneven distribution of charge; covalent bond in which the bonded atoms have an unequal attraction for the shared electrons • Nonpolar – equal distribution of charge; bonding electrons are shared equally.


Covalent bonds or Binary molecular compounds A. composed of 2 non-metallic elements B. Naming and writing formulas are different from ionic compounds b/c: Electrons are shared (covalent bond) rather than transferred (ionic bond) C. Ionic charges are not used in writing formulas.

Covalent Bonds D. Naming and writing formulas covalent compounds: 1. First element: Use prefixes for identifying the # of each atom in the compound. Omit prefix if only one is present 2. Second element always has a prefix, and the name ends in -ide 3. The “o” at the end of the prefix mono and “a” at end of prefix are dropped if the element is oxygen: monoxide not monooxide

4. Prefixes mono tri penta hepta nona 1 3 5 7 9 di tetra hexa octa deca 2 4 6 8 10
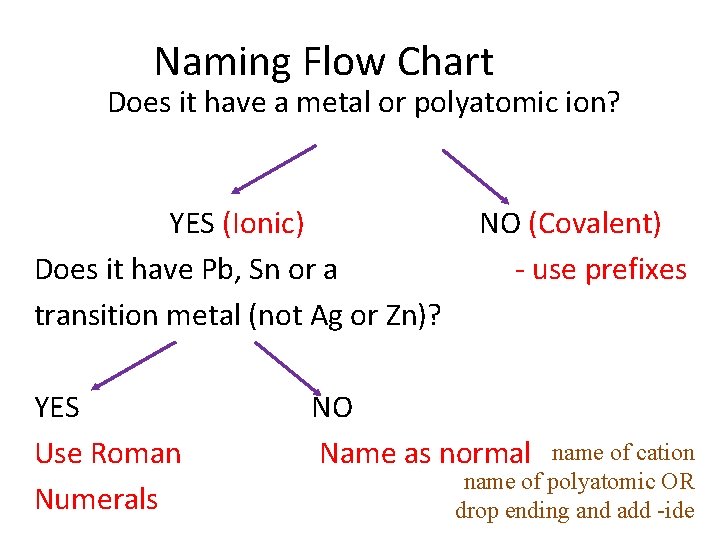
Naming Flow Chart Does it have a metal or polyatomic ion? YES (Ionic) NO (Covalent) Does it have Pb, Sn or a - use prefixes transition metal (not Ag or Zn)? YES NO Use Roman Name as normal name of cation name of polyatomic OR Numerals drop ending and add -ide

Dog’s and Chemical Bonds https: //www. youtube. com/watch? v=_M 9 khs 87 x. Q 8

VIII. Metallic Bonds • The chemical bonding that results from the attraction between metal atoms and the surrounding sea of electrons • This freedom of e- accounts for the high electrical and thermal conductivity of metals.

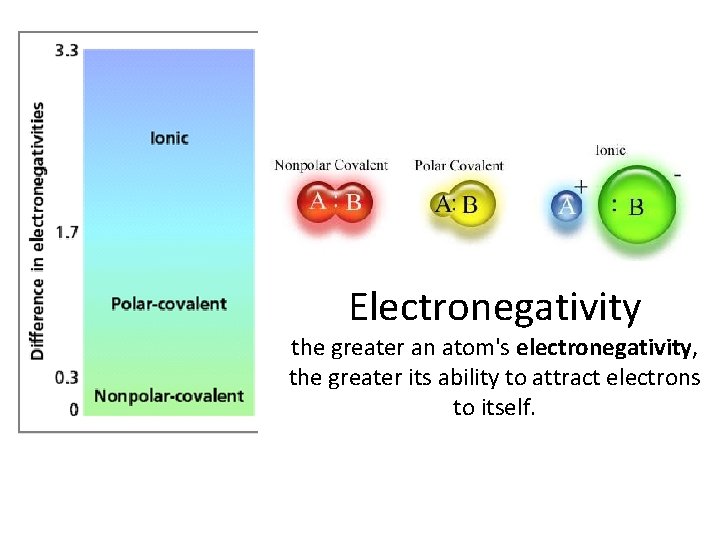
Electronegativity the greater an atom's electronegativity, the greater its ability to attract electrons to itself.

Electronegativity • Where are the most electronegative elements? • Where are the leas electronegative elements?

Formula for Electronegativity Δ electronegativity = most e negative – least e negative
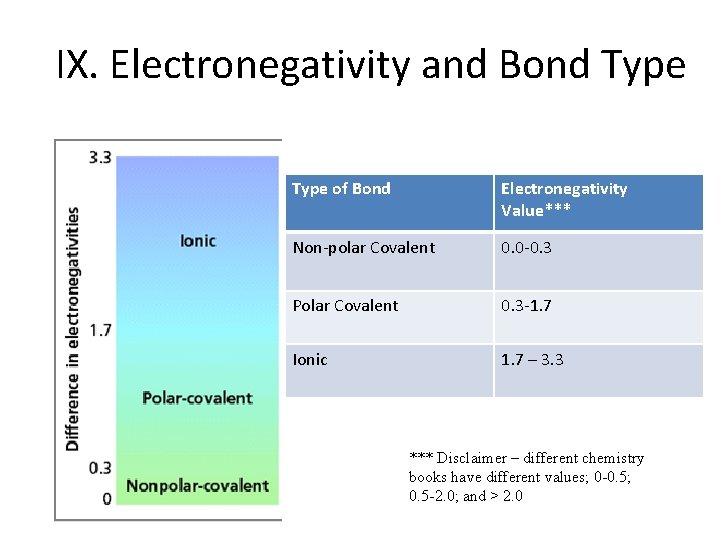
IX. Electronegativity and Bond Type of Bond Electronegativity Value*** Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 3 -1. 7 Ionic 1. 7 – 3. 3 *** Disclaimer – different chemistry books have different values; 0 -0. 5; 0. 5 -2. 0; and > 2. 0
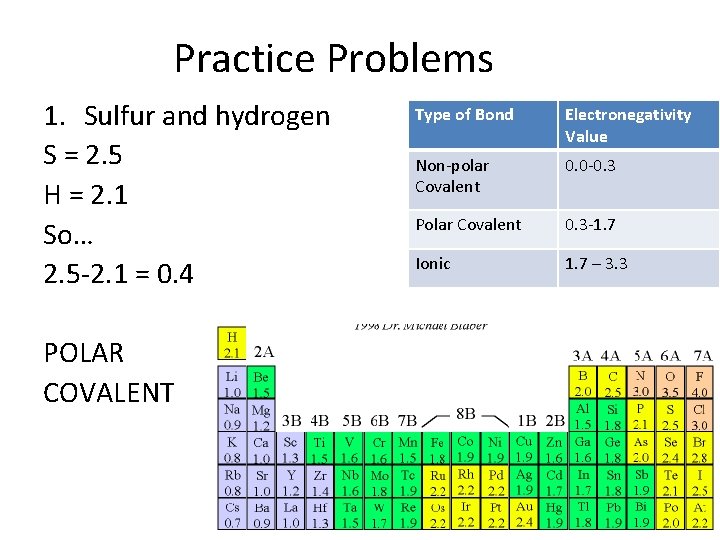
Practice Problems 1. Sulfur and hydrogen S = 2. 5 H = 2. 1 So… 2. 5 -2. 1 = 0. 4 POLAR COVALENT Type of Bond Electronegativity Value Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 3 -1. 7 Ionic 1. 7 – 3. 3

Practice Problems 2. Sulfur and cesium S = 2. 5 Cs = 0. 7 So… 2. 5 -0. 7 = 1. 8 IONIC Type of Bond Electronegativity Value Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 3 -1. 7 Ionic 1. 7 – 3. 3

Practice Problems 3. Sulfur and Chlorine S = 2. 5 Cs = 3. 0 So… 3. 0 - 2. 5 = 0. 5 POLAR COVALENT Type of Bond Electronegativity Value Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 31 -1. 7 Ionic > 1. 7
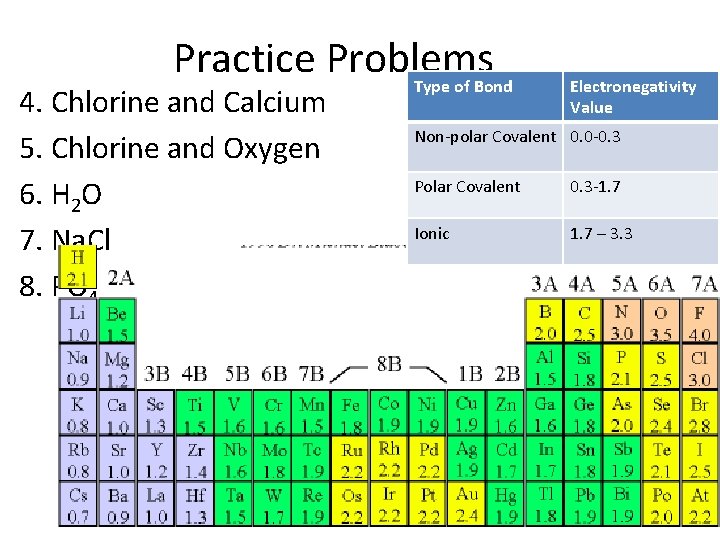
Practice Problems Type of Bond 4. Chlorine and Calcium 5. Chlorine and Oxygen 6. H 2 O 7. Na. Cl 8. PO 4 Electronegativity Value Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 3 -1. 7 Ionic 1. 7 – 3. 3
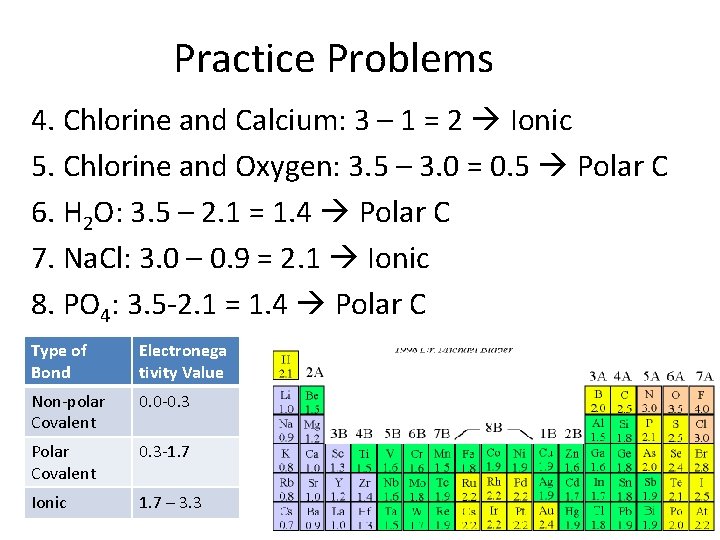
Practice Problems 4. Chlorine and Calcium: 3 – 1 = 2 Ionic 5. Chlorine and Oxygen: 3. 5 – 3. 0 = 0. 5 Polar C 6. H 2 O: 3. 5 – 2. 1 = 1. 4 Polar C 7. Na. Cl: 3. 0 – 0. 9 = 2. 1 Ionic 8. PO 4: 3. 5 -2. 1 = 1. 4 Polar C Type of Bond Electronega tivity Value Non-polar Covalent 0. 0 -0. 3 Polar Covalent 0. 3 -1. 7 Ionic 1. 7 – 3. 3
- Slides: 57