Indicaciones de quimioterapia adyuvante en cncer de endometrio

















- Slides: 64

Indicaciones de quimioterapia adyuvante en cáncer de endometrio Nuria Lainez Milagro Complejo Hospitalario de Navarra Complejo hospitalario de Navarra

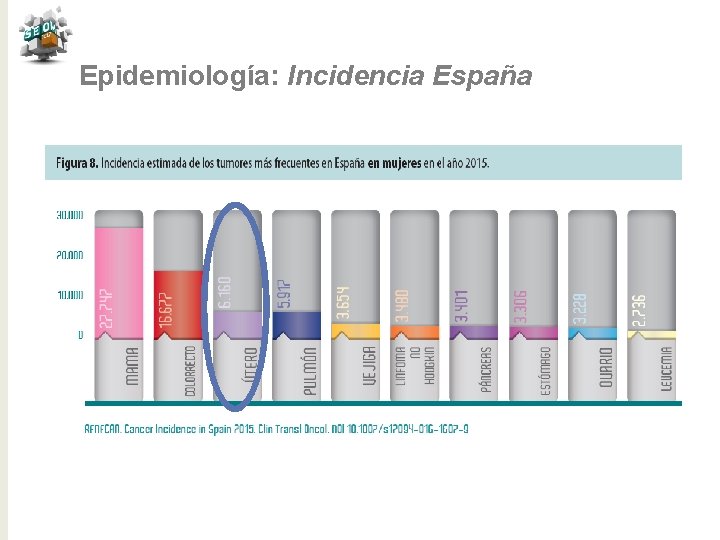
Epidemiología: Incidencia España

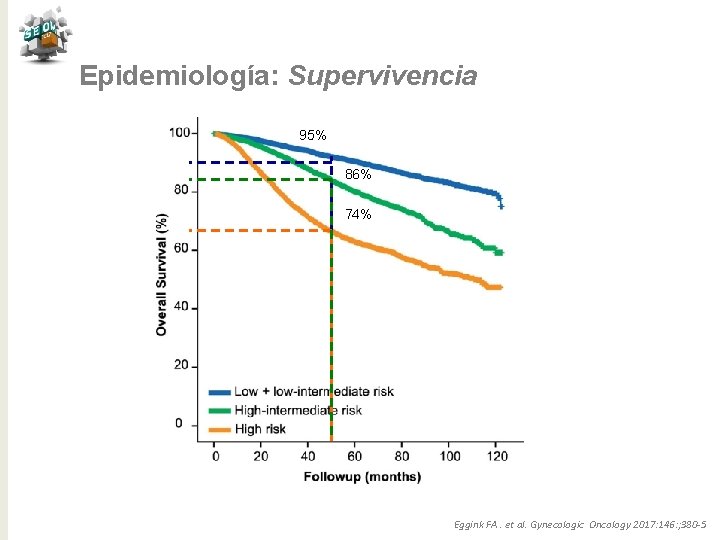
Epidemiología: Supervivencia 95% 86% 74% Eggink FA. et al. Gynecologic Oncology 2017: 146: ; 380 -5

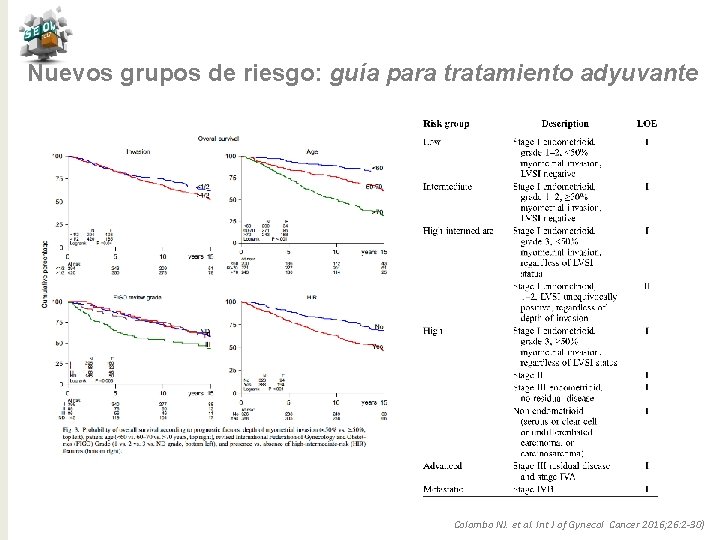
Nuevos grupos de riesgo: guía para tratamiento adyuvante Colombo NJ. et al. Int J of Gynecol Cancer 2016; 26: 2 -30)

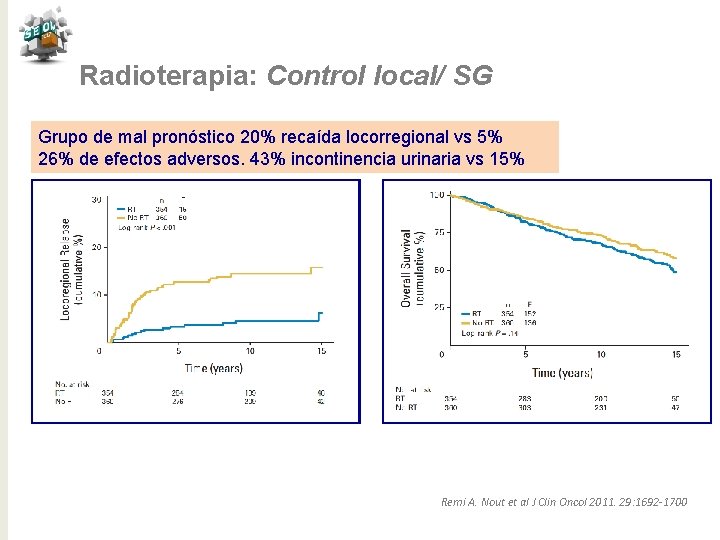
Radioterapia: Control local/ SG Grupo de mal pronóstico 20% recaída locorregional vs 5% 26% de efectos adversos. 43% incontinencia urinaria vs 15% Remi A. Nout et al J Clin Oncol 2011. 29: 1692 -1700

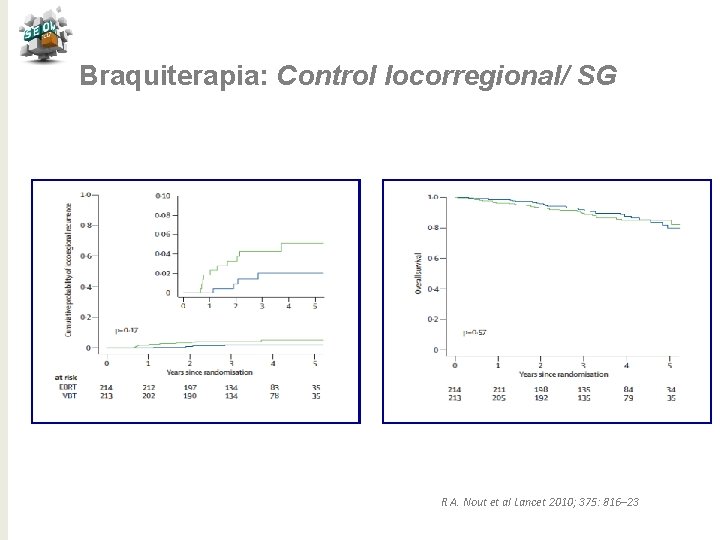
Braquiterapia: Control locorregional/ SG R A. Nout et al Lancet 2010; 375: 816– 23

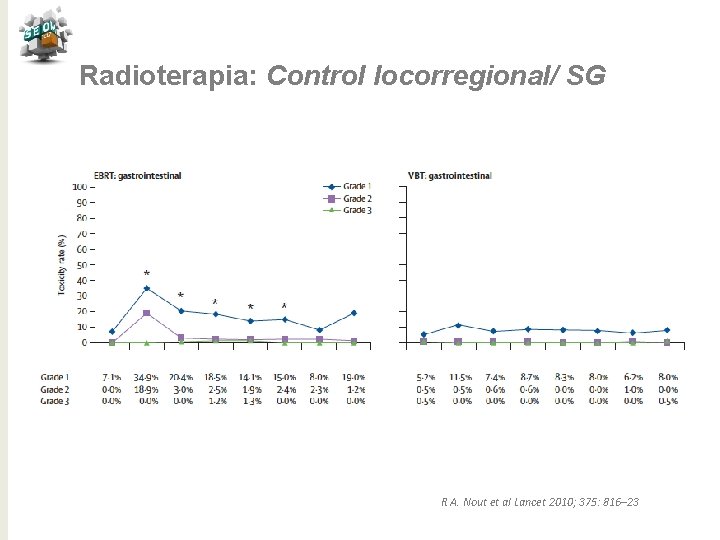
Radioterapia: Control locorregional/ SG R A. Nout et al Lancet 2010; 375: 816– 23

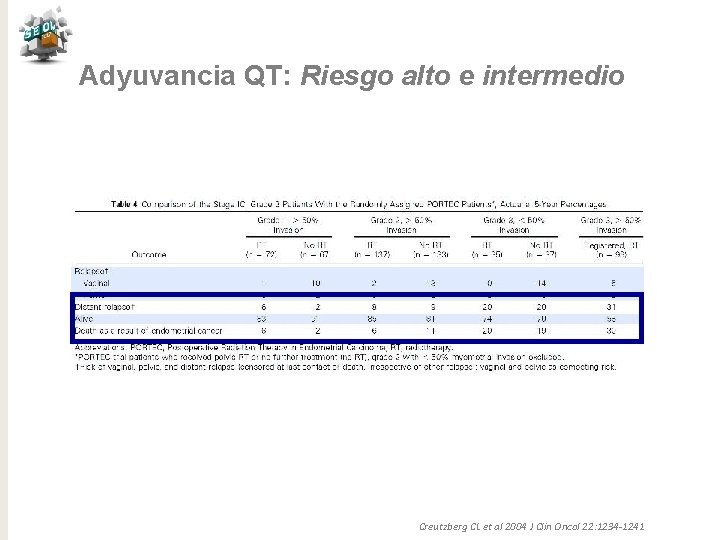
Adyuvancia QT: Riesgo alto e intermedio Creutzberg CL et al 2004 J Clin Oncol 22: 1234 -1241

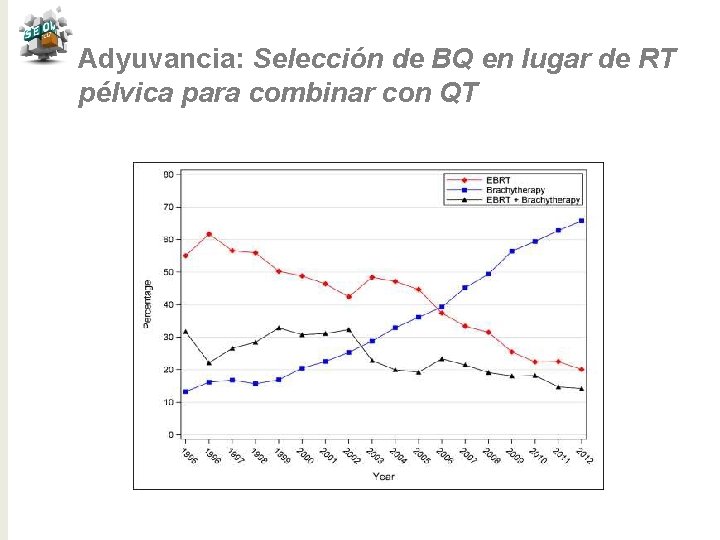
Adyuvancia: Selección de BQ en lugar de RT pélvica para combinar con QT

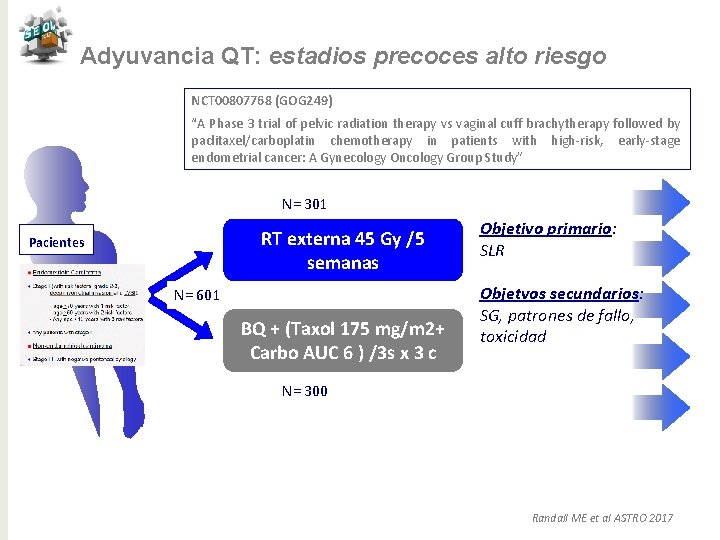
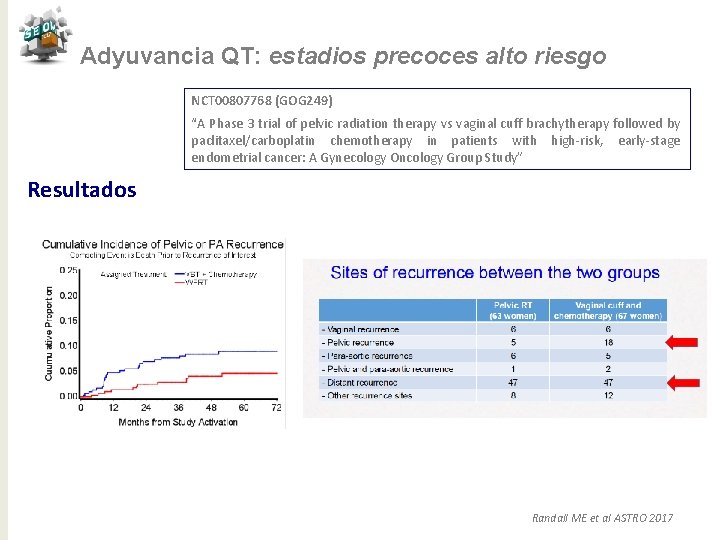
Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” N= 301 RT externa 45 Gy /5 semanas Pacientes N= 601 BQ + (Taxol 175 mg/m 2+ Carbo AUC 6 ) /3 s x 3 c Objetivo primario: SLR Objetvos secundarios: SG, patrones de fallo, toxicidad N= 300 Randall ME et al ASTRO 2017

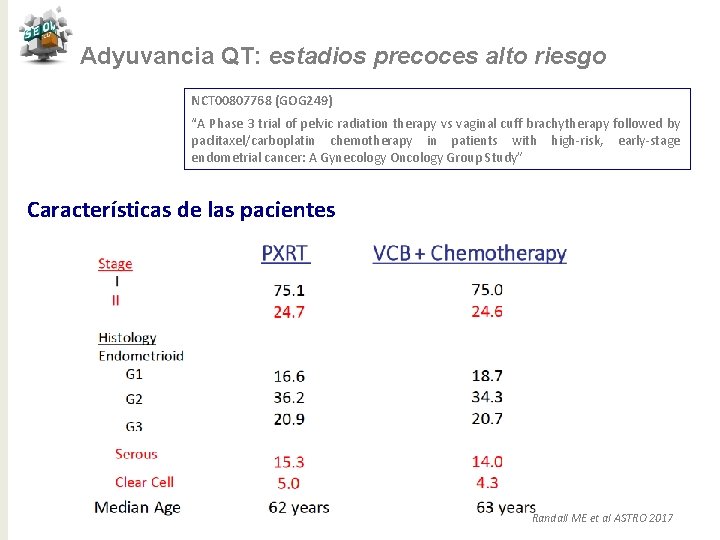
Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Características de las pacientes Randall ME et al ASTRO 2017

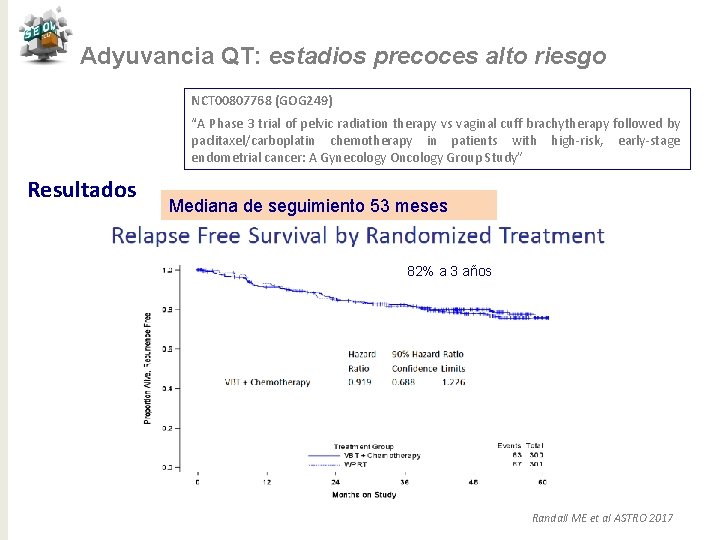
Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Resultados Mediana de seguimiento 53 meses 82% a 3 años Randall ME et al ASTRO 2017

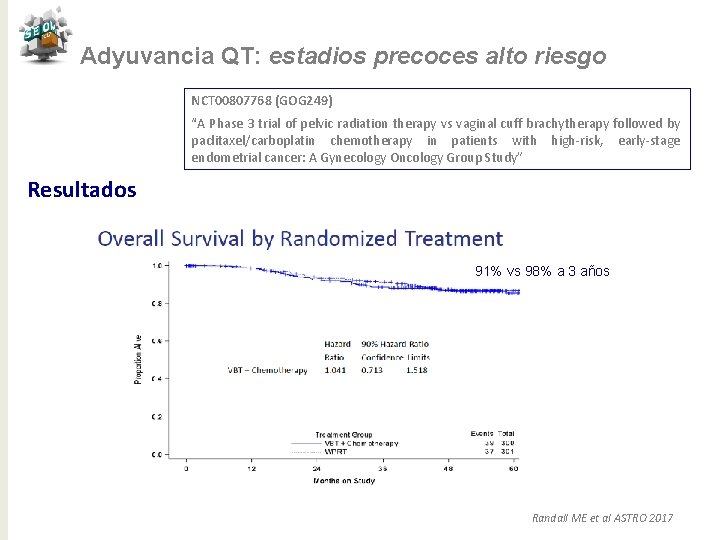
Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Resultados 91% vs 98% a 3 años Randall ME et al ASTRO 2017

Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Resultados Randall ME et al ASTRO 2017

Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Resultados Randall ME et al ASTRO 2017

Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Resultados Randall ME et al ASTRO 2017

Adyuvancia QT: estadios precoces alto riesgo NCT 00807768 (GOG 249) “A Phase 3 trial of pelvic radiation therapy vs vaginal cuff brachytherapy followed by paclitaxel/carboplatin chemotherapy in patients with high-risk, early-stage endometrial cancer: A Gynecology Oncology Group Study” Conclusiones ü El estudio no demuestra superioridad de BQ/QT frente a RT ü Toxicidad aguda mayor en rama experimental ü Recaída ganglionar más frecuente en rama experimental ü RT sola sigue siendo un tratamiento apropiado para las pacientes con tumores en estadíos precoces de alto riesgo ( la modalidad de RT se ha de individualizar en base a factores pronósticos) Randall ME et al ASTRO 2017

Adyuvancia QT: estadios precoces alto riesgo

Radioterapia vs Quimioterapia 19

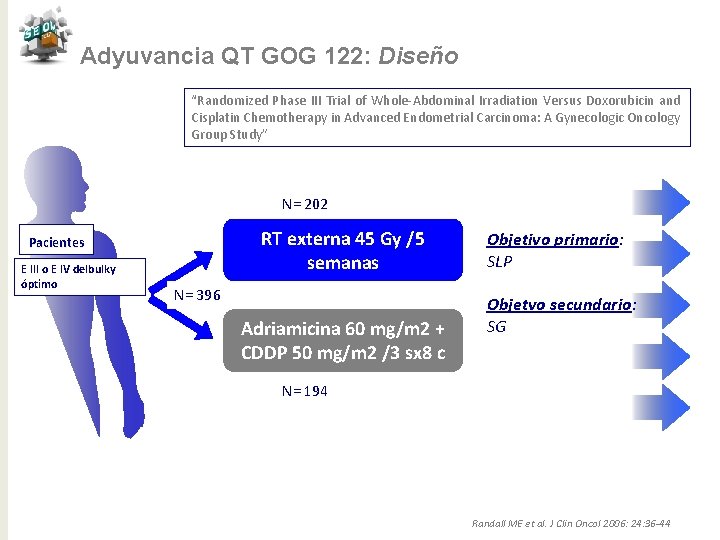
Adyuvancia QT GOG 122: Diseño “Randomized Phase III Trial of Whole-Abdominal Irradiation Versus Doxorubicin and Cisplatin Chemotherapy in Advanced Endometrial Carcinoma: A Gynecologic Oncology Group Study” N= 202 RT externa 45 Gy /5 semanas Pacientes E III o E IV delbulky óptimo N= 396 Adriamicina 60 mg/m 2 + CDDP 50 mg/m 2 /3 sx 8 c Objetivo primario: SLP Objetvo secundario: SG N= 194 Randall ME et al. J Clin Oncol 2006: 24: 36 -44

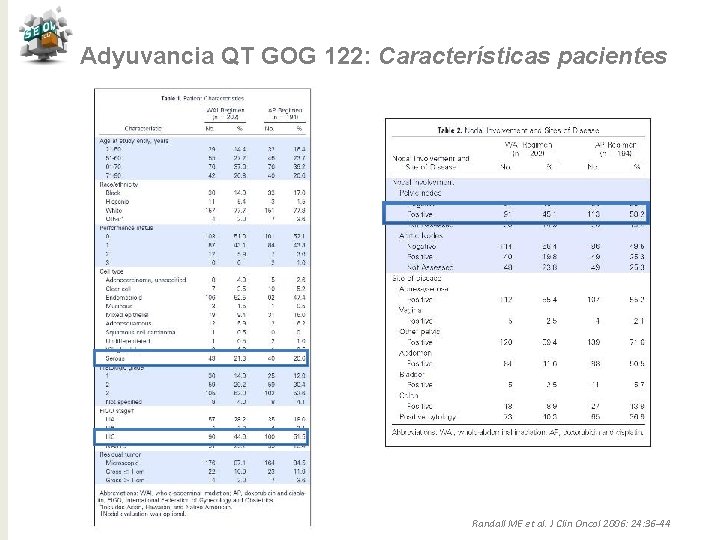
Adyuvancia QT GOG 122: Características pacientes Randall ME et al. J Clin Oncol 2006: 24: 36 -44

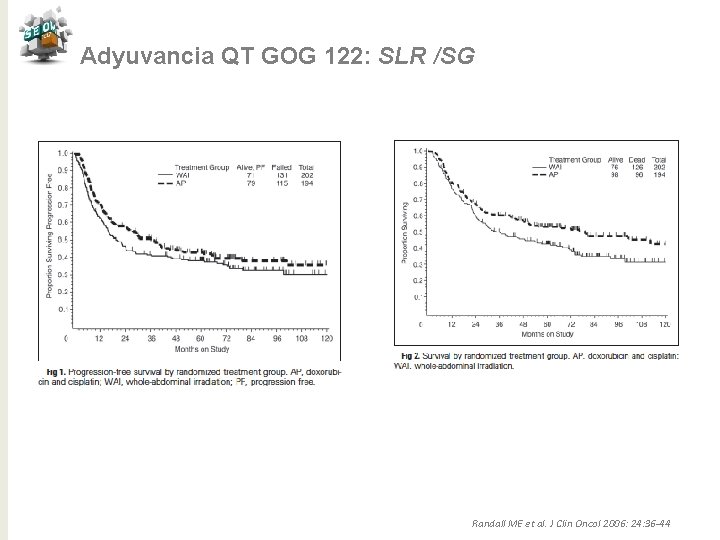
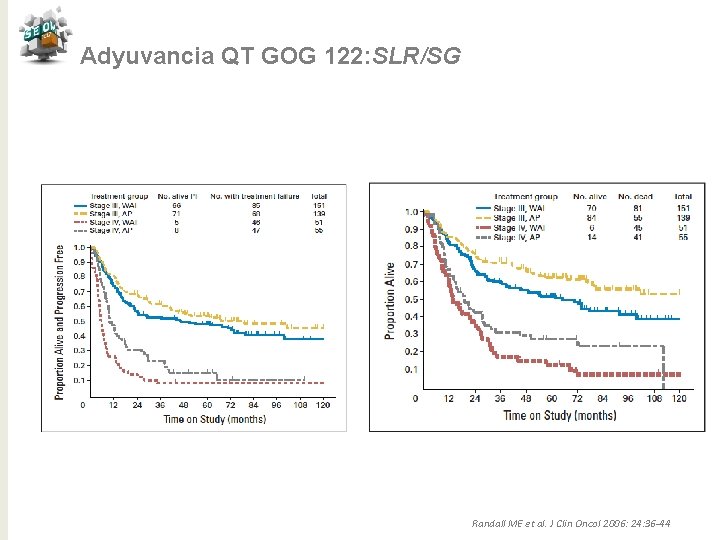
Adyuvancia QT GOG 122: SLR /SG Randall ME et al. J Clin Oncol 2006: 24: 36 -44

Adyuvancia QT GOG 122: SLR/SG Randall ME et al. J Clin Oncol 2006: 24: 36 -44

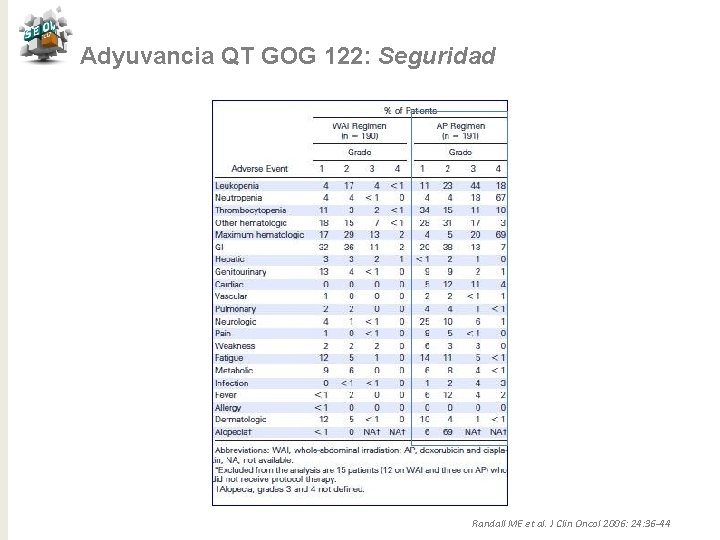
Adyuvancia QT GOG 122: Seguridad Randall ME et al. J Clin Oncol 2006: 24: 36 -44

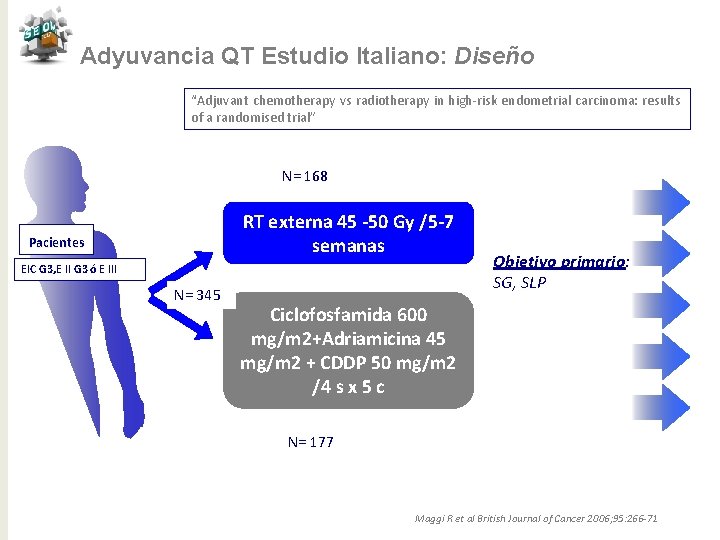
Adyuvancia QT Estudio Italiano: Diseño “Adjuvant chemotherapy vs radiotherapy in high-risk endometrial carcinoma: results of a randomised trial” N= 168 RT externa 45 -50 Gy /5 -7 semanas Pacientes EIC G 3, E II G 3 ó E III N= 345 Objetivo primario: SG, SLP Ciclofosfamida 600 mg/m 2+Adriamicina 45 mg/m 2 + CDDP 50 mg/m 2 /4 s x 5 c N= 177 Maggi R et al British Journal of Cancer 2006; 95: 266 -71

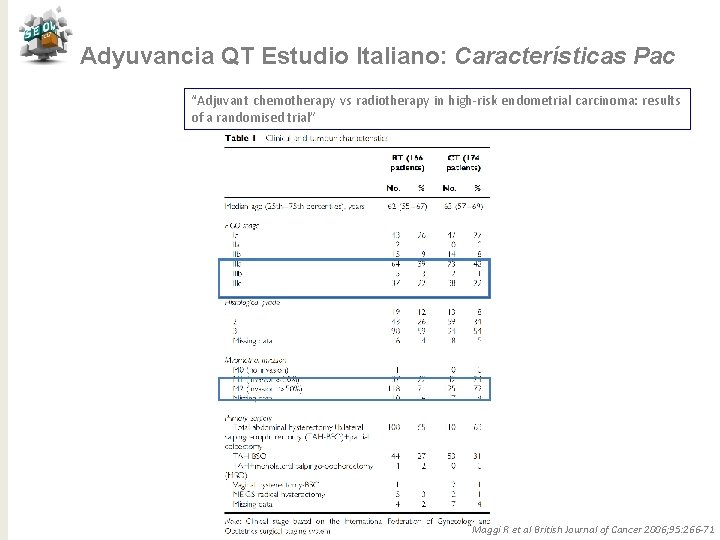
Adyuvancia QT Estudio Italiano: Características Pac “Adjuvant chemotherapy vs radiotherapy in high-risk endometrial carcinoma: results of a randomised trial” Maggi R et al British Journal of Cancer 2006; 95: 266 -71

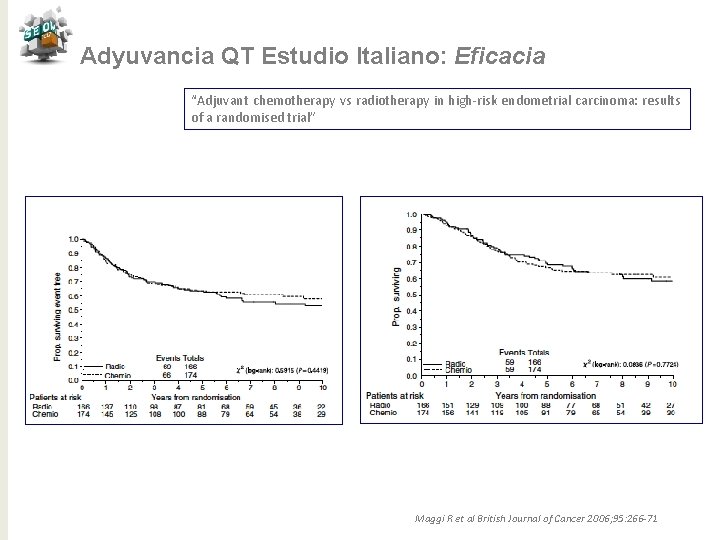
Adyuvancia QT Estudio Italiano: Eficacia “Adjuvant chemotherapy vs radiotherapy in high-risk endometrial carcinoma: results of a randomised trial” Maggi R et al British Journal of Cancer 2006; 95: 266 -71

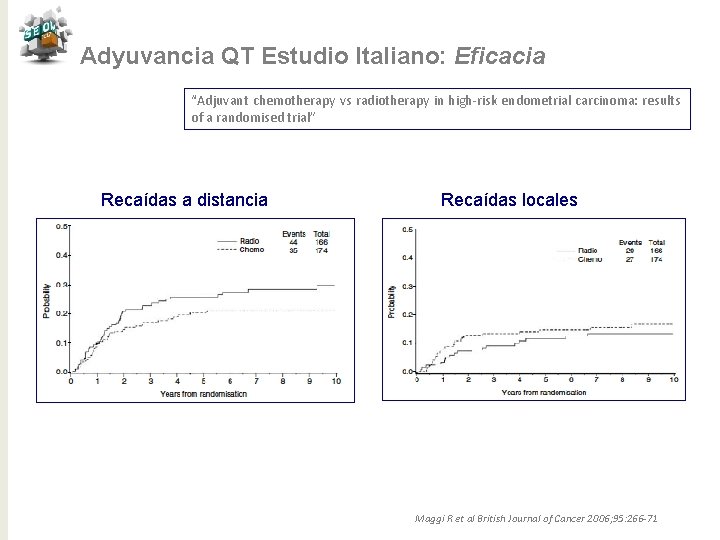
Adyuvancia QT Estudio Italiano: Eficacia “Adjuvant chemotherapy vs radiotherapy in high-risk endometrial carcinoma: results of a randomised trial” Recaídas a distancia Recaídas locales Maggi R et al British Journal of Cancer 2006; 95: 266 -71

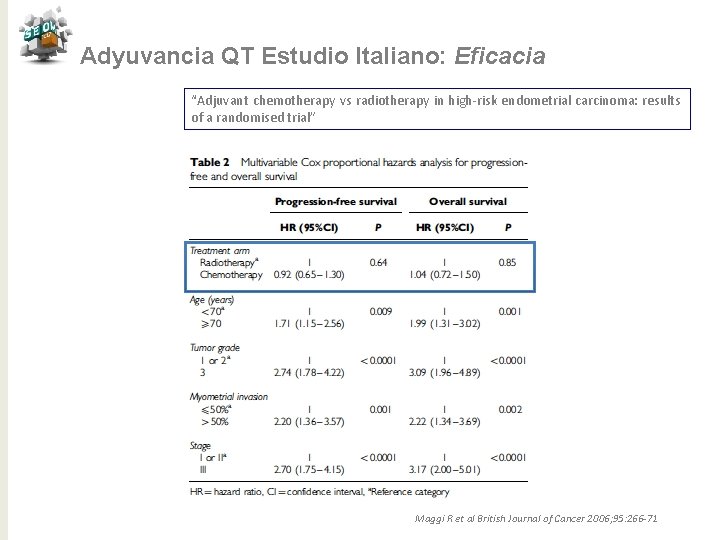
Adyuvancia QT Estudio Italiano: Eficacia “Adjuvant chemotherapy vs radiotherapy in high-risk endometrial carcinoma: results of a randomised trial” Maggi R et al British Journal of Cancer 2006; 95: 266 -71

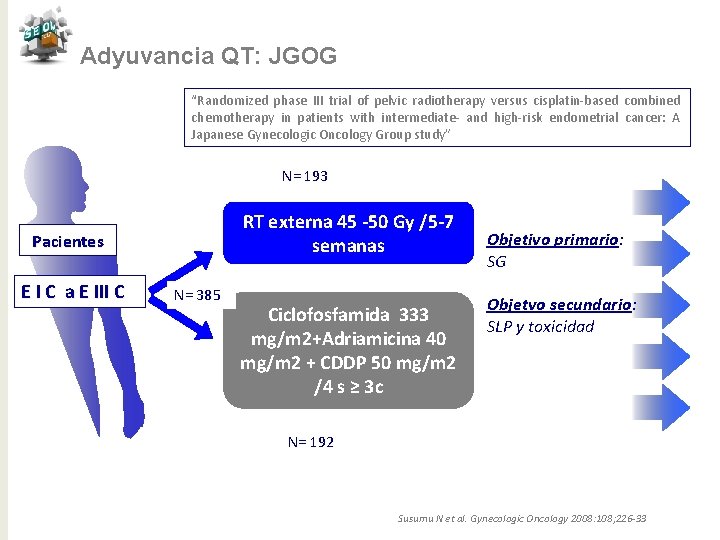
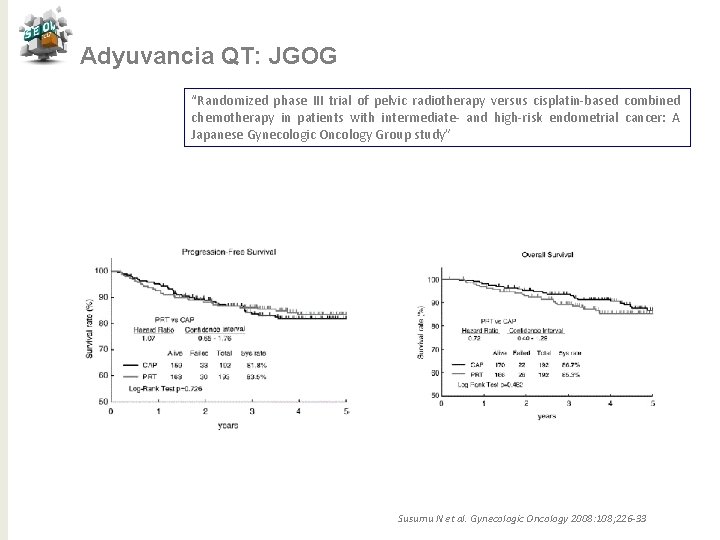
Adyuvancia QT: JGOG “Randomized phase III trial of pelvic radiotherapy versus cisplatin-based combined chemotherapy in patients with intermediate- and high-risk endometrial cancer: A Japanese Gynecologic Oncology Group study” N= 193 RT externa 45 -50 Gy /5 -7 semanas Pacientes E I C a E III C N= 385 Ciclofosfamida 333 mg/m 2+Adriamicina 40 mg/m 2 + CDDP 50 mg/m 2 /4 s ≥ 3 c Objetivo primario: SG Objetvo secundario: SLP y toxicidad N= 192 Susumu N et al. Gynecologic Oncology 2008: 108; 226 -33

Adyuvancia QT: JGOG “Randomized phase III trial of pelvic radiotherapy versus cisplatin-based combined chemotherapy in patients with intermediate- and high-risk endometrial cancer: A Japanese Gynecologic Oncology Group study” Susumu N et al. Gynecologic Oncology 2008: 108; 226 -33

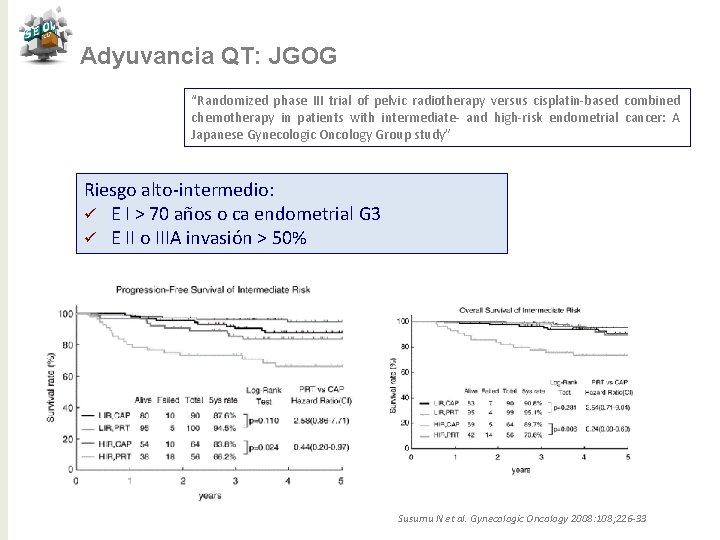
Adyuvancia QT: JGOG “Randomized phase III trial of pelvic radiotherapy versus cisplatin-based combined chemotherapy in patients with intermediate- and high-risk endometrial cancer: A Japanese Gynecologic Oncology Group study” Riesgo alto-intermedio: ü E I > 70 años o ca endometrial G 3 ü E II o IIIA invasión > 50% Susumu N et al. Gynecologic Oncology 2008: 108; 226 -33

Radioterapia vs Quimioradio 33

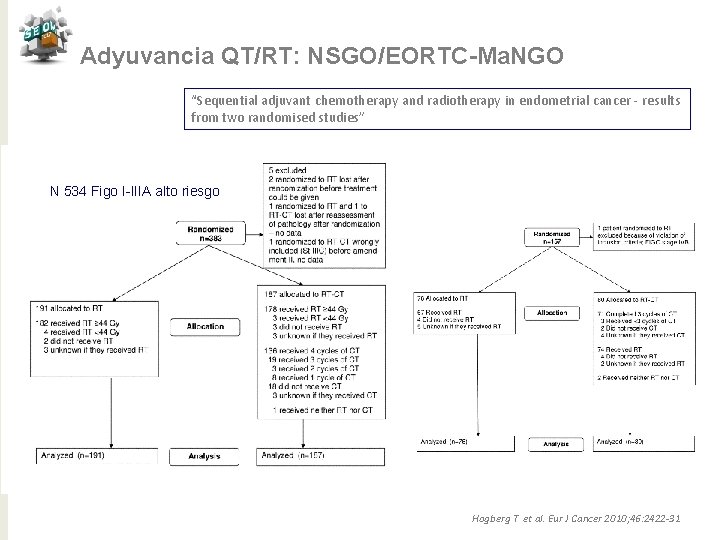
Adyuvancia QT/RT: NSGO/EORTC-Ma. NGO “Sequential adjuvant chemotherapy and radiotherapy in endometrial cancer - results from two randomised studies” N 534 Figo I-IIIA alto riesgo Hogberg T et al. Eur J Cancer 2010; 46: 2422 -31

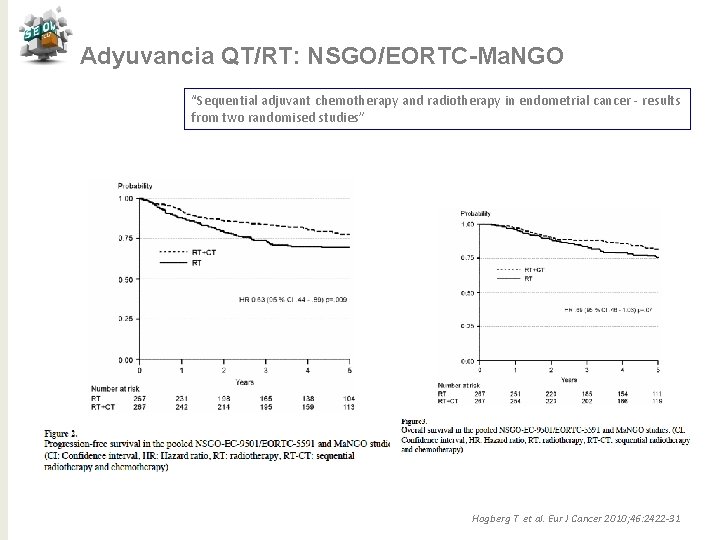
Adyuvancia QT/RT: NSGO/EORTC-Ma. NGO “Sequential adjuvant chemotherapy and radiotherapy in endometrial cancer - results from two randomised studies” Hogberg T et al. Eur J Cancer 2010; 46: 2422 -31

Adyuvancia QT: Racional para uso taxanos • Adriamicina + platino ha sido el regimen estándar en cáncer de endometrio • GOG 177: Primera línea superioridad de añadir taxol a ciaplatino + adriamicina • GOG 209: demuestra que taxol+ platino+ adriamicina es igual a carboplatino + taxol

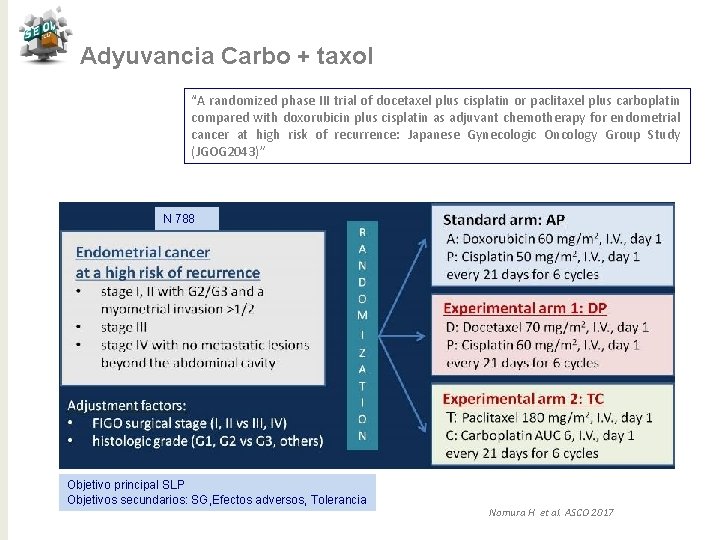
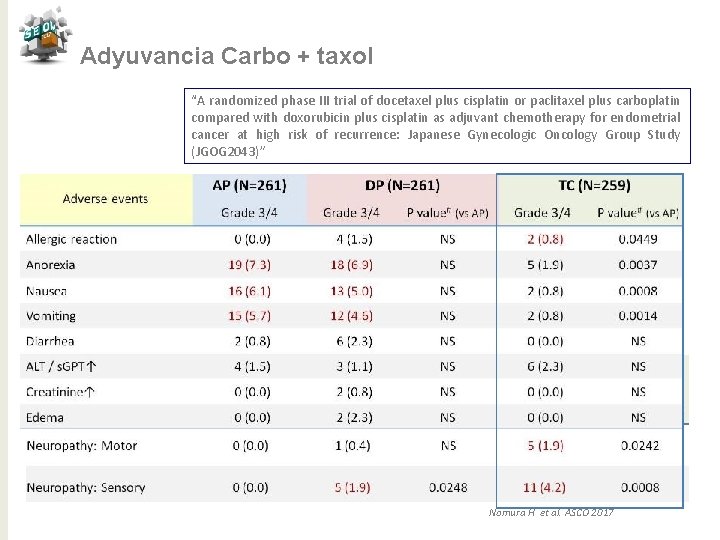
Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” N 788 Objetivo principal SLP Objetivos secundarios: SG, Efectos adversos, Tolerancia Nomura H et al. ASCO 2017

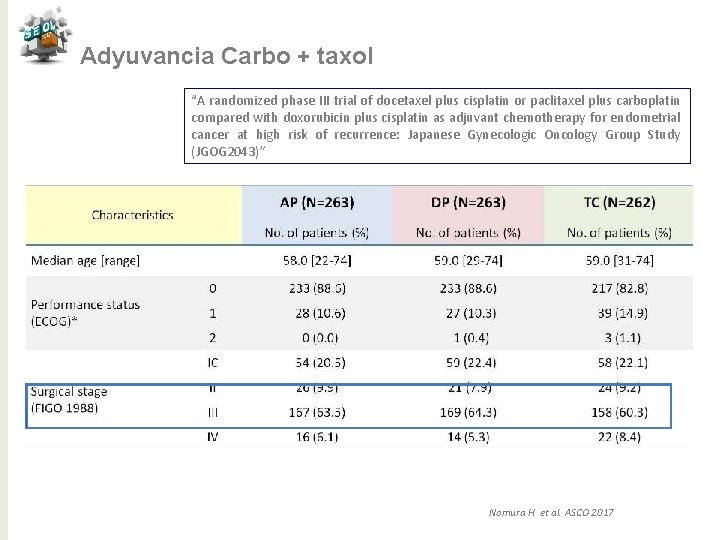
Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

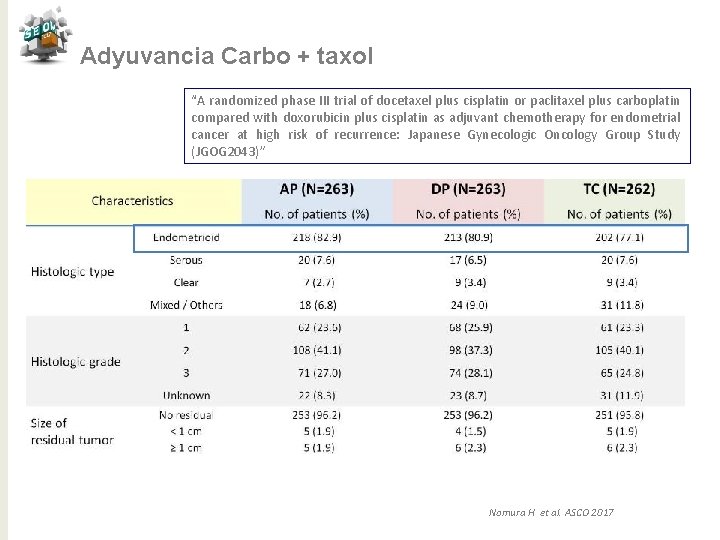
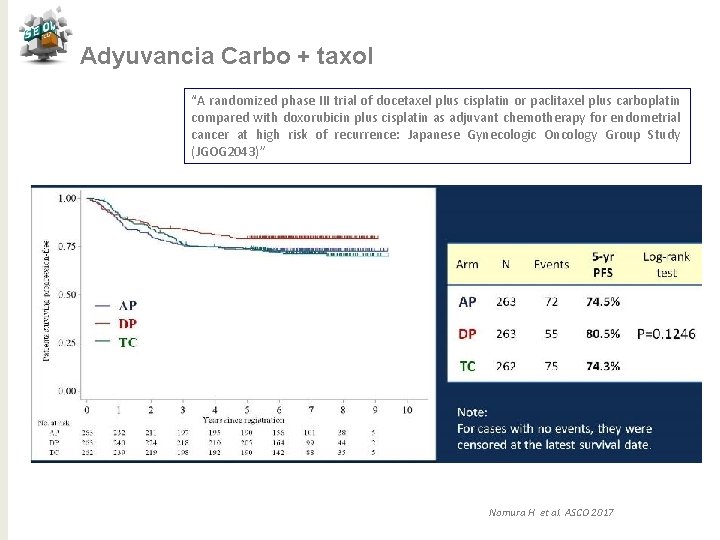
Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

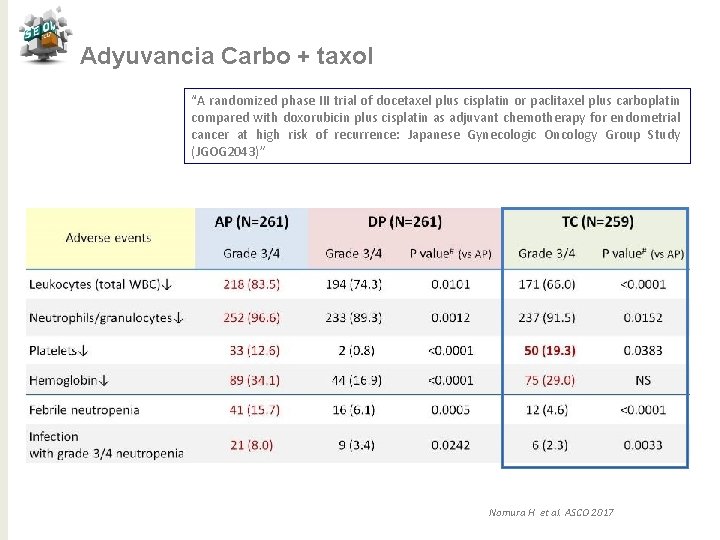
Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

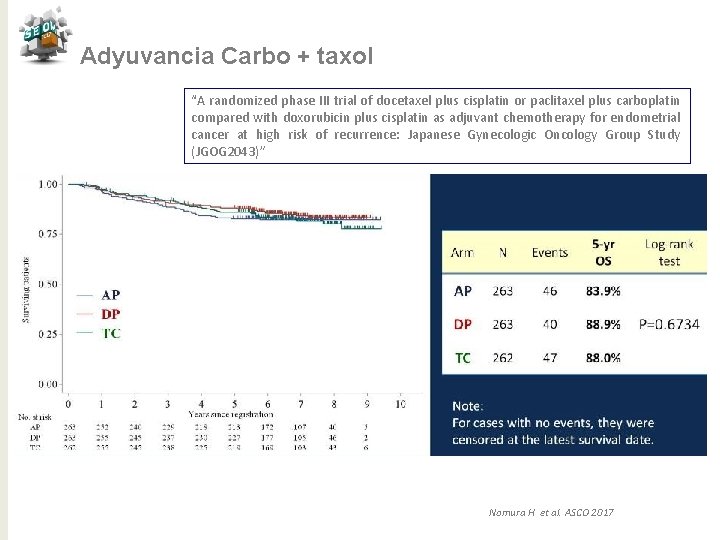
Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

Adyuvancia Carbo + taxol “A randomized phase III trial of docetaxel plus cisplatin or paclitaxel plus carboplatin compared with doxorubicin plus cisplatin as adjuvant chemotherapy for endometrial cancer at high risk of recurrence: Japanese Gynecologic Oncology Group Study (JGOG 2043)” Nomura H et al. ASCO 2017

Quimioradio+QT vs Quimioterapia 44

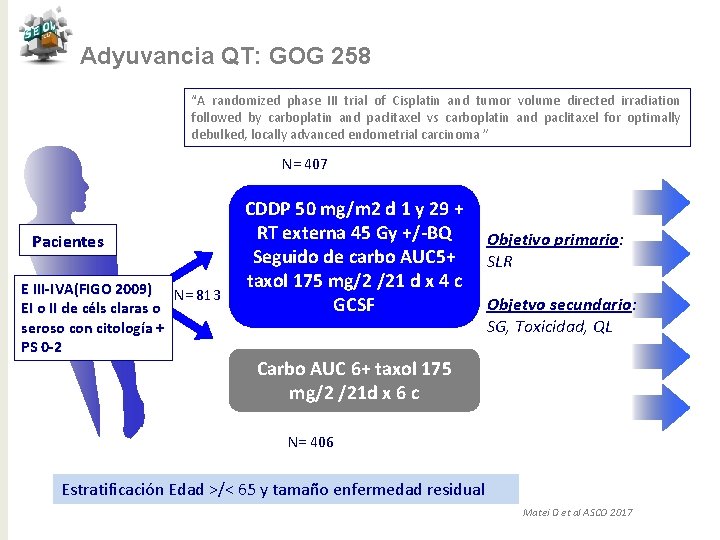
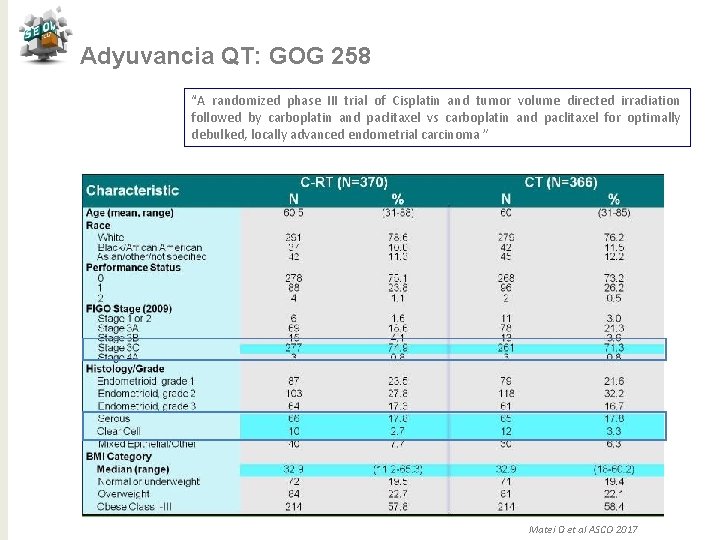
Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” N= 407 Pacientes E III-IVA(FIGO 2009) N= 813 EI o II de céls claras o seroso con citología + PS 0 -2 CDDP 50 mg/m 2 d 1 y 29 + RT externa 45 Gy +/-BQ Seguido de carbo AUC 5+ taxol 175 mg/2 /21 d x 4 c GCSF Objetivo primario: SLR Objetvo secundario: SG, Toxicidad, QL Carbo AUC 6+ taxol 175 mg/2 /21 d x 6 c N= 406 Estratificación Edad >/< 65 y tamaño enfermedad residual Matei D et al ASCO 2017

Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” Matei D et al ASCO 2017

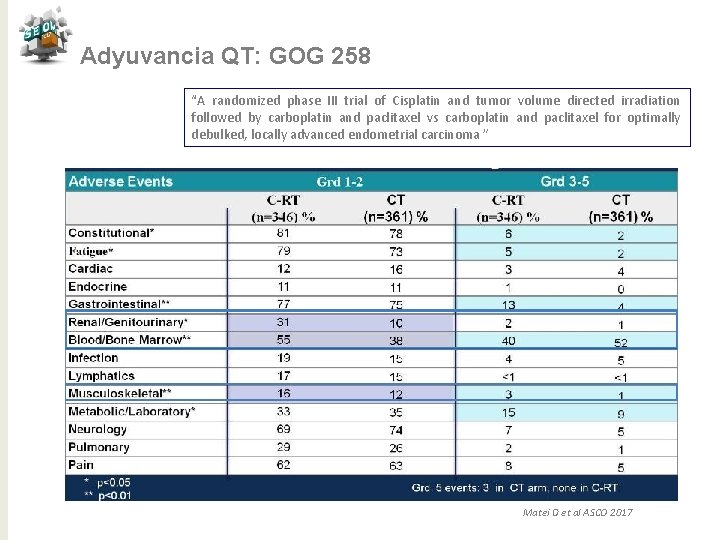
Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” Matei D et al ASCO 2017

Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” Matei D et al ASCO 2017

Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” Matei D et al ASCO 2017

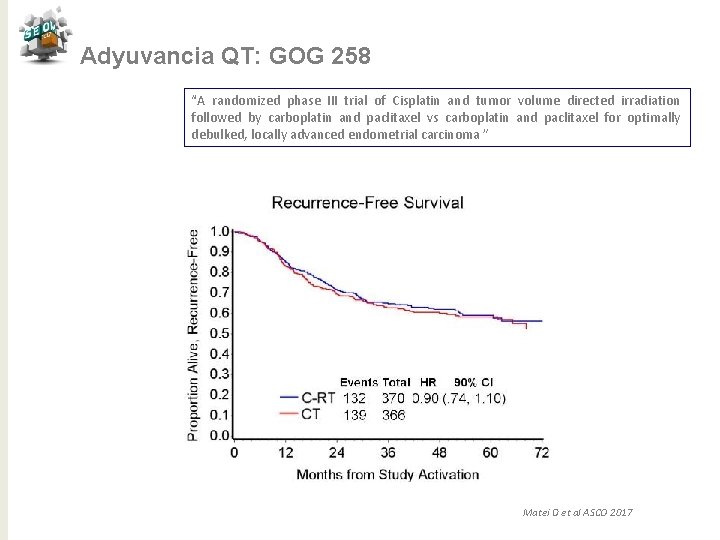
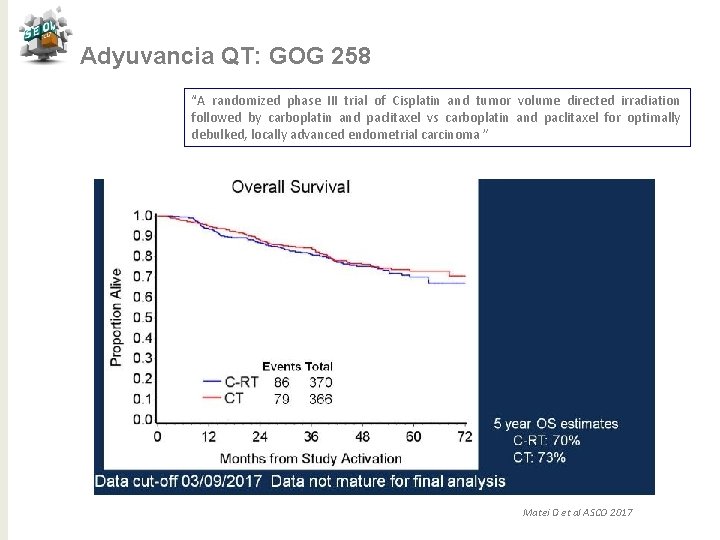
Adyuvancia QT: GOG 258 “A randomized phase III trial of Cisplatin and tumor volume directed irradiation followed by carboplatin and paclitaxel vs carboplatin and paclitaxel for optimally debulked, locally advanced endometrial carcinoma ” Conclusiones ü QT-RT no demuestra superioridad comparado con QT (HR 0. 9, CI 0. 74 -1. 1) ü Toxicidad aguda similar ambas ramas ü QT-RT reduce la incidencia de recaídas vaginales, pélvicas y paraaórticas con respecto a QT ü La recaída a distancia es más frecuente en QT-RT vs QT ü Los resultados en SG y QL se reportarán en el futuro

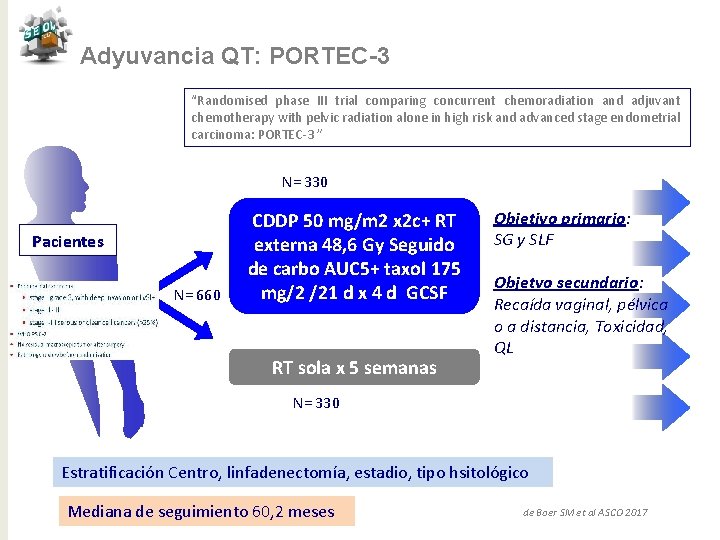
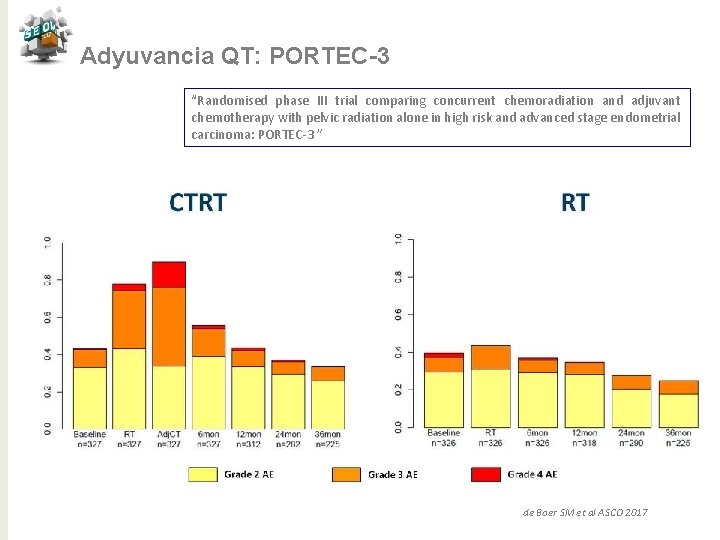
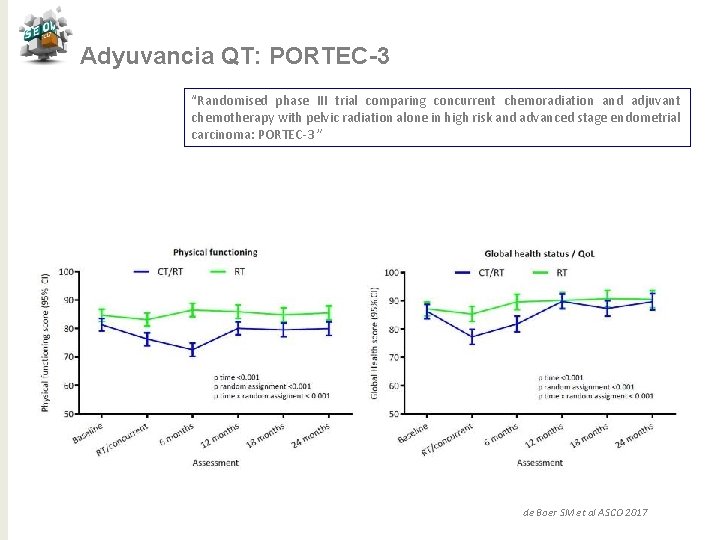
Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” N= 330 Pacientes N= 660 CDDP 50 mg/m 2 x 2 c+ RT externa 48, 6 Gy Seguido de carbo AUC 5+ taxol 175 mg/2 /21 d x 4 d GCSF RT sola x 5 semanas Objetivo primario: SG y SLF Objetvo secundario: Recaída vaginal, pélvica o a distancia, Toxicidad, QL N= 330 Estratificación Centro, linfadenectomía, estadio, tipo hsitológico Mediana de seguimiento 60, 2 meses de Boer SM et al ASCO 2017

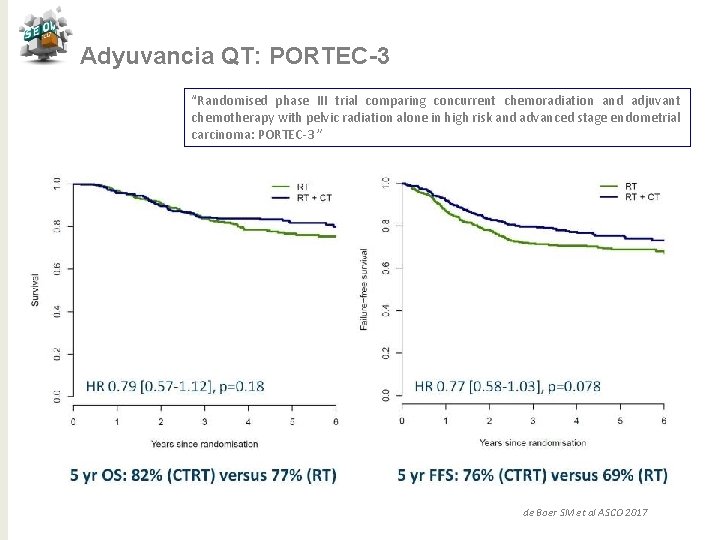
Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

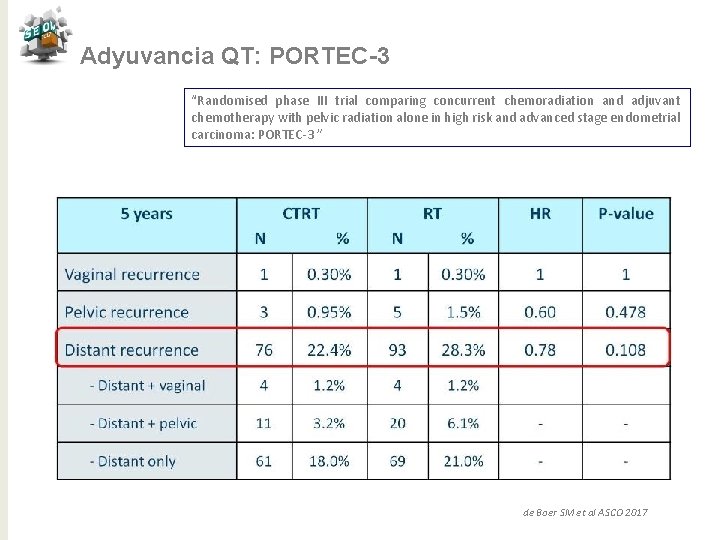
Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

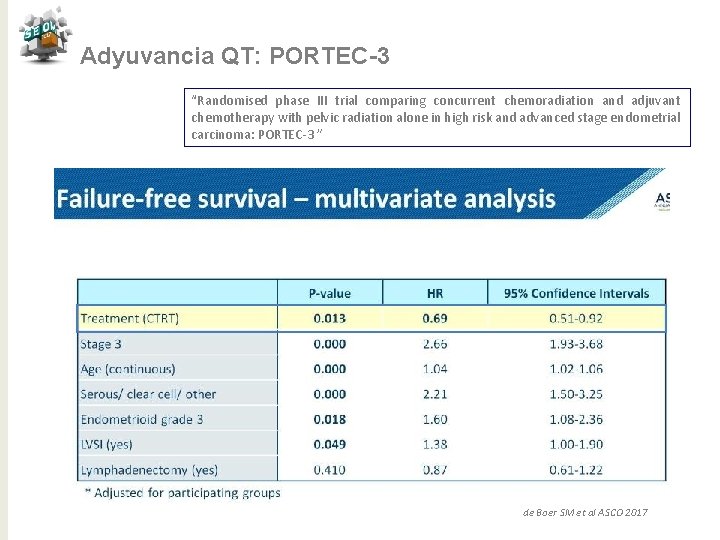
Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” de Boer SM et al ASCO 2017

Adyuvancia QT: PORTEC-3 “Randomised phase III trial comparing concurrent chemoradiation and adjuvant chemotherapy with pelvic radiation alone in high risk and advanced stage endometrial carcinoma: PORTEC-3 ” Conclusiones QTRT vs RT para alto riesgo ü Tendencia de mejora en SV a 5 años con reducción 7% (SLF) y 5% (SG) ü Beneficio significativo del 11% en SLF con QTRT para E III ü No puede ser recomendado para E I-II ü Mayor toxicidad para QTRT en los primeros 12 meses ü Necesitamos > seguimiento para SG de Boer SM et al ASCO 2017 j





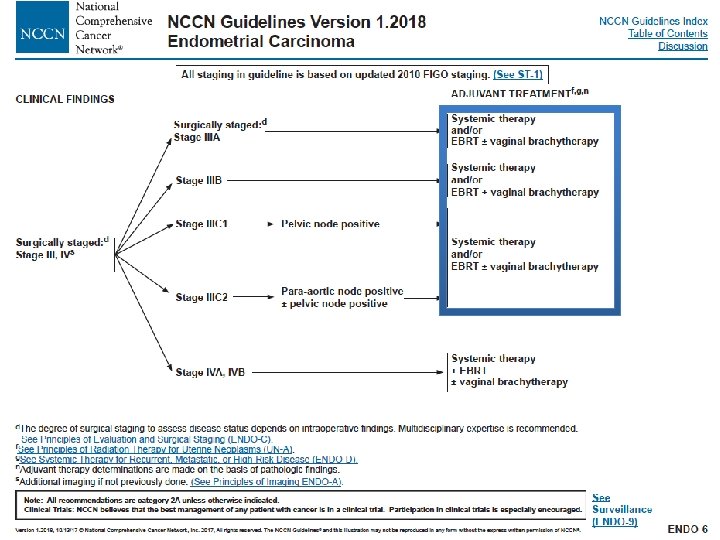
Conclusiones ü No indicación de rutina para tratamiento sistémico en pacientes con estadios iniciales de alto riesgo ü En estadios III y IV A indicación de QT adyuvante ( aumento SG y SLP) o RT QT frente a RT exclusiva ( Aumento SLR y SGCE) ü Pendiente de resultados de SG para indicación de QT/RT QT ü En tumores serosos y de células claras tratamiento QT adyuvante también en estadios precoces

Gracias por su atención!!!!! Nuria Lainez Milagro Complejo hospitalario de Navarra