Modern Chemistry Chapter 9 Stoichiometry Modern Chemistry Chapter














































- Slides: 47

Modern Chemistry Chapter 9 Stoichiometry

Modern Chemistry Chapter 9 Stoichiometry n n composition stoichiometry deals with the mass relationships of elements in compounds. reaction stoichiometry involves the mass relationships between reactants and products in a chemical reaction.

Types of Stoichiometry Problems 1) 2) 3) 4) mole to mole (Both the given and the unknown quantities are amounts in moles. ) mole to mass (The given amount is in moles and the unknown amount is in grams. ) mass to mole (The given amount is in grams and the unknown amount is in moles. ) mass to mass (Both the given and the unknown amount is in grams. )

Mole Ratio & Molar Mass n n mole ratio- A conversion factor that relates the amounts in moles of any two substances involved in a chemical reaction. Found by using the coefficients in the balanced formula equation of the reaction. n n molar mass- Equal to the mass in grams of one mole of an element or a compound. Found by adding the individual element atomic masses from the formula of the compound.

Section Review Problems n Do section review problems #1 through #4 on page 301 of the textbook.

Section Review page 301 1 - The branch of chemistry that deals with mass relationships in compounds and in chemical reactions. 2 Hg. O 2 Hg + O 2 2 - a) 2 mol Hg. O 2 mol Hg & 2 mol Hg. O 1 mol O 2 2 mol Hg. O & 2 mol Hg 1 mol O 2 2 mol Hg. O & 1 mol O 2 2 mol Hg

Section Review page 301 3 - It is used to convert moles of one substance into moles of another substance. 4 - The formula equation MUST be BALANCED so mole ratios can be determined.

Using Conversion Factors Amount coefficient amount of of given x of unknown = unknown substance coefficient substance in moles of known in moles # moles given x mole ratio = #moles of unknown Do practice problems #1 & #2 on page 306 of text.

Page 306 #1 3 H 2 + N 2 2 NH 3 6 mol H 2 x 2 mol NH 3 = 4 mol NH 3 3 mol H 2

Page 306 #2 2 KCl. O 3 2 KCl + 3 O 2 15 mol O 2 x 2 mol KCl. O 3 = 10 mol KCl. O 3 3 mol O 2

Chapter 9 quiz #1 - mole to mole calculations 6 Na. Br + Mg 3(PO 4)2 2 Na 3 PO 4 + 3 Mg. Br 2 Use the above balanced formula equation to solve the following: 1 - 7. 0 moles of Na. Br will produce ? moles Na 3 PO 4 2 - 3. 0 moles of Mg 3(PO 4)2 will yield ? moles Mg. Br 2 3 - 0. 5 moles of Na. Br will react with ? moles Mg 3(PO 4)2 4 - 2. 5 moles of Na. Br will yield ? moles Na 3 PO 4 5 - 2. 5 moles Na. Br will produce ? moles Mg. Br 2

6 Na. Br + Mg 3(PO 4)2 2 Na 3 PO 4 + 3 Mg. Br 2 1 - 7 mol Na. Br x 2 mol Na 3 PO 4 = 2. 3 mol Na 3 PO 4 6 mol Na. Br 2 - 3 mol Mg 3(PO 4)2 x 3. 0 mol Mg. Br 2 = 9 mol Mg. Br 2 1 mol Mg 3(PO 4)2 3 - 0. 5 mol Na. Br x 1 mol Mg 3(PO 4)2 = 0. 083 mol 6 mol Na. Br 4 - 2. 5 mol Na. Br x 2 mol Na 3 PO 4 = 0. 83 mol Na 3 PO 4 6 mol Na. Br 5 - 2. 5 mol Na. Br x 3 mol Mg. Br 2 = 1. 25 mol Mg. Br 2 6 mol Na. Br

Using Conversion Factors amount of mass in given x moles unknown x molar = grams of substance moles known mass of unknown in moles unknown substance # moles given x mole ratio x molar = mass of unknown mass (in grams) unknown Do practice problems #1 & #2 on page 308 of the textbook.

Page 308 #1 & 2 2 Mg + O 2 2 Mg. O 2. 00 mol Mg x 2 mol Mg. O x 40. 3 g Mg. O = 80. 6 g Mg. O 2 mol Mg 1 mol Mg. O 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 10 mol CO 2 x 1 mol C 6 H 12 O 6 x 180 g C 6 H 12 O 6 = 300 g C 6 H 12 O 6 6 mol CO 2 1 mol C 6 H 12 O 6

Chapter 9 Quiz #2 - mole-mass problems 3 Mg. F 2 + Al 2 O 3 3 Mg. O + 2 Al. F 3 Use the above balanced formula equation to answer the following questions. 1 - 2. 0 mol Mg. F 2 will yield ? grams of Mg. O 2 - 4. 0 mol of Al 2 O 3 ? grams of Al. F 3 3 - If 6. 0 mol of Mg. O is produced, ? grams of Al. F 3 4 - 0. 6 mol Mg. F 2 ? grams of Al. F 3 5 - 2. 75 mol Al 2 O 3 ? grams of Mg. O

3 Mg. F 2 + Al 2 O 3 3 Mg. O + 2 Al. F 3 1 - 2. 0 mol Mg. F 2 x 3 mol Mg. O x 40. 3 g Mg. O = 80. 6 g 3 mol Mg. F 2 1 mol Mg. O 2 - 4. 0 mol Al 2 O 3 x 2 mol Al. F 3 x 84. 0 g Al. F 3 = 672. 0 g 1 mol Al 2 O 3 1 mol Al. F 3 3 - 6. 0 mol Mg. O x 2 mol Al. F 3 x 84. 0 g Al. F 3 = 336 g Al. F 3 3 mol Mg. O 1 mol Al. F 3 4 - 0. 6 mol Mg. F 2 x 2 mol Al. F 3 x 84. 0 g Al. F 3 = 33. 6 g Al. F 3 3 mol Mg. F 2 1 mol Al. F 3 5 - 2. 75 mol Al 2 O 3 x 3 mol Mg. O x 40. 3 g Mg. O = 332 g. Mg. O 1 mol Al 2 O 3 1 mol Mg. O

Using Conversion Factors mass (g) x 1 mol given x mol unknown = moles of of given molar mass mol given unknown substance of given substance grams x 1 x mole ratio = moles unknown molar mass Do practice problems #1 & #2 on page 309 of the textbook.

Practice problems page 309 2 Hg. O 2 Hg + O 2 125 g O 2 x 1 mol O 2 x 2 mol Hg. O = 7. 81 mol Hg. O 32 g O 2 1 mol O 2 125 g O 2 x 1 mol O 2 x 2 mol Hg = 7. 81 mol Hg 32 g O 2 1 mol O 2

chapter 9 quiz #3 - mass-mole problems Na 2 O + Ca. F 2 2 Na. F + Ca. O Use the above equation to solve the problems. 1 - 156. 1 grams of Ca. F 2 ? mol Ca. O 2 - 186 g Na 2 O ? mol Na. F 3 - 31 g Na 2 O ? mol Ca. O 4 - 31 g Na 2 O ? mol Na. F 5 - A yield of 84 g Na. F ? mol Ca. O

Na 2 O + Ca. F 2 2 Na. F + Ca. O 1 - 156. 1 g Ca. F 2 x 1 mol Ca. F 2 x 1 mol Ca. O = 2. 00 mol Ca. O 78. 1 g Ca. F 2 1 mol Ca. F 2 2 - 186. 0 g Na 2 O x 1 mol Na 2 O x 2 mol Na. F = 6. 0 mol Na. F 62 g Na 2 O 1 mol Na 2 O 3 - 31. 0 g Na 2 O x 1 mol Ca. O = 0. 5 mol Ca. O 62. 0 g Na 2 O 1 mol Na 2 O 4 - 31. 0 g Na 2 O x 1 mol Na 2 O x 2 mol Na. F = 1. 0 mol Na. F 62. 0 g Na 2 O 1 mol Na 2 O 5 - 84. 0 g Na. F x 1 mol Na. F x 1 mol Ca. O = 1. 0 mol Ca. O 42. 0 g Na. F 2 mol Na. F

Chemistry Chapter 9 - Stoichiometry Practice Problems 2 Na. F + Ca. O Na 2 O + Ca. F 2 1 - 4. 5 moles of Na. F will produce -? - moles of Na 2 O ? 4. 5 mol Na. F x 1 mol Na 2 O/2 mol Na. F = 2. 25 moles Na 2 O 2 - 3. 2 moles of Ca. O will produce -? - grams of Ca. F 2 ? 3. 2 mol Ca. O x 1 mol Ca. F 2/1 mol Ca. O x 78. 1 g Ca. F 2/mol Ca. F 2 = 249. 9 g Ca. F 2

2 Na. F + Ca. O Na 2 O + Ca. F 2 3 - 168. 0 grams of Na. F will produce -? - moles of Na 2 O ? 168. 0 /42. 0 x 1 /2 = 2. 0 mol Na 2 O 4 - 112. 2 grams of Ca. O will produce -? - moles of Ca. F 2 ? 112. 2/56. 1 x 1/1 = 2. 0 mol Ca. F 2

5 - Calculate the molar mass of each of the reactants & products of the above balanced formula equation. Use the molar masses in the following problems. a- Al. N = (1 x 27. 0) + (1 x 14. 0) = 41. 0 g/mol b- Na 2 O = (2 x 23. 0) + (1 x 16. 0) = 62. 0 g/mol c- Al 2 O 3 = (2 x 27. 0) + (3 x 16. 0) = 102. 0 g/mol d- Na 3 N = (3 x 23. 0) + (1 x 14. 0) = 83. 0 g/mol

2 Al. N + 3 Na 2 O Al 2 O 3 + 2 Na 3 N 6 - 82. 0 grams of Al. N will produce -? - moles of Al 2 O 3 ? 82. 0/41. 0 x 1/2 = 1. 0 mol Al 2 O 3 7 - 164. 0 grams Al. N will produce -? - moles of Na 3 N ? 164. 0/41. 0 x 2/2 = 4. 0 mol Na 3 N

2 Al. N + 3 Na 2 O Al 2 O 3 + 2 Na 3 N 8 - 9 - 2. 5 moles of Na 2 O will produce -? - grams of Na 3 N ? 2. 5 x 2/3 x 83. 0 = 138. 3 g Na 3 N 0. 75 moles of Na 2 O will produce -? - moles of Al 2 O 3 ? 0. 75 x 1/3 = 0. 25 mol Al 2 O 3 10 - 11. 0 moles of Na 2 O will produce -? - grams of Na 3 N ? 11. 0 x 2/3 x 83. 0 = 608. 7 g Na 3 N

2 H 2 + O 2 2 H 2 O 11 - 8. 0 grams of H 2 will react with -? - moles of O 2 ? 8. 0/2. 0 x 1/2 = 2. 0 mol O 2 12 - 64. 0 grams of O 2 will produce -? - moles of H 2 O ? 64. 0/32. 0 x 2/1 = 4. 0 mol H 2 O 13 - 0. 25 moles of H 2 will produce -? - moles of H 2 O ? 0. 25 x 2/2 = 0. 25 mol H 2 O

2 H 2 + O 2 2 H 2 O 14 - 1. 5 moles of H 2 will produce -? - grams of H 2 O ? 1. 5 x 2/2 x 18. 0 = 27. 0 g H 2 O 15 - 14 moles of O 2 will produce -? - moles of H 2 O ? 14 x 2/1 = 28. 0 mol H 2 O

Using Conversion Factors mass (g) x 1 mol given x mol unknown x molar mass unknown = mass of given molar mass mol given 1 mol unknown of unsubstance of given known grams x 1 x mole ratio x molar mass = mass of unknown given molar mass Do practice problems #1, #2, & #3 on page 311 of textbook. Do section review problems #1 - #5 on page 311 of textbook.

Practice problems page 311 NH 4 NO 3 N 2 O + 2 H 2 O 32 g N 2 O x 1 mol NH 4 NO 3 x 80 g NH 4 NO 3 = 60 g NH 4 NO 3 44 g N 2 O 1 mol N 2 O 1 mol NH 4 NO 3

Chapter 9 quiz #4 - mass-mass problems Na 2 O + Ca. F 2 2 Na. F + Ca. O 1 - 124 g Na 2 O ? grams Na. F 2 - 124 g Na 2 O ? grams Ca. O 3 - 234. 3 g Ca. F 2 ? g Na. F 4 - 234. 3 g Ca. F 2 ? g Ca. O 5 - 84. 0 g Na. F ? g Ca. O

Na 2 O + Ca. F 2 2 Na. F + Ca. O 1 - 124 g Na 2 O x 1 mol Na 2 O x 2 mol Na. F x 42. 0 g Na. F = 168 g 62 g Na 2 O 1 mol Na. F 2 - 124 g Na 2 O x 1 mol Ca. O x 56. 1 g Ca. O = 112. 2 g 62 g Na 2 O 1 mol Na 2 O 1 mol Ca. O 3 - 234. 3 g Ca. F 2 x 1 mol Ca. F 2 x 2 mol Na. F x 42. 0 g Na. F = 252 g 78. 1 g. Ca. F 2 1 mol Ca. F 2 1 mol Na. F 4 - 234. 3 g Ca. F 2 x 1 mol Ca. O x 56. 1 g Ca. O = 168. 3 g 78. 1 g. Ca. F 2 1 mol Ca. F 2 1 mol Ca. O 5 - 84. 0 g Na. F x 1 mol Ca. O x 56. 1 g Ca. O = 56. 1 g 42. 0 g Na. F 2 mol Na. F 1 mol Ca. O

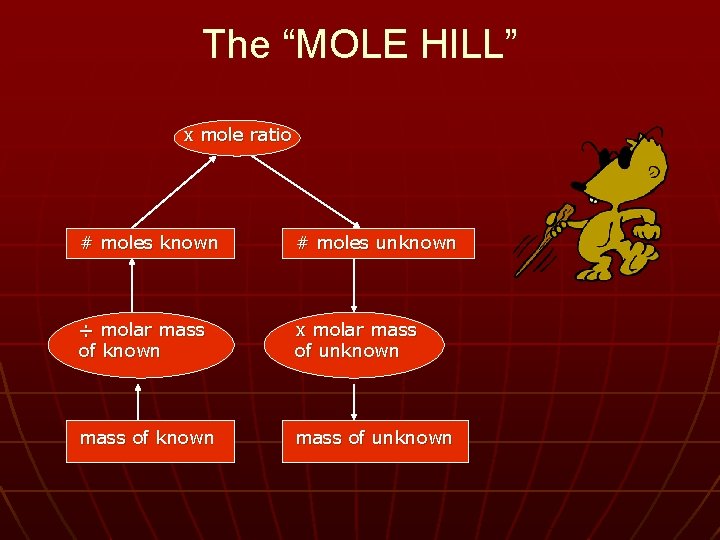
The “MOLE HILL” x mole ratio # moles known # moles unknown ÷ molar mass of known x molar mass of unknown

Stoichiometry Practice Problems 2 H 2 + O 2 2 H 2 O 1) 2. 5 mol H 2 x 1 mol O 2 = 1. 25 mol O 2 2 mol H 2 2) 2. 5 mol H 2 x 2 mol H 2 O = 2. 5 mol H 2 O 2 mol H 2 3) 2. 5 mol H 2 x 1 mol O 2 x 32 g O 2 = 40 g O 2 2 mol H 2 1 mol O 2 4) 2. 5 mol H 2 x 2 mol H 2 O x 18 g H 2 O = 45 g H 2 O 2 mol H 2 1 mol H 2 O

Stoichiometry Practice Problems 2 H 2 + O 2 2 H 2 O 5) 16 g H 2 x 1 mol O 2 = 4. 0 mol O 2 2 g H 2 2 mol H 2 6) 16 g H 2 x 1 mol H 2 x 2 mol H 2 O = 8. 0 mol H 2 O 2. 0 g H 2 2 mol H 2 7) 16 g H 2 x 1 mol O 2 x 32 g O 2 = 128 g O 2 2. 0 g H 2 2 mol H 2 1 mol O 2 8) 16 g H 2 x 1 mol H 2 x 2 mol H 2 O x 18 g H 2 O = 144 g H 2 O 2. 0 g H 2 2 mol H 2 1 mol H 2 O

Limiting Reactant & Percentage Yield n n limiting reactant is the reactant that limits the amount of the other reactant that can combine and the amount of product that can be formed in a chemical reaction. excess reactant is the substance that is NOT completely used up in a chemical reaction.

Sample & Practice Problems See sample problem F on page 313 of textbook. Do practice problems #1 on page 313. See sample problem G on pages 314 -315. Do practice problems #1 & #2 on page 315.

Quick. LAB n Do the Quick. LAB titled “Limiting Reactants in a Recipe” on page 316 of the textbook. Yes, cooking IS chemistry!

Percentage Yield § § theoretical yield is the maximum amount of product that can be produced from a given amount of reactant actual yield of a product is the measured amount of a product obtained from a reaction percentage yield is the ratio of the actual yield to theoretical yield multiplied by 100 percentage yield = actual yield x 100 theoretical yield

Problems n see sample problem H on pages 317 -318 of textbook n Do practice problems #1 & #2 on page 318. n Do section review problems #1 - #4 on page 318. n Do critical thinking problems #37, #38, #39, & #40 on pages 322 & 323.

Chapter 9 test review n 20 multiple choice questions • definitions of composition & reaction stoichiometry • mole ratios: their definition & use • SI units of molar mass • identify mole ratio from balanced formula equation • 5 mole-mole problems • Definitions & practical applications of excess reactant & limiting reactant • definitions & practical applications of theoretical yield, actual yield, & % yield

Honors Chemistry Chapter 9 Test Review n n n n n 25 multiple choice Know the definitions of reaction & composition stoichiometry, mole ratio, and units of molar mass. Know what mole ratio means and how it is used in stoichiometry. Determine mole ratio using balanced formula equation. (2) Perform mole to mole stoichiometric calculations (4). Perform mole to mass stoichiometric calculations (1). Perform mass to mole stoichiometric calculations (1). Perform mass to mass stoichiometric calculations (1). Know definitions and applications of limiting and excess reactants. Know the definitions & applications of actual yield, theoretical yield, and percent yield.

Practice #2 2 Na 3 PO 4 + 3 Ca. SO 4 3 Na 2 SO 4 + Ca 3(PO 4)2 Known = 3 moles Ca. SO 4 unknown = ? Moles Na 3 PO 4 3 x 2/3 = 2 moles Na 3 PO 4 Known = 2. 0 moles Na 3 PO 4 unknown = ? Moles Na 2 SO 4 2 x 3/2 = 3 moles Na 2 SO 4 Known = 1. 5 moles Ca 3(PO 4)2 unknown = ? Moles Ca. SO 4 1. 5 x 3/1 = 4. 5 moles Ca. SO 4 Known = 4. 4 moles Ca. SO 4 unknown = ? Moles Na 3 PO 4 4. 4 x 2/3 = 2. 9 moles Na 3 PO 4

2 Na 3 PO 4 + 3 Ca. SO 4 3 Na 2 SO 4 + Ca 3(PO 4)2 Known = 6. 0 moles Na 3 PO 4 unknown = ? Moles Na 2 SO 4 6. 0 x 3/2 = 9. 0 moles Na 2 SO 4 Known = 5. 4 moles Ca. SO 4 unknown = ? Moles Na 3 PO 4 5. 4 x 2/3 = 3. 6 moles Na 3 PO 4 Known = 0. 6 moles Na 3 PO 4 unknown = ? Moles Ca. SO 4 0. 6 x 3/2 = 0. 9 moles Ca. SO 4

Stoichiometry Practice #3 4 Na 3 N + 3 O 2 6 Na 2 O + 2 N 2 Assume you have 12. 0 moles of Na 3 N. 1) 2) 3) How many moles of O 2 do you need? How many moles of Na 2 O will you get? How many moles of N 2 will you get?

Stoichiometry Practice #3 4 Na 3 N + 3 O 2 6 Na 2 O + 2 N 2 Assume you have 12. 0 moles of Na 3 N. How many moles of O 2 do you need? 12 x 3/4 = 9 moles O 2 How many moles of Na 2 O will you get? 12 x 6/4 = 18 moles Na 2 O How many moles of N 2 will you get? 12 x 2/4 = 6 moles N 2

Practice #3 4 Na + O 2 2 Na 2 O If you have 4 moles of Na, how many grams of O 2 will you need? If you have 64 grams of O 2, how many moles of Na 2 O will you produce? If you have 46 grams of Na, how many grams of O 2 will you need?

Practice #3 4 Na + O 2 2 Na 2 O If you have 4 moles of Na, how many grams of O 2 will you need? 4 x 1/4 x 32 = 32 grams O 2 If you have 64 grams of O 2, how many moles of Na 2 O will you produce? 64/32 x 2/1 = 4 moles Na 2 O If you have 46 grams of Na, how many grams of O 2 will you need? 46/23 x 1/4 x 32 = 16 grams O 2