Stoichiometry Introduction to Stoichiometry Stoichiometry Objectives Define stoichiometry
Stoichiometry Introduction to Stoichiometry

Stoichiometry Objectives Define stoichiometry Describe the importance of the mole ratio in stoichiometric calculations Write a mole ratio relating two substances in a chemical equation

Stoichiometry Definitions Composition stoichiometry deals with the mass relationships of elements in compounds Reaction stoichiometry involves the mass relationships between reactants and products in a chemical reaction

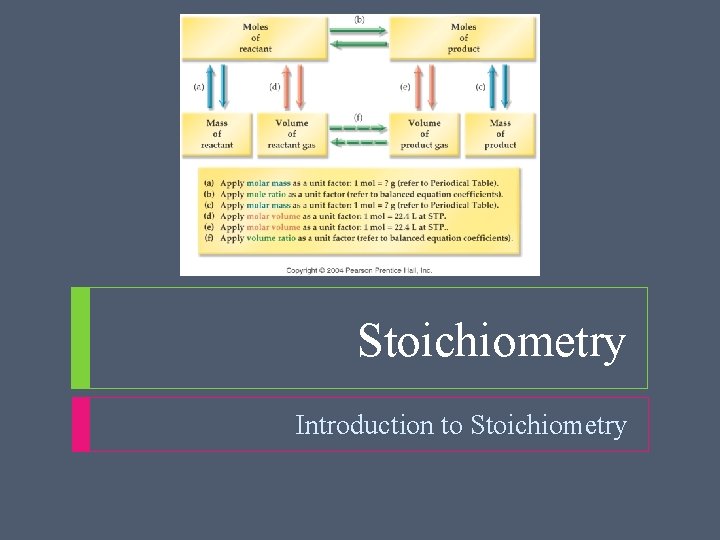
Stoichiometry Reaction Stoichiometry Problems Problem Type 1: Given and unknown quantities are amounts in moles Amount of given substance (mol) Amount of unknown substance (mol)

Stoichiometry Reaction Stoichiometry Problems Problem Type 2: Given is an amount in moles and unknown is a mass Amount of given substance (mol) Amount of unknown substance (mol) Mass of unknown substance (g)

Stoichiometry Reaction Stoichiometry Problems Problem Type 3: Given is a mass and unknown is an amount in moles Mass of given substance (g) Amount of given substance (mol) Amount of unknown substance (mol)

Stoichiometry Reaction Stoichiometry Problems Problem Type 4: Given is a mass and unknown is a mass Mass of a given substance (g) Amount of given substance (mol) Amount of unknown substance (mol) Mass of unknown substance (g)

Stoichiometry Mole Ratio A mole ratio is a conversion factor that relates the amounts in moles of any two substances involved in a chemical reaction 2 Al 2 O 3(l) → 4 Al(s) + 3 O 2(g) 2 mol Al 2 O 3 4 mol Al , 2 mol Al 2 O 3 3 mol O 2 , 4 mol Al 3 mol O 2
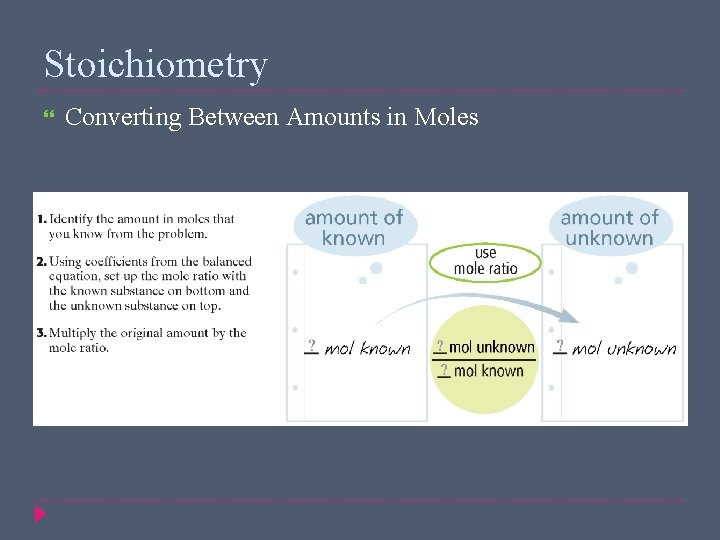
Stoichiometry Converting Between Amounts in Moles
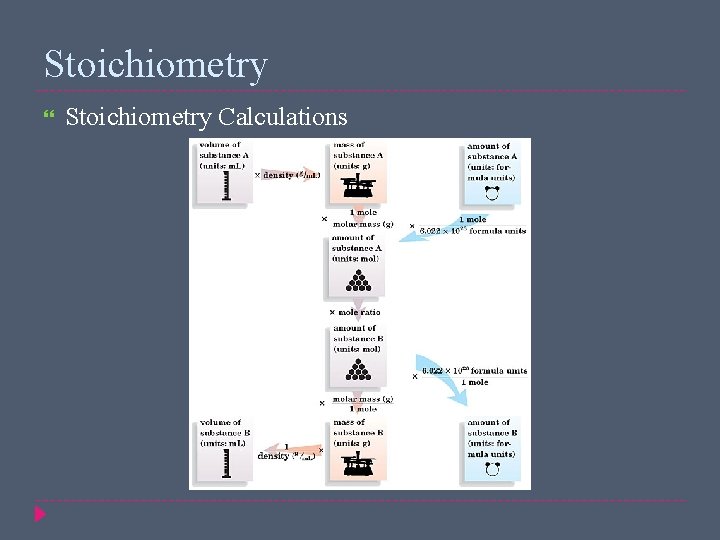
Stoichiometry Calculations

Stoichiometry Calculations Sample Problem For each equation, balance and write all possible mole ratios Hg. O (s) NH 3 (g) + NO (g) Hg (l) + O 2 (g) N 2 (l) + H 2 O (l)

Stoichiometry Calculations Sample Problem How many moles of sodium will react with water to produce 4. 0 mol of hydrogen in the following reaction? Na(s) + H 2 O(l) Na. OH(aq) + H 2(g)

Stoichiometry Calculations Sample Problem How many moles of lithium chloride will be formed by the reaction of chlorine with 0. 046 mol of lithium bromide in the following reaction? Li. Br(aq) + Cl 2(g) Li. Cl(aq) + Br 2(l)

Stoichiometry Calculations Sample Problem Aluminum will react with sulfuric acid in the following reaction. Al(s) + H 2 SO 4(l) Al 2(SO 4)3(aq) + H 2(g) How many moles of H 2 SO 4 will react with 18 mol Al? How many moles of each product will be produced from 18 mol Al?

Stoichiometry Calculations Sample Problem Propane burns in excess oxygen according to the following reaction. C 3 H 8 + O 2 CO 2 + H 2 O How many moles each of CO 2 and H 2 O are formed from 3. 85 mol of propane? If 0. 647 mol of oxygen is used in the burning of propane, how many moles each of CO 2 and H 2 O are produced? How many moles of C 3 H 8 are consumed?
- Slides: 15