Stoichiometry Chapter 3 Email benzene 4 presidentgmail com

Stoichiometry Chapter 3 E-mail: benzene 4 president@gmail. com Web-site: http: //clas. sa. ucsb. edu/staff/terri/

Stoichiometry - Ch. 3 1. For a new element, 67. 16% is an isotope with mass 280. 8 amu, 2. 76% is an isotope with mass 283. 7 amu and 30. 08% is an isotope with mass 284. 8 amu. Calculate the average atomic mass for this new element?

Stoichiometry - Ch. 3 Average Atomic Mass = (fraction of isotope A)(mass of isotope A) + (fraction of isotope B)(mass of isotope B) + etc.

Stoichiometry - Ch. 3 2. For which of the following compounds does 1. 00 g represent 3. 32 × 10 -2 mol? a. NO 2 b. H 2 O c. C 2 H 6 d. NH 3 e. CO
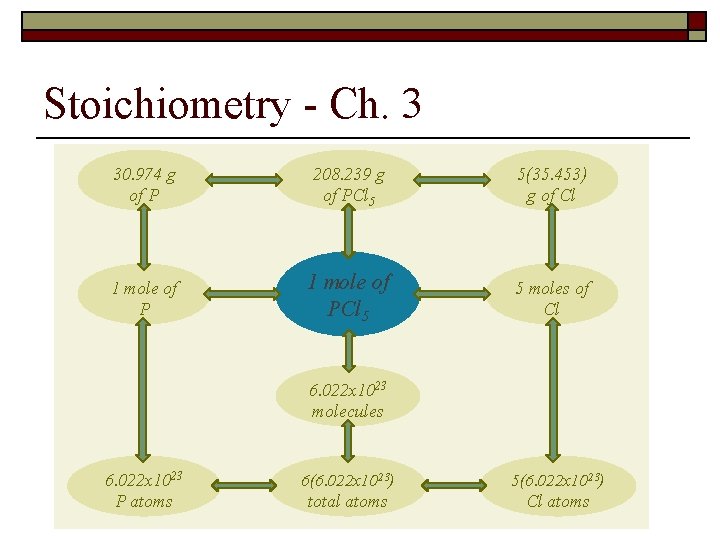
Stoichiometry - Ch. 3 30. 974 g of P 208. 239 g of PCl 5 5(35. 453) g of Cl 1 mole of PCl 5 5 moles of Cl 6. 022 x 1023 molecules 6. 022 x 1023 P atoms 6(6. 022 x 1023) 5(6. 022 x 1023) total atoms Cl atoms

Stoichiometry - Ch. 3 3. If a sample of diatomic element weighs 131. 3 g and contains 4. 162 x 1024 atoms. Identify the element.

Stoichiometry - Ch. 3 4. If you have 0. 63 mg of H 2 SO 4 a. How many H 2 SO 4 molecules are in your sample? b. How many oxygen atoms?

Stoichiometry - Ch. 3 5. An alkali metal oxide contains 83. 01% metal by mass. Determine the identity of the metal oxide. How many grams of oxygen are in a 25. 0 g sample of the metal oxide?

Stoichiometry - Ch. 3 o

Stoichiometry - Ch. 3 6. Compound X 2 Y is 60% X by mass. Calculate the percent Y by mass of the compound XY 3?

Stoichiometry - Ch. 3 7. Tryptophan is 64. 7% carbon, 5. 9% hydrogen, 13. 7% nitrogen and 15. 7% oxygen. What is the empirical formula for tryptophan?

Stoichiometry - Ch. 3 Empirical Formula ⇒ The lowest whole number molar ratio of the elements in a compound 1. Convert given values into moles 2. Divide all moles by the smallest mole value 3. If you have all whole numbers you have the EF – if not try multiplying them all by 2 or 3 etc.

Stoichiometry - Ch. 3 8. The empirical formula for xylene is C 4 H 5 and xylene has a molar mass of 106. 16 g/mol. Determine the molecular formula for xylene.

Stoichiometry - Ch. 3 Molecular Formula ⇒ The actual molar ratio of the elements in a compound – it is some multiple of the empirical formula (x 1, x 2 etc) 1. 2. 3. 4. Derive empirical formula Determine the empirical mass (Molar mass)/(empirical mass) = multiple Multiply the empirical formula by the multiple

Stoichiometry - Ch. 3 9. Consider the following unbalanced reaction: NH 3 + O 2 NO 2 + H 2 O a. How many moles of oxygen gas are required to make 12. 8 moles of nitrogen dioxide? b. How many grams of water can be produced from 9. 64 g of ammonia? c. Identify the limiting reagent if 3 moles of ammonia is combined with 5 moles of oxygen d. Identify the limiting reagent if 10 g of ammonia is combined with 28 g of oxygen

Stoichiometry - Ch. 3 1. 2. 3. 4. Methodology for Reaction Stoichiometry Problems Write a balanced chemical reaction Convert given value(s) into moles (you may have to ID the limiting reagent – next slide) Use reaction coefficients as a molar ratio Convert moles of your unknown into the desired units

Stoichiometry - Ch. 3 Limiting Reagent ⇒ Limits the amount of product that is produced due to running out 1 st The limiting reagent is used to determine the maximum yield of product/s aka theoretical yield and the maximum consumption of reactants Identifying Limiting Reagents: 1. Convert all given values into moles 2. Divide each mole value by the coefficient 3. The smallest number identifies the LR

Stoichiometry - Ch. 3 10. Consider the following unbalanced reaction: P 4 H 10 (s) + H 2 O (l) H 3 PO 4 (s) a. How many grams of H 3 PO 4 can be produced from a mixture of 10 g of each reactant? b. How many grams of excess reactant remains (assume reaction goes to completion)? c. What is the percent yield if 22. 8 g of H 3 PO 4 is produced?

Stoichiometry - Ch. 3 o

Stoichiometry - Ch. 3 11. How many grams of fluorine are required if you want to produce 83 g of PF 3 if the reaction has 63. 2% yield? P 4 + F 2 PF 3 (unbalanced)

Stoichiometry - Ch. 3 12. A 0. 4647 -g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxygen to yield 0. 8635 g of CO 2 and 0. 1767 g of H 2 O. What is the empirical formula of the compound?

Stoichiometry - Ch. 3 You have completed ch. 3

Ch. 3 – Answer Key 1. For a new element, 67. 16% is an isotope with mass 280. 8 amu, 2. 76% is an isotope with mass 283. 7 amu and 30. 08% is an isotope with mass 284. 8 amu. Calculate the average atomic mass for this new element? Average atomic mass = (0. 6716)(280. 8 amu) + (0. 0276)(283. 7 amu) + (0. 3008)(284. 8 amu) = 282. 1 amu 2. For which of the following compounds does 1. 00 g represent 3. 32 × 10 -2 mol? Molar mass is a useful value for identification Molar mass = (1. 00 g)/ (3. 32 × 10 -2 mol) = 30. 1 g/mol => C 2 H 6

Ch. 3 – Answer Key 3. If a sample of diatomic element weighs 131. 3 g and contains 4. 162 x 1024 atoms. Identify the element. Diatomic tells us the formula for the element is X 2. To get the molar mass you need the grams (given) and the moles (not given). 4. 162 x 1024 atoms 1 molecule X 2 1 mole X 2 = 3. 456 mole X 2 2 atoms 6. 022 x 1023 molecules Molar mass = 131. 3 g/3. 456 mole = 38. 0 g/mol => F 2

Ch. 3 – Answer Key 4. If you have 0. 63 mg of H 2 SO 4 a. How many H 2 SO 4 molecules are in your sample? The molar mass of H 2 SO 4 = 2(1. 01) + 32. 06 + 4(16. 0) = 98. 08 g/mol 0. 63 mg 1. 0 g 1 mole 6. 022 x 1023 molecules = 3. 9 x 1018 molecules 1000 mg 98. 08 g H 2 SO 4 1 mole b. How many oxygen atoms are in your sample? 3. 9 x 1018 molecules 4 oxygen atoms = 1. 5 x 10 O atoms 1 H 2 SO 4 molecule

Ch. 3 – Answer Key 5. An alkali metal oxide contains 83. 01% metal by mass. Determine the identity of the metal. How many grams of oxygen are in a 25. 0 g sample of the metal oxide? Since an alkali metal has a charge of 1+ the chemical formula for the metal oxide is M 2 O If the molar mass of the metal is denoted by x ⇒ 83. 01 = ((2 x)/(2 x + 16))(100) x = 39. 1 g/mol => K Potassium is the unknown metal

Ch. 3 – Answer Key 6. Compound X 2 Y is 60% X by mass. Calculate the percent Y by mass of the compound XY 3? If you have 100 g of X 2 Y there would be 60 g of X and 40 g of Y For XY 3 since it has only one X atom you can think of that as ½(60 g) or 30 g of X and since there’s three Y atoms you can think of that as 3(40 g) or 120 g of Y. So in XY 3 there’s 30 g of X for every 120 g of Y – so %Y = (120 g/150 g)100 = 80%

Ch. 3 – Answer Key 7. Tryptophan is 64. 7% carbon, 5. 9% hydrogen, 13. 7% nitrogen and 15. 7% oxygen. What is the empirical formula for tryptophan? First we need to get a moles for the molar ratio If we have 100 g sample of typtophan ⇒ 64. 7 g C/12. 01 g/mol = 5. 347 mol C 5. 9 g H/1. 008 g/mol = 5. 853 mol H 13. 7 g N/14. 01 g/mol = 0. 978 mol N 15. 7 g O/16 g/mol = 0. 981 mol O continue to next slide. . .

Ch. 3 – Answer Key 7. …continued Divide each by the smallest mole value to simplify the ratio ⇒ 5. 347 mol C/0. 978 = 5. 5 mol C 5. 853 mol H/0. 978 = 6 mol H 0. 978 mol N/0. 978 = 1 mol N 0. 981 mol O/0. 978 = 1 mol O Double each to get whole numbers ⇒ 11 mol C: 12 mol H : 2 mol N: 2 mol O Empirical Formula ⇒ C 11 H 12 N 2 O 2

Ch. 3 – Answer Key 8. The empirical formula for xylene is C 4 H 5 and xylene has a molar mass of 106. 16 g/mol. Determine the molecular formula for xylene. First determine the molar mass of the empirical formula ⇒ 4(12. 01)g/mol + 5(1. 008)g/mol = 53. 08 g/mol Divide molar mass of empirical formula into molar mass of the compound ⇒ (106. 16 g/mol)/(53. 08 g/mol) = 2 Multiply the empirical formula by 2⇒ Molecular formula ⇒ C 8 H 10

Ch. 3 – Answer Key 9. Consider the following unbalanced reaction: 4 NH 3 + 7 O 2 4 NO 2 + 6 H 2 O a. How many moles of oxygen gas are required to make 12. 8 moles of nitrogen dioxide? 12. 8 mol NO 2 7 mol O 2 = 22. 4 mol O 2 4 mol NO 2 b. How many grams of water can be produced from 9. 64 g of ammonia? 9. 64 g NH 3 1 mol NH 3 6 mol H 2 O 18. 02 g H 2 O = 15. 3 g H 2 O 17. 04 g NH 3 4 mol NH 3 1 mol H 2 O Continue to next slide…

Ch. 3 – Answer Key 9. …continued c. Identify the limiting reagent if 3 moles of ammonia is combined with 5 moles of oxygen divide each mole by the molar coefficient and look for smaller value (3 mol NH 3)/4 = 0. 75 vs. (5 mol O 2)/7 = 0. 71 ⇒ O 2 is the LR d. Identify the limiting reagent if 10. g of ammonia is combined with 28 g of oxygen convert to moles then divide by molar coefficient (10 g NH 3)/(17. 034 g/mol) = 0. 587 mol NH 3/4 = 0. 147 vs. (28 g O 2)/(32 g/mol) = 0. 875 mol O 2/7 = 0. 125 ⇒ O 2 is the LR

Ch. 3 – Answer Key o

Ch. 3 – Answer Key o

Ch. 3 – Answer Key 11. How many grams of fluorine are needed to produce 83 g of phosphorus trifluoride, if the reaction has 63. 2% yield? P 4 + F 2 PF 3 (unbalanced) First ⇒ balance reaction ⇒ P 4 + 6 F 2 4 PF 3 Determine theoretical yield from the actual and the % yield ⇒ 63. 2% = (83 g/Theo)x 100 ⇒ Theoretical yield = 131. 3 g PF 3 Use theoretical yield to determine grams of F 2 131. 3 g PF 3 1 mol PF 3 6 mol F 2 38 g F 2 = 85. 1 g F 2 87. 97 g PF 3 4 mol PF 3 1 mol F 2

Ch. 3 – Answer Key 12. A 0. 4647 -g sample of a compound known to contain only carbon, hydrogen, and oxygen was burned in oxygen to yield 0. 8635 g of CO 2 and 0. 1767 g of H 2 O. What is the empirical formula of the compound? Cx. Hy. Oz + O 2 CO 2 + H 2 O ? All of the carbon in the compound will end up in the CO 2 and all of the hydrogen will end up in the water the oxygen is unpredictable so we need determine how much C and H there is in our compound …continue to next slide

Ch. 3 – Answer Key 12. …continued C ⇒ 0. 8635 g CO 2 12. 01 g C = 0. 2356 g C 44. 01 g CO 2 H ⇒ 0. 1767 g H 2 O 2. 016 g H = 0. 01977 g H 18. 016 g H 2 O O ⇒ 0. 4647 g cmpd – 0. 2356 g C – 0. 01977 g H = 0. 2093 g O …continue to next slide

Ch. 3 – Answer Key 12. …continued Covert each to moles and divide by smallest value ⇒ (0. 2356 g C)/(12. 01 g/mol) = 0. 01962 mol C/0. 01308 = 1. 5 mol C (0. 01977 g H)/(1. 008 g/mol) = 0. 01961 mol H/0. 01308 = 1. 5 mol H (0. 2093 g O)/(16 g/mol) = 0. 01308 mol O/0. 01308 = 1 mol O Double each value 3 mol C: 3 mol H: 2 mol O Empirical formula ⇒ C 3 H 3 O 2
- Slides: 38