Chapter 11 Stoichiometry Stoichiometry Basics DEFINITION Stoichiometry is

Chapter 11 - Stoichiometry

Stoichiometry Basics DEFINITION - Stoichiometry is the study of the quantitative relationships that exist in chemical formulas and balanced chemical equations. “stoicheion” - element “metron” - measure Stoichiometry allows us to measure amounts of substances that are related by a balanced chemical equation. Chemical reactions are like a recipe

Interpreting Balanced Equations Balance the equation. . . Cu + Ag. NO 3 --> Cu(NO 3)2 + Ag

Interpreting Balanced Equations Balance the equation. . . Cu + 2 Ag. NO 3 --> Cu(NO 3)2 + 2 Ag This means that 1 mole of copper reacts with 2 moles of silver nitrate to form 1 mole of copper (II) nitrate and 2 moles of silver.

Mole - Mole Problems Balance the equation. . . Li + N 2 --> Li 3 N

Mole - Mole Problems Balance the equation. . . 6 Li + N 2 --> 2 Li 3 N EX 1 : How many moles of Li 3 N can be produced when 3. 55 moles of Lithium react with nitrogen?

Example 2 6 Li + N 2 --> 2 Li 3 N EX 2: How many moles of Li 3 N can be produced when 56. 8 grams of Lithium react with nitrogen?

Mass - Mass Conversions Mass-Mass conversions follow a very specific roadmap. . . EX 1 : How many grams of Sn. F 2 can be formed from 12. 4 g of HF? Sn + 2 HF --> Sn. F 2 + H 2

Volume - Volume Conversions Volume-Volume conversions also follow a very specific roadmap. . . EX 1 : How many Liters of H 2 O(g) can be formed from 48. 9 L of CH 4(g) at STP? CH 4 + O 2 --> CO 2 + H 2 O

Volume - Volume Conversions Volume-Volume also conversions follow a very specific roadmap. . . EX 1 : How many Liters of H 2 O(g) can be formed from 48. 9 L of CH 4(g) at STP? CH 4 + 2 O 2 --> CO 2 + 2 H 2 O
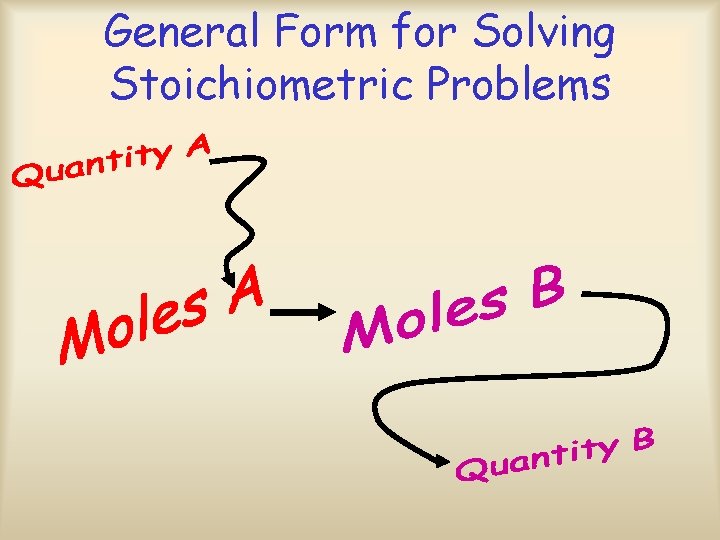
General Form for Solving Stoichiometric Problems

Percent Yield To this point, we have assumed that nothing “goes wrong” in our reactions. We know better! Stoichiometry gives us the amount we should get. This is called theoretical or expected yield. In the lab, we often get different results. What we get in the lab is called the actual or measured yield.

Percent Yield Theoretical Yield - The amount you should get based upon stoichiometry. Actual Yield - The amount you got in the lab.

Percent Yield Example In the lab, you reacted silver nitrate with 32. 5 g of magnesium. When the reaction was complete, you had formed 213 g of silver. What was the percent yield of the experiment?

Limiting Reagents (Reactants) Limiting Reagent - The reactant that will run out first. It limits the amount of products that can be made. Excess Reagent(s) - The reactant(s) that there are more than enough of. Will not run out.

Limiting Reagent Steps • STEP 1 : Balance the equation. • STEP 2 : Convert all reactant quantities to grams or Liters of a product. • STEP 3 : The reactant that would produce the least amount of product is the limiting reagent. The other(s) are excess reagent(s). • STEP 4 : Use only the limiting reagent to calculate all further values.

Limiting Reagent Example EX : How many grams of water can be formed by the reaction of 25 g of CH 4 with 55 L of O 2 at STP? STEP 1 : Balance the equation. CH 4 + 2 O O 2 --> CO 2 + 2 H H 2 O STEP 2 : Answer the question using each reactant quantity.

Limiting Reagent Example STEP 2 : Answer the question using each reactant quantity. STEP 3 : The reactant that makes the least amount of product is the limiting reagent!

Limiting Reagent Example STEP 3 : The reactant that makes the least amount of product is the limiting reagent! STEP 4 : Use only the limiting reagent to calculate all further values.

Limiting Reagents: Example 2 • Ex 2: If 6. 35 g of Al reacts with 86. 5 g of Cu. SO 4: – A) Identify the limiting reagent – B) Calculate the mass of Cu produced – C) Calculate the mass of excess reagent remaining
- Slides: 20