STOICHIOMETRY THEORY Stoichiometry deals with relations between the

STOICHIOMETRY

THEORY: Stoichiometry deals with relations between the mass and chemical reactions. There are two types of chemical formulas; Molecular Formula: A molecular formula of a compound is based on the actual numbers of atoms that make up a molecule of that compound. Simplest Formula: A formula, which is written using the simplest whole number ratio, is called the simplest formula or empirical formula.

Purpose: • Determination of the reaction stoichiometry. • Determination the limiting reagent in a reaction.

Introduction: In this experiment, we will be investigating reaction stoichiometry by doing a series of mixing experiments using Ni. SO 4. 6 H 2 O and Dimethylglyoxime (DMG) solution.

Experiment: Nickel (II) forms a precipitate with the organic compound DMG. The formation of the red chelate occur quantitatively in a solution in which the p. H is buffered in the range of 5 to 9. The chelation reaction that occurs in illustrated next slayt.
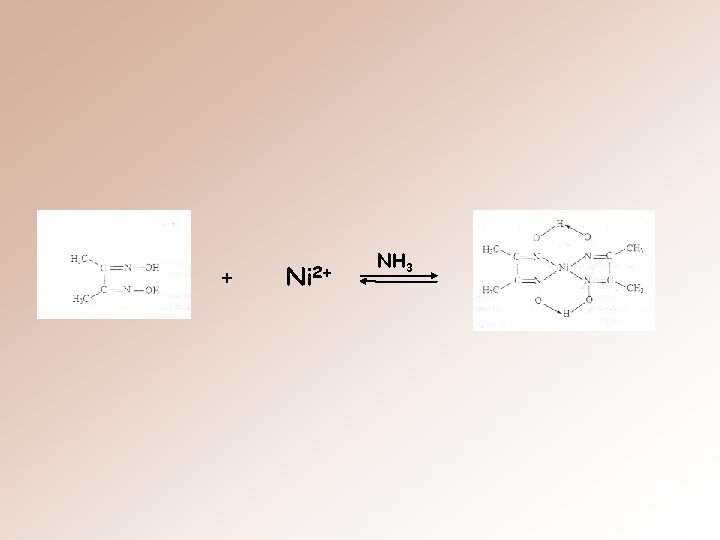
+ Ni 2+ NH 3

Although the loss of one proton occurs from one oxime group on each of the two molecules at DMG, the chelation reaction occurs due to donation of the electron pairs on the four nitrogen atoms, not by electrons the oxygen atoms.

The reaction is performed in a solution buffered by either an ammonia or citrate buffer to prevent the p. H of the solution from falling below 5. If the p. H becomes too low, the equilibrium of the above reaction favors the formation of the nickel(II) ion, causing the dissolution of Ni(DMG)2 back into the mother solution.

An alcoholic solution of DMG is used as the precipitating reagent during the experiment because DMG is only slightly soluble in water. It is therefore crucial to avoid the addition of too large an excess of DMG because it may crystallize out with the chelate.
- Slides: 9