Stoichiometry Dr Ron Rusay Chemical Stoichiometry Stoichiometry is


























- Slides: 34

Stoichiometry Dr. Ron Rusay

Chemical Stoichiometry ð Stoichiometry is the study of mass in chemical reactions. It deals with both reactants and products. ð It quantitatively and empirically relates the behavior of atoms and molecules in a balanced chemical equation to observable chemical change and measurable mass effects. ð It accounts for mass and the conservation of mass, just as the conservation of atoms in a balanced chemical equation.

Chemical Reactions Atoms, Mass & Balance: eg. Zn(s) + S(s)

Stoichiometry Must begin with a correctly balanced equation: _ C 2 H 5 OH + _ O 2 _ CO 2 + _ H 2 O Reactants Products • C=2; H =5+1=6; O=2+1 • C=1; H=2; O=2+1 1 C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O

https: //phet. colorado. edu/sims/html/balancing-chemicalequations/latest/balancing-chemical-equations_en. html

Chemical Equation C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O The balanced equation can be completely stated as: ð 1 mole of ethanol reacts with 3 moles of oxygen to produce 2 moles of carbon dioxide and 3 moles of water.

Chemical Equation All Balanced Equations relate on a molar mass basis. For example the combustion of octane: 2 C 8 H 18(l)+ 25 O 2(g) 16 CO 2(g) +18 H 2 O(l) • moles of octane react with 25 moles of oxygen to produce 16 moles of carbon dioxide and 18 moles of water.

The Chemical Equation: Mole & Masses C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O ð 46 g (1 mole) of ethanol reacts with 3 moles of oxygen (96 g) to produce 2 moles of carbon dioxide and 3 moles of water. ð How many grams of carbon dioxide and water are respectively produced from 46 g (1 mole) of ethanol ? 2 mol x 44 g/mol = 88 g 3 mol x 18 g/mol = 54 g

The Chemical Equation: Moles & Masses • C 2 H 5 OH + 3 O 2 2 CO 2 + 3 H 2 O ð How many grams of oxygen are needed to react with 15. 3 g of ethanol in a 12 oz. beer ? 15. 3 gethanol x molethanol /46. 0 gethanol = 0. 333 molethanol x 3 moloxygen / 1 molethanol = 0. 999 moloxygen 32. 0 goxygen /moloxygen x 0. 999 moloxygen = 32. 0 goxygen NOTE: It takes approximately 1 hour for the biologically equivalent amount of oxygen available from cytochrome p 450 to consume the alcohol in a human in 1 beer to a level below the legal limit of 0. 08%.

Chemical Stoichiometry Epsom salt (magnesium sulfate heptahydrate) is one of five possible hydrates: mono-, di-, tri-, hexa-, or hepta- hydrate. ð How can stoichiometry be used to determine, which hydrate is present in a pure unknown sample, by heating the sample in a kitchen oven at 400 o C for 45 minutes? ð Mg. SO 4. x H 2 O(s) Mg. SO 4 (s) + x H 2 O (g) Refer to % Hydrate Lab Experiment for answer.

Mass Calculations All Balanced Equations relate on a molar and mass basis. For the combustion of octane: 2 C 8 H 18(l)+ 25 O 2(g) 16 CO 2(g) +18 H 2 O(l) 228 g of octane (2 moles)* will react with 800 g of oxygen (25 moles) to produce (16 moles) 704 g of carbon dioxide and (18 moles) 324 g of water. *(2 moles octane x 114 g/mol = 228 g )

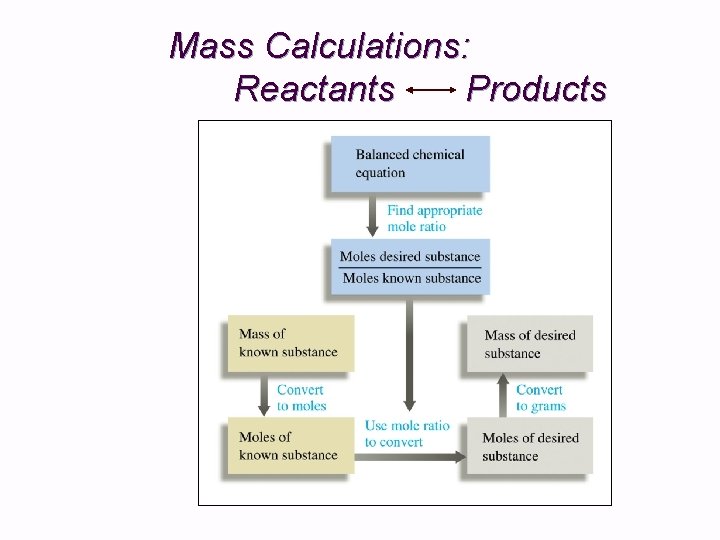
Mass Calculations: Reactants Products

Mass Calculations: Reactants Products 1. 2. 3. 4. 5. Balance the chemical equation. Convert mass of reactant or product to moles. Identify mole ratios in balanced equation: They serve as the “Gatekeeper”. Calculate moles of desired product or reactant. Convert moles to grams.

Mass Calculations: Reactants Products

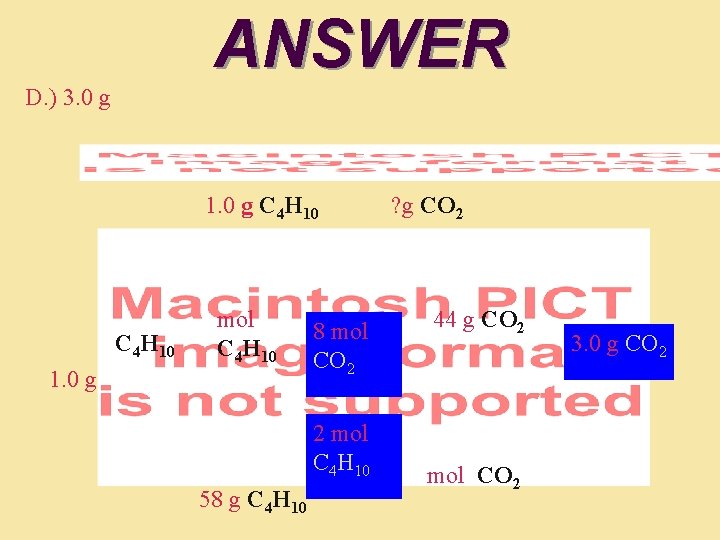
QUESTION The fuel in small portable lighters is butane (C 4 H 10). After using a lighter for a few minutes, 1. 0 gram of fuel was used. How many moles of carbon dioxide would it produce? • • 58 moles 0. 017 moles 1. 7 10– 24 moles 0. 068 moles

ANSWER The fuel in small portable lighters is butane (C 4 H 10). After using a lighter for a few minutes, 1. 0 gram of fuel was used. How many moles of carbon dioxide would it produce? A. B. C. D. 58 moles 0. 017 moles 1. 7 10– 24 moles 0. 068 moles The molar mass of butane: 4 12 g/mol = 48 for carbon + 10 1 g/mol = 10. 0 for hydrogen. Total = 48. 0 + 10. 0 = 58. 0 g/mol. Next; 1. 0 gram of butane 1 mol/58. 0 g = 0. 017 mol

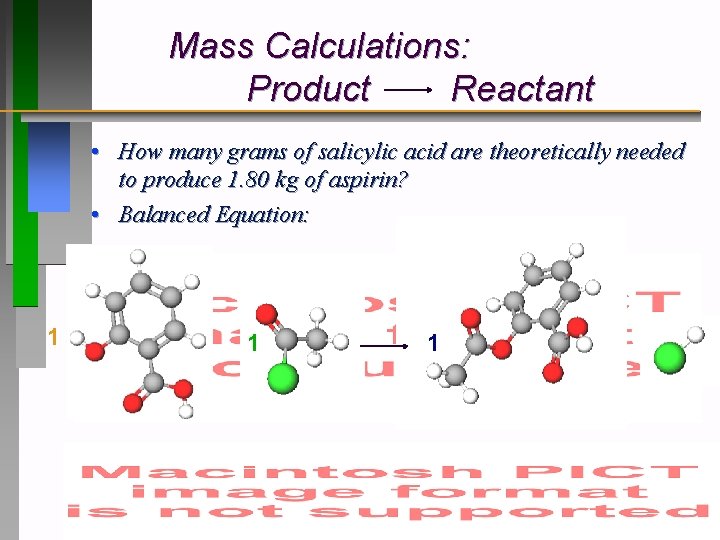
Mass Calculations: Reactants Products • How many grams of salicylic acid are theoretically needed to produce 1. 80 kg of aspirin? • Balanced Equation: 1 1

Mass Calculations: Product Reactant • How many grams of salicylic acid are theoretically needed to produce 1. 80 kg of aspirin? • Balanced Equation: 1 1 1

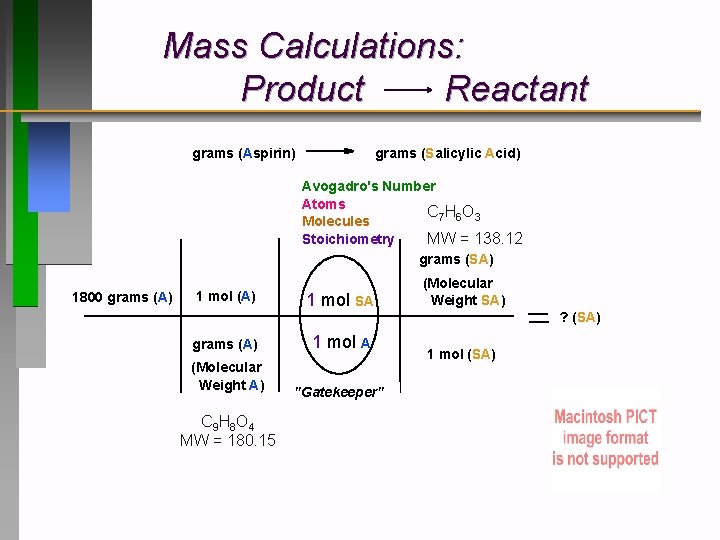
Mass Calculations: Product Reactant grams (Aspirin) grams (Salicylic Acid) Avogadro's Number Atoms C 7 H 6 O 3 Molecules Stoichiometry MW = 138. 12 grams (SA) 1800 grams (A) 1 mol SA grams (A) 1 mol A (Molecular Weight A) "Gatekeeper" C 9 H 8 O 4 MW = 180. 15 (Molecular Weight SA) ? (SA) 1 mol (SA)

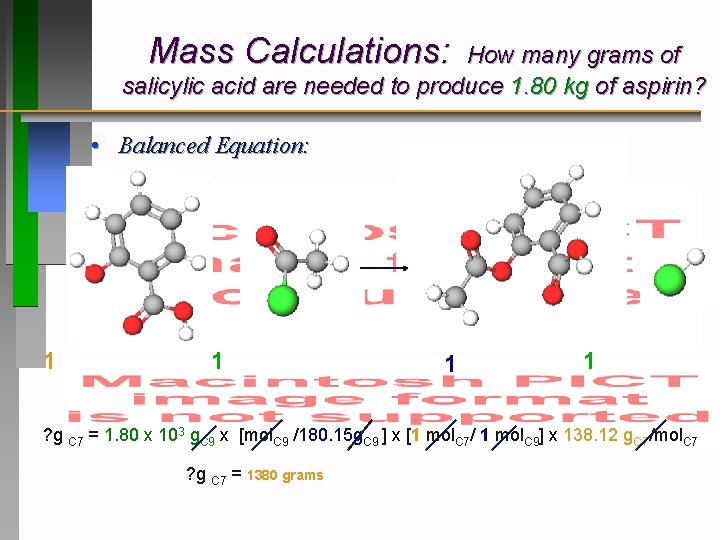
Mass Calculations: How many grams of salicylic acid are needed to produce 1. 80 kg of aspirin? • Balanced Equation: 1 1 ? g C 7 = 1. 80 x 103 g. C 9 x [mol. C 9 /180. 15 g. C 9 ] x [1 mol. C 7/ 1 mol. C 9] x 138. 12 g. C 7/mol. C 7 ? g C 7 = 1380 grams

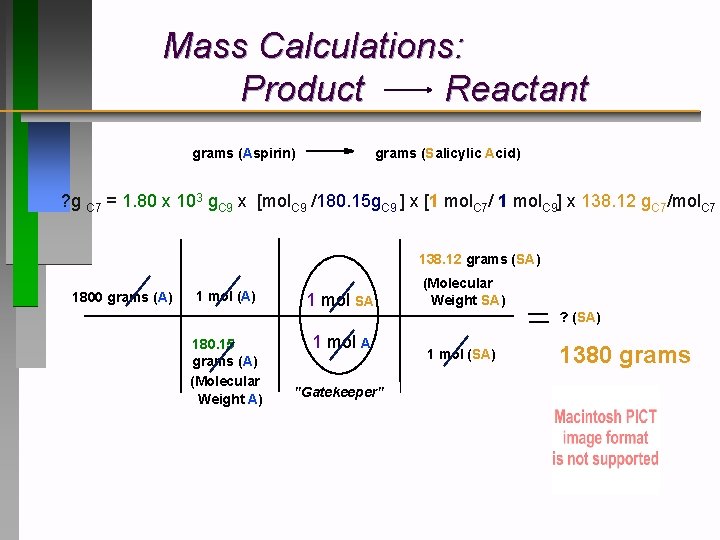
Mass Calculations: Product Reactant grams (Aspirin) grams (Salicylic Acid) ? g C 7 = 1. 80 x 103 g. C 9 x [mol. C 9 /180. 15 g. C 9 ] x [1 mol. C 7/ 1 mol. C 9] x 138. 12 g. C 7/mol. C 7 138. 12 grams (SA) 1800 grams (A) 1 mol (A) 180. 15 grams (A) (Molecular Weight A) 1 mol SA 1 mol A "Gatekeeper" (Molecular Weight SA) ? (SA) 1 mol (SA) 1380 grams

Mass Calculations: Reactants Products

QUESTION The fuel in small portable lighters is butane (C 4 H 10). After using a lighter for a few minutes, 1. 0 gram (0. 017 moles) of fuel was used. How many grams of carbon dioxide would it produce? How many grams of carbon dioxide would this produce? A. ) 750 mg B. ) 6. 0 g C) 1. 5 g D. ) 3. 0 g

ANSWER D. ) 3. 0 g 1. 0 g C 4 H 10 mol C 4 H 10 1. 0 g 8 mol CO 2 2 mol C 4 H 10 58 g C 4 H 10 ? g CO 2 44 g CO 2 mol CO 2 3. 0 g CO 2 ? g CO 2

Percent Yield In synthesis as in any experimentation, it is very difficult and at most times impossible to be perfect. Therefore the actual yield (g) is measured and compared to theoretical calculated yield (g). This is the percent yield: % Yield = actual (g) / theoretical (g) x 100 Some DVC students may report percent yields greater than 100% in their first synthesis experiment. Hmmm? . . . . Why is this not possible? How would Walter White do?

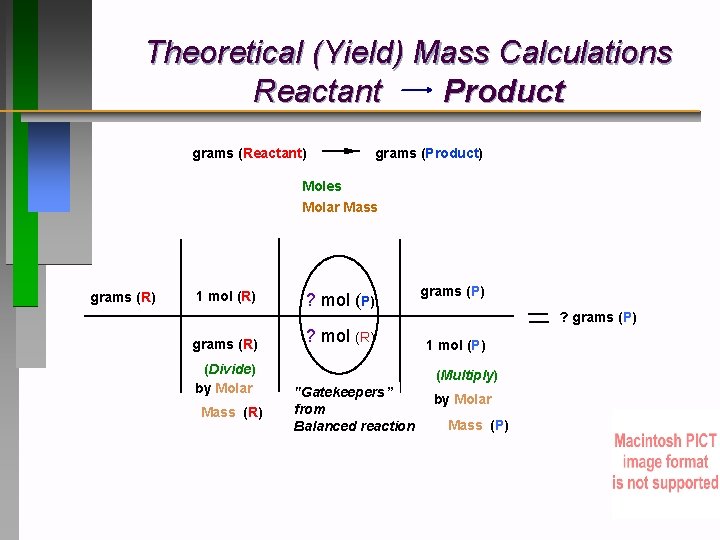
Theoretical (Yield) Mass Calculations Reactant Product grams (Reactant) grams (Product) Moles Molar Mass grams (R) 1 mol (R) grams (R) (Divide) by Molar Mass (R) ? mol (P) ? mol (R) grams (P) ? grams (P) 1 mol (P) (Multiply) "Gatekeepers” from Balanced reaction by Molar Mass (P)

QUESTION A synthetic reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction (at least in Europe), the calculated theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% C) 80. 3% D) 160. 6%

ANSWER If a reaction produced 2. 45 g of Ibogaine, C 20 H 26 N 2 O, a natural product with strong promise in treating heroin addiction, and theoretical yield was 3. 05 g, what is the % yield? A) 19. 7% B) 39. 4% % yield = 2. 45 g / 3. 05 g x 100 = 80. 3% C) 80. 3% D) 160. 6%

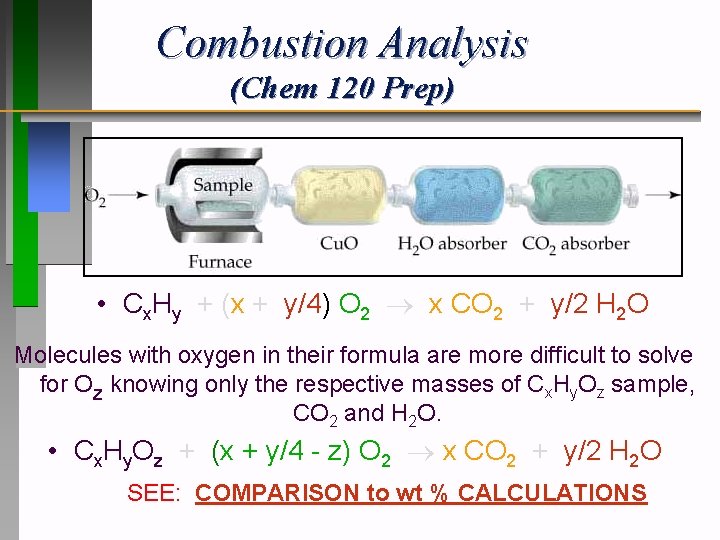
Combustion Analysis (Chem 120 Prep) • Cx. Hy + (x + y/4) O 2 x CO 2 + y/2 H 2 O Molecules with oxygen in their formula are more difficult to solve for Oz knowing only the respective masses of Cx. Hy. Oz sample, CO 2 and H 2 O. • Cx. Hy. Oz + (x + y/4 - z) O 2 x CO 2 + y/2 H 2 O SEE: COMPARISON to wt % CALCULATIONS

Combustion Analysis Calculation Ascorbic Acid ( Vitamin C ) • Combustion of a 6. 49 mg sample in excess oxygen, yielded 9. 74 mg CO 2 and 2. 64 mg H 2 O • Calculate it’s Empirical formula! • C: 9. 74 x 10 -3 g CO 2 x(12. 01 g C/44. 01 g CO 2) = 2. 65 x 10 -3 g C • H: 2. 64 x 10 -3 g H 2 O x (2. 016 g H 2/18. 02 g. H 2 O) = 2. 92 x 10 -4 g H • Mass Oxygen = 6. 49 mg - 2. 65 mg - 0. 30 mg = 3. 54 mg O

Vitamin C: Calculation (continued) • C = 2. 65 x 10 -3 g C / ( 12. 01 g C / mol C ) = = 2. 21 x 10 -4 mol C • H = 0. 295 x 10 -3 g H / ( 1. 008 g H / mol H ) = = 2. 92 x 10 -4 mol H • O = 3. 54 x 10 -3 g O / ( 16. 00 g O / mol O ) = = 2. 21 x 10 -4 mol O • Divide each by 2. 21 x 10 -4 • C = 1. 00 Multiply each by 3 = 3. 00 = 3. 0 • H = 1. 32 = 3. 96 = 4. 0 • O = 1. 00 = 3. 0 C 3 H 4 O 3

QUESTION Erythrose contains carbon, hydrogen and oxygen (MM = 120. 0 g/mol). It is an important sugar that is used in many chemical syntheses. Combustion analysis of a 700. 0 mg sample yielded 1. 027 g CO 2 and 0. 4194 g H 2 O. Mass Spectrometry produced a molecular ion @ 120 mass units (m/z). What is the molecular formula of erythrose? A) B) C) D) CH 2 O C 6 H 12 O 6 C 3 H 6 O 3 C 4 H 8 O 4

ANSWER Alternative Solution Method Mass ratio of C in CO 2 = D) C 4 H 8 O 4 mol C x M of C = mass of 1 mol CO 2 = 1 mol C x 12. 01 g C/ 1 mol C = 44. 01 g CO 2 0. 2729 g C / 1 g CO 2 mol H x M of H = mass of 1 mol H 2 O 2 mol H x 1. 008 g H / 1 mol H = = 0. 1119 g H / 1 g H 2 O 18. 02 g H 2 O Mass ratio of H in H 2 O = Calculating masses of C and H: Mass of Element = mass of compound x mass ratio of element

Alternative Method Mass (g) of C = 1. 027 g CO 2 x 0. 2729 g C 1 g CO 2 = 0. 2803 g C 0. 1119 g H Mass (g) of H = 0. 4194 g H 2 O x = 0. 04693 g H 1 g H 2 O Calculating the mass of O: Mass (g) of O = Sample mass -( mass of C + mass of H ) = 0. 700 g - 0. 2803 g C - 0. 04693 g H = 0. 37277 g O Calculating moles of each element: C = 0. 2803 g C / 12. 01 g C/ mol C = 0. 02334 mol C H = 0. 04693 g H / 1. 008 g H / mol H = 0. 04656 mol H O = 0. 37277 g O / 16. 00 g O / mol O = 0. 02330 mol O C 0. 02334 H 0. 04656 O 0. 02330 = CH 2 O formula weight = 30 g / formula 120 g /mol / 30 g / formula = 4 formula units / cmpd = C 4 H 8 O 4