Chemical Quantities and Chemical Reactions Chemical Quantities Measuring



















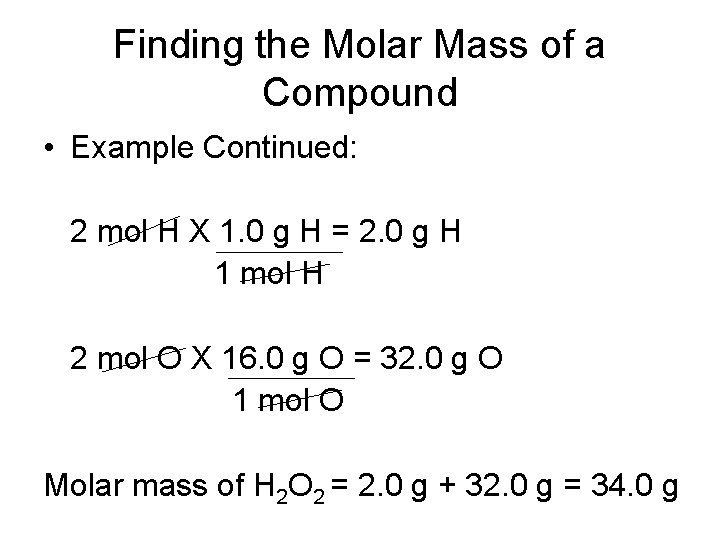













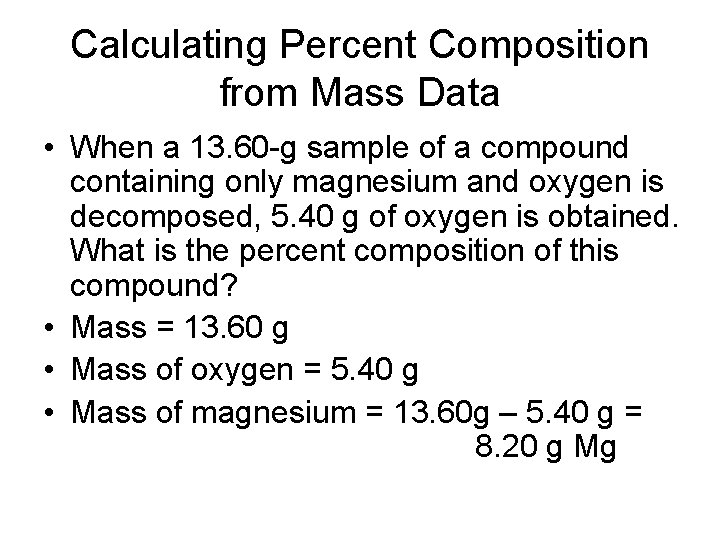
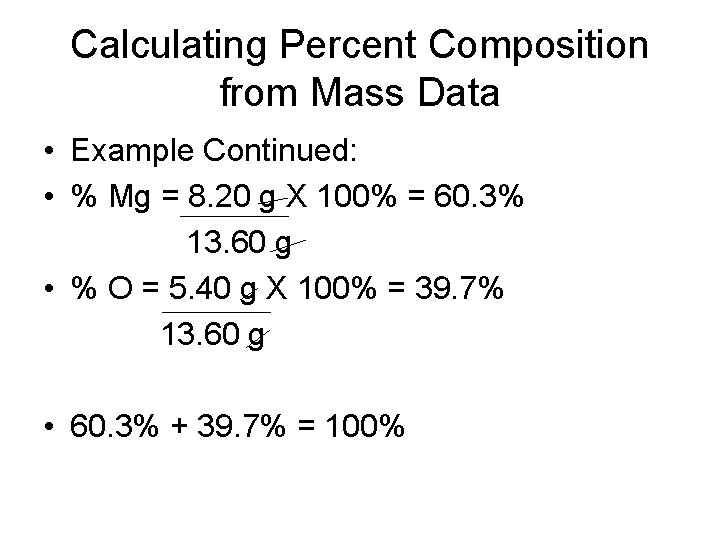

















































- Slides: 94

Chemical Quantities and Chemical Reactions

Chemical Quantities

Measuring Matter • Often measure the amount of something by one of three different methods: –count –mass –volume

Measuring Matter • By count: - Example: 1 dozen apples = 12 apples • By mass: - Example: 1 dozen apples = 2. 0 kg apples • By volume: - Example: 1 dozen apples = 0. 20 bushel apples

Measuring Matter • Can convert among units: • Example: 1 dozen apples 12 apples 1 dozen apples 0. 20 bushel apples 2. 0 kg apples 1 dozen apples

Finding Mass from a Count • What is the mass of 90 average-sized apples if 1 dozen of the apples has a mass of 2. 0 kg? • # apples dozens apples mass apples • Conversion factors: 1 dozen apples 12 apples 2. 0 kg apples 1 dozen apples

Finding Mass from a Count 90 apples X 1 dozen apples X 2. 0 kg apples 12 apples 1 dozen apples = 15 kg apples

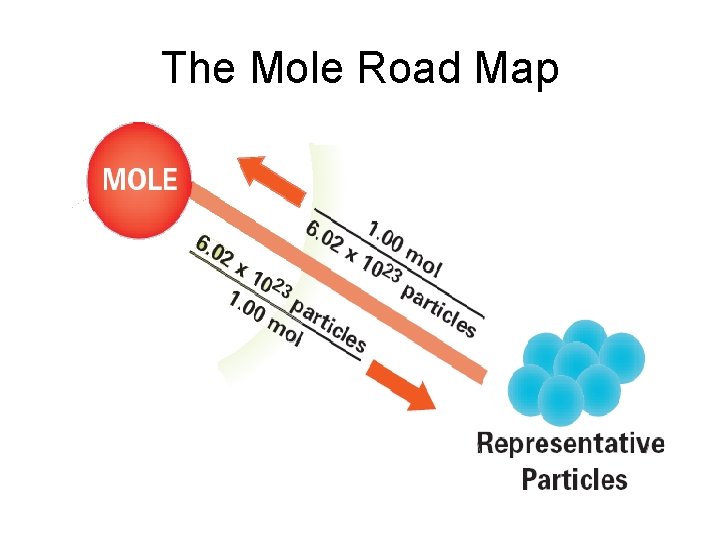
What is a Mole? • mole - (mol) 6. 02 1023 representative particles of a substance - SI unit for measuring the amount of a substance -The number 6. 02 1023 is called Avogadro’s number (Italian scientist) - helped clarify difference between atoms and molecules

What is a Mole? • Mole of any substance contains Avogadro’s number of representative particles, or 6. 02 1023 representative particles • Representative particle - refers to species present in a substance - usually atoms, molecules, or formula units

What is a Mole?

Converting Number of Atoms to Moles • Magnesium is a light metal used in the manufacture of aircraft, automobile wheels, tools, and garden furniture. How many moles of magnesium is 1. 25 X 1023 atoms of magnesium? • Conversion factors: 1 mol Mg 6. 02 X 10 23 atoms Mg

Converting Number of Atoms to Moles 1. 25 X 10 23 atoms Mg X 1 mol Mg 6. 02 X 1023 atoms Mg = 2. 08 X 10 -1 mol Mg = 0. 208 mol Mg

What is a Mole? • Converting Moles to Number of Particles

Converting Moles to Number of Atoms • Propane is a gas used for cooking and heating. How many atoms are in 2. 12 mol of propane (C 3 H 8)? • Conversion factors: 6. 02 X 1023 molecules C 3 H 8 1 mol C 3 H 8 11 atoms 1 molecule C 3 H 8

Converting Moles to Number of Atoms • Example Continued: 2. 12 mol C 3 H 8 X 6. 02 X 1023 molecules C 3 H 8 1 mol C 3 H 8 X 11 atoms 1 molecule C 3 H 8 = 1. 4039 X 1025 atoms = 1. 40 X 1025 atoms

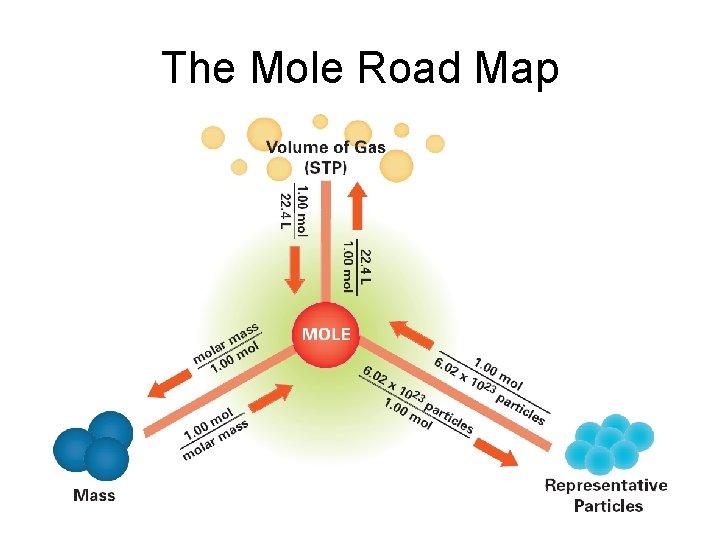
The Mass of a Mole of an Element • Atomic mass of an element expressed in grams is the mass of a mole of the element • Molar mass- mass of a mole of an element - Examples: molar mass of Carbon = 12. 0 g molar mass of Hydrogen = 1. 0 g

The Mass of a Mole of an Element

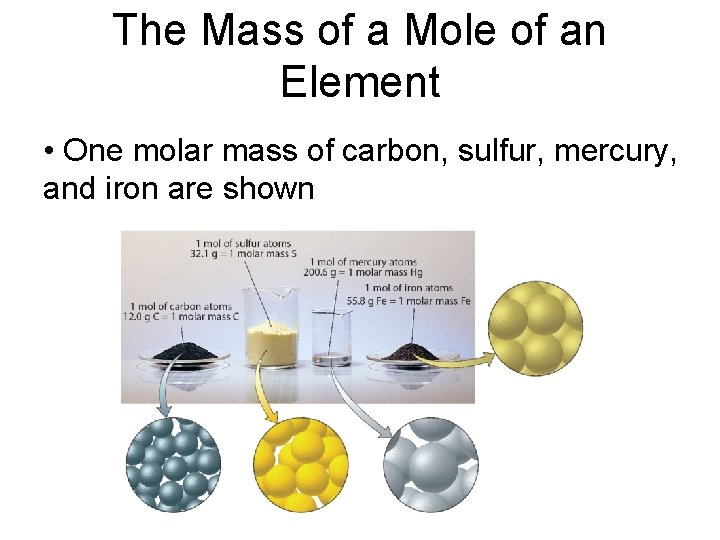
The Mass of a Mole of an Element • One molar mass of carbon, sulfur, mercury, and iron are shown

The Mass of a Mole of an Element • You must know formula of compound to calculate the mass of a mole of an element • To calculate the molar mass of a compound: - find the number of grams of each element in one mole of the compound - add the masses of the elements in the compound

Finding the Molar Mass of a Compound • The decomposition of hydrogen peroxide (H 2 O 2) provides sufficient energy to launch a rocket. What is the molar mass of hydrogen peroxide? • 1 mole H = 1. 0 g H • 1 mol O = 16. 0 g O
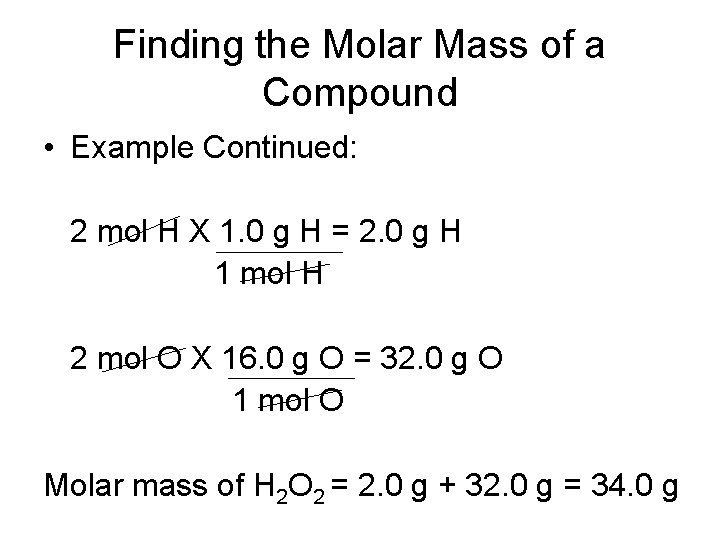
Finding the Molar Mass of a Compound • Example Continued: 2 mol H X 1. 0 g H = 2. 0 g H 1 mol H 2 mol O X 16. 0 g O = 32. 0 g O 1 mol O Molar mass of H 2 O 2 = 2. 0 g + 32. 0 g = 34. 0 g

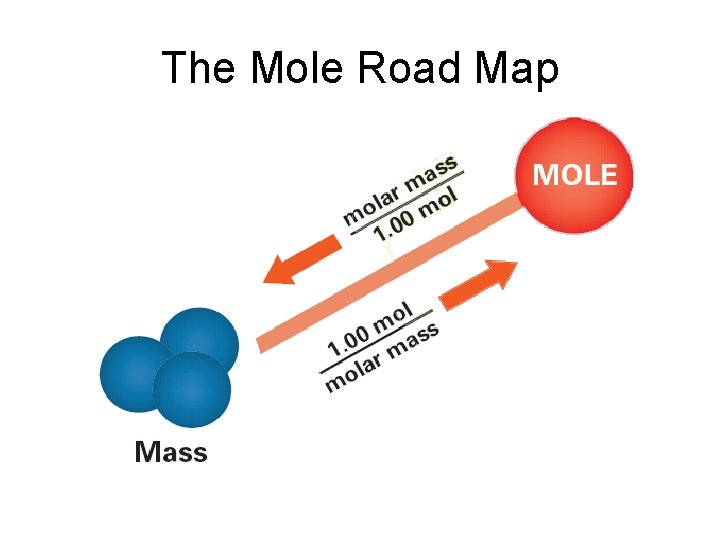
The Mole-Mass Relationship • It’s very important to use the formula of the substance when calculating molar mass • Use molar mass of an element or compound to convert between the mass of a substance and the moles of a substance

Converting Moles to Mass • The aluminum satellite dishes are resistant to corrosion because aluminum reacts with oxygen in the air to form a coating of aluminum oxide (Al 2 O 3). This tough, resistant coating prevents any further corrosion. What is the mass of 9. 45 mol of aluminum oxide? • Moles = 9. 45 mol Al 2 O 3 • 1 mol Al 2 O 3 = 102. 0 g Al 2 O 3

Converting Moles to Mass • Example Continued: mass = 9. 45 mol Al 2 O 3 X 102. 0 g Al 2 O 3 1 mol Al 2 O 3 = 964 g Al 2 O 3

Converting Mass to Moles • When iron is exposed to air, it corrodes to form red-brown rust. Rust is iron(III) oxide (Fe 2 O 3). How many moles of iron(III) oxide are contained in 92. 2 g of pure Fe 2 O 3? • Mass = 92. 2 g Fe 2 O 3 • 1 mol Fe 2 O 3 = 159. 6 g Fe 2 O 3

Converting Mass to Moles • Example Continued: Moles = 92. 2 g Fe 2 O 3 X 1 mol Fe 2 O 3 159. 6 g Fe 2 O 3 = 0. 578 mol Fe 2 O 3

Mole-Volume Relationship • Avogadro’s hypothesis – equal volumes of gases at same temperature and pressure contain equal numbers of particles

Mole–Volume Relationship • Volume of a gas varies with temperature and pressure - Example: balloon filled with helium shrinks when outside on a cold day - The volume of a gas is usually measured at a standard temperature and pressure • Standard temperature and pressure (STP) - means a temperature of 0°C and a pressure of 101. 3 k. Pa, or 1 atmosphere (atm)

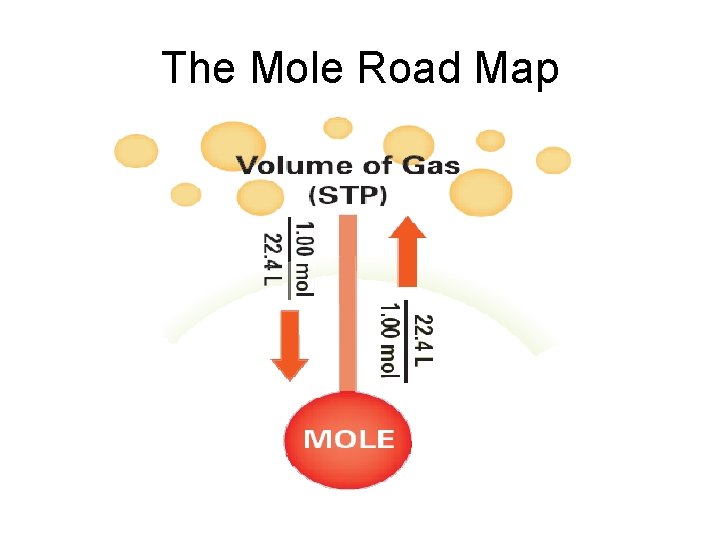
Mole-Volume Relationship • At STP, 1 mol or, 6. 02 1023 representative particles, of any gas occupies a volume of 22. 4 L • Molar volume – quantity of 22. 4 L

Calculating the Volume of Gas at STP • Sulfur dioxide (SO 2) is a gas produced by burning coal. It is an air pollutant and one of the causes of acid rain. Determine the volume, in liters of 0. 60 mol SO 2 gas at STP. • Moles = 0. 60 mol SO 2 • 1 mol SO 2 = 22. 4 L SO 2 • Conversion factor: 22. 4 L SO 2 1 mol SO 2

Calculating the Volume of a Gas at STP • Example Continued: Volume = 0. 60 mol SO 2 X 22. 4 L SO 2 1 mol SO 2 = 13 L SO 2

The Mole–Volume Relationship • Calculating Molar Mass from Density:

Calculating Molar Mass of a Gas at STP • The density of a gaseous compound containing carbon and oxygen is found to be 1. 964 g/L at STP. What is the molar mass of the compound? • Density = 1. 964 g/L • 1 mol (gas at STP) = 22. 4 L Molar mass = 1. 964 g X 22. 4 L 1 L 1 mol = 44. 0 g/mol

The Mole Road Map

The Mole Road Map

The Mole Road Map

The Mole Road Map

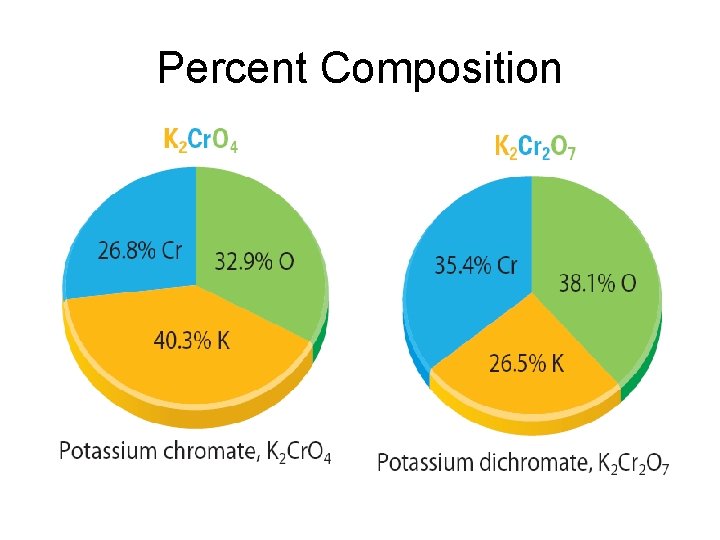
The Percent Composition of a Compound • Percent composition- percent by mass of each element in the compound • Percent by mass of an element in a compound is the number of grams of the element divided by the mass in grams of the compound, multiplied by 100%

Percent Composition
Calculating Percent Composition from Mass Data • When a 13. 60 -g sample of a compound containing only magnesium and oxygen is decomposed, 5. 40 g of oxygen is obtained. What is the percent composition of this compound? • Mass = 13. 60 g • Mass of oxygen = 5. 40 g • Mass of magnesium = 13. 60 g – 5. 40 g = 8. 20 g Mg
Calculating Percent Composition from Mass Data • Example Continued: • % Mg = 8. 20 g X 100% = 60. 3% 13. 60 g • % O = 5. 40 g X 100% = 39. 7% 13. 60 g • 60. 3% + 39. 7% = 100%

The Percent Composition of a Compound • Percent Composition from the Chemical Formula

Calculating Percent Composition from a Formula • Propane (C 3 H 8), the fuel commonly used in gas grills, is one of the compounds obtained from petroleum. Calculate the percent composition of propane. • Mass of C in 1 mol C 3 H 8 = 36. 0 g • Mass of H in 1 mole C 3 H 8 = 8. 0 g • Molar mass of C 3 H 8 = 44. 0 g/mol

Calculating Percent Composition from a Formula • Example Continued: • % C = 36. 0 g X 100% = 81. 8% 44. 0 g • % H = 8. 0 g X 100% = 18% 44. 0 g Add up to 100% when expressed as 2 significant figures

The Percent Composition of a Compound • Percent Composition as a Conversion Factor –You can use percent composition to calculate the number of grams of any element in a specific mass of a compound.

The Percent Composition of a Compound • Propane (C 3 H 8) is 81. 8% carbon and 18% hydrogen. You can calculate the mass of carbon and the mass of hydrogen in an 82. 0 g sample of C 3 H 8.

Empirical Formulas • Empirical formula- gives the lowest whole -number ratio of the atoms of the elements in a compound –The empirical formula of a compound shows the smallest whole-number ratio of the atoms in the compound

Determining Empirical Formula • A compound is analyzed and found to contain 25. 9% nitrogen and 74. 1% oxygen. What is the empirical formula of the compound? • Percent of nitrogen = 25. 9% N • Percent of oxygen = 74. 1% O

Determining Empirical Formula • Since percent means parts per 100, you can assume that 100. 0 g of the compound contains 25. 9 g N and 74. 1 g O. • • 25. 9 g N X 1 mol N = 1. 85 mol N 14. 0 g N 74. 1 g O X 1 mol O = 4. 63 mol O 16. 0 g O

Determining Empirical Formula • Divide each molar quantity by the smaller number of moles: 1. 85 mol N = 1 mol N 1. 85 4. 63 mol O = 2. 50 mol O 1. 85

Determining Empirical Formula • Can’t have fraction : N 1 O 2. 5 • Obtain lowest whole-number ratio by multiplying each part of the ratio by the smallest whole number that will convert both subscripts into whole numbers 1 mol N X 2 = 2 mol N 2. 5 mol O X 2 = 5 mol O Empirical Formula = N 2 O 5

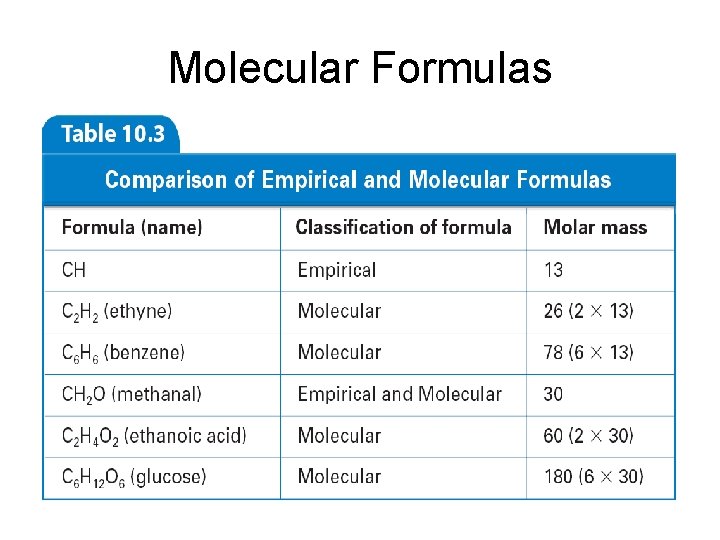
Empirical Formulas • Ethyne (C 2 H 2) is a gas used in welder’s torches. Styrene (C 8 H 8) is used in making polystyrene • These two compounds of carbon have the same empirical formula (CH) but different molecular formulas

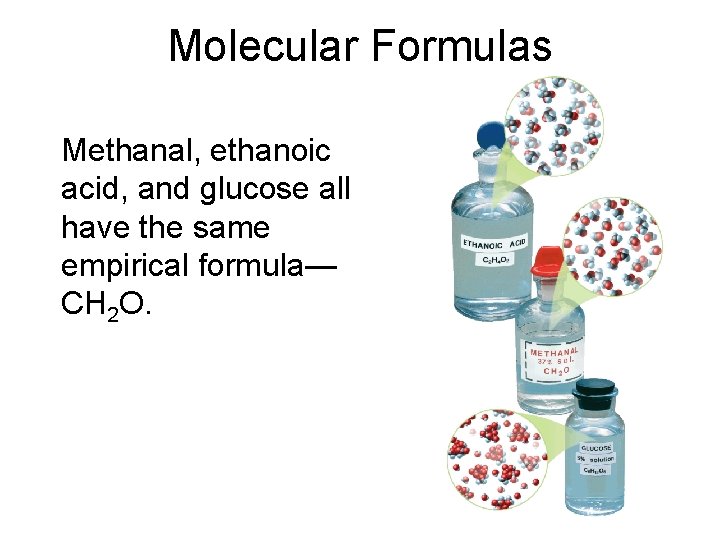
Molecular Formulas • Molecular formula of a compound is either the same as its experimentally determined empirical formula, or it is a simple wholenumber multiple of its empirical formula - must know compound’s molar mass to determine molecular formulas

Molecular Formulas Methanal, ethanoic acid, and glucose all have the same empirical formula— CH 2 O.

Molecular Formulas

Finding Molecular Formula • Calculate the molecular formula of a compound whose molar mass is 60. 0 g/mol and empirical formula is CH 4 N. • Empirical formula = CH 4 N • Molar mass = 60. 0 g/mol • Empirical formula mass = 30. 0 g/mol • 60. 0/30. 0 = 2 (multiply empirical by 2) • Molecular formula = C 2 H 8 N 2

Chemical Reactions

Writing Chemical Equations • Word Equations –To write a word equation: • write the names of the reactants to the left of the arrow separated by plus signs • write the names of the products to the right of the arrow, also separated by plus signs • Reactant + Reactant Product + Product

Writing Chemical Equations • Methane + Oxygen Carbon dioxide + Water

Writing Chemical Equations • iron + oxygen iron(III) oxide

Writing Chemical Equations • Hydrogen Peroxide Water and Oxygen

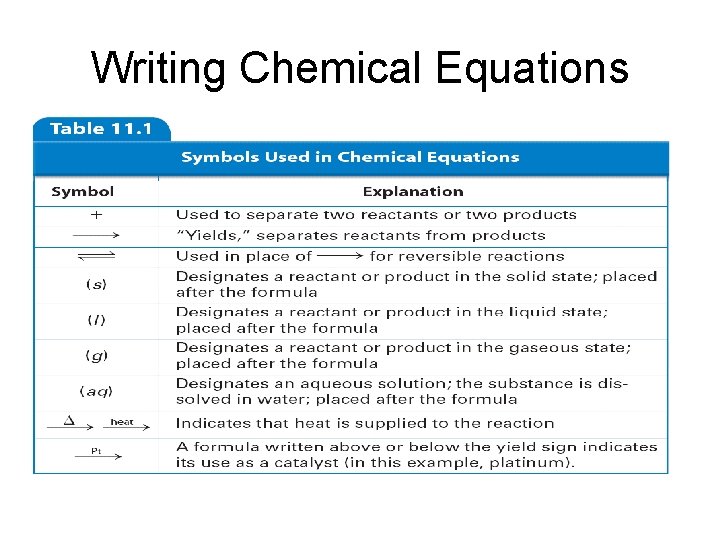
Writing Chemical Equations • Chemical Equations – Chemical equation- is a representation of a chemical reaction • the formulas of the reactants (on the left) are connected by an arrow with the formulas of the products (on the right)

Writing Chemical Equations • Skeleton equation- a chemical equation that does not indicate the relative amounts of the reactants and products • Here is the equation for rusting: Fe + O 2 Fe 2 O 3

Writing Chemical Equations • You can indicate the physical states of substances by using symbols in a chemical reaction: - use (s) for a solid - use (l) for a liquid - use (g) for a gas - use (aq) for aqueous or a substance dissolved in water Fe(s) + O 2(g) Fe 2 O 3(s)

Writing Chemical Equations

Writing Chemical Equations • Catalyst- is a substance that speeds up the reaction but is not used up in the reaction • Without Catalyst With Catalyst

Writing Chemical Equations • A catalyst is neither a reactant or product - its formula is written above the arrow in a chemical equation Example: H 2 O 2(aq) Mn. O 2 H 2 O(l) + O 2(g)

Writing a Skeleton Equation • Hydrochloric acid and solid sodium hydrogen carbonate are reacted together. The products formed are aqueous sodium chloride, water, and carbon dioxide gas. Write a skeleton equation for this reaction. • Solid sodium hydrogen carbonate: Na. HCO 3(s) • Hydrochloric acid: HCl(aq) • Water: H 2 O(l) • Carbon dioxide gas: CO 2(g)

Writing a Skeleton Equation • Example Continued: Na. HCO 3(s) + HCl(aq) Na. Cl(aq) + H 2 O(l) + CO 2(g)

Balancing Chemical Equations • This is a balanced equation for making a bicycle: • Coefficients—small whole numbers that are placed in front of the formulas in an equation in order to balance it

Balancing Chemical Equations • A chemical reaction is also described by a balanced equation • Balanced equation- each side of the equation has the same number of atoms of each element and mass is conserved

Balancing Chemical Equations • To write a balanced chemical equation: - write the skeleton equation - use coefficients to balance the equation so that it obeys the law of conservation of mass

Writing a Balanced Chemical Equation • Hydrogen and oxygen react to form water. The reaction releases enough energy to launch a rocket. Write a balanced equation for the reaction. • Determine the correct formulas for all of the reactants and products – Hydrogen = H 2 – Oxygen = O 2 – Water = H 2 O

Writing a Balanced Chemical Equation • Write the skeleton equation: H 2(g) + O 2(g) H 2 O(l) • Determine the number of atoms of each element between the reactants and products: - reactants = 2 hydrogen, 2 oxygen - products = 2 hydrogen, 1 oxygen

Writing a Balanced Chemical Equation • Balance the elements one at a time • If no coefficient is written, it is assumed to be 1 H 2(g) + O 2(g) 2 H 2 O(l) 2 H 2(g) + O 2(g) 2 H 2 O(l)

Writing a Balanced Chemical Equation • Practice Problem: • Balance the following: Cu(s) + Ag. NO 3(aq) Cu(NO 3)2(aq) + Ag(s) Answer: Cu(s) + 2 Ag. NO 3(aq) Cu(NO 3)2(aq) + 2 Ag(s)

Classifying Reactions • The five general types of reaction are: -combination -decomposition -single-replacement -double-replacement -combustion

Classifying Reactions • Combination reaction- is a chemical change in which two or more substances react to form a single new substance • Example: 2 Mg(s) + O 2(g) 2 Mg. O(s)

Writing Combination Reactions • Copper and sulfur are reactants in a combination reaction. Complete the equation for the reaction: Cu(s) + S(s) ? Cu(s) + S(s) Cu. S(s)

Classifying Reactions • Decomposition reaction- is a chemical change in which a single compound breaks down into two or more simpler products

Decomposition Reaction • Example: 2 Hg. O(s) 2 Hg(l) + O 2(g) Practice: Decomposition of water using electrolysis H 2 O(l) electricity ? electricity H 2 O(l) H 2(g) + O 2(g) electricity 2 H 2 O(l) 2 H 2(g) + O 2(g) (balanced)

Classifying Reactions • Single-replacement reaction- is a chemical change in which one element replaces a second element in a compound

Single-Replacement Reaction • Example: 2 K(s) + 2 H 2 O(l) 2 KOH(aq) + H 2(g) Practice: Write a balanced equation for the following: Zn(s) + H 2 SO 4(aq) ? Zn. SO 4(aq) + H 2(g)

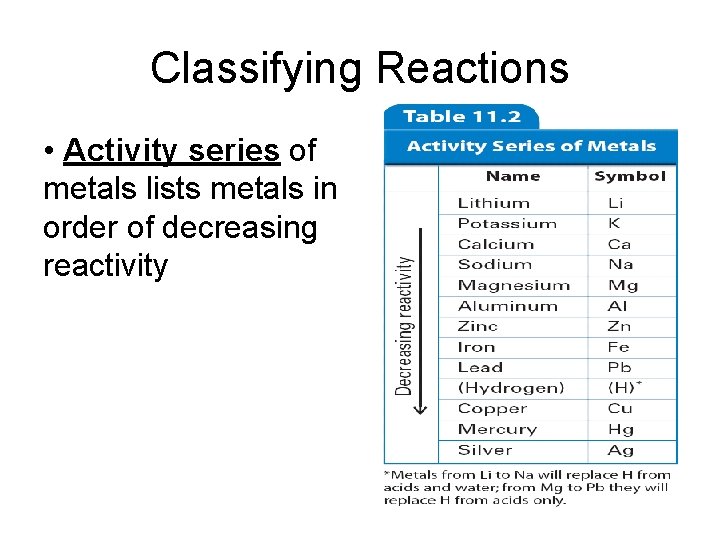
Classifying Reactions • Activity series of metals lists metals in order of decreasing reactivity

Classifying Reactions • Double-replacement reaction- is a chemical change involving an exchange of positive ions between two compounds

Double-replacement reaction • Example: Na 2 S(aq) + Cd(NO 3)2(aq) Cd. S(s) + 2 Na. NO 3(aq) Practice: Write the equation for the following double-replacement reaction: • Ca. Br 2(aq) + Ag. NO 3(aq) ? • Ca. Br 2(aq) + Ag. NO 3(aq) Ag. Br(s) + Ca(NO 3)2(aq) • Ca. Br 2(aq) + 2 Ag. NO 3(aq) 2 Ag. Br(s) + Ca(NO 3)2(aq)

Classifying Reactions • Combustion reaction- is a chemical change in which an element or a compound reacts with oxygen, often producing energy in the form of heat and light

Combustion Reactions • Example: Combustion of gasoline 2 C 8 H 18(l) + 25 O 2(g) 16 CO 2(g) + 18 H 2 O(l) Practice: write the combustion of benzene C 6 H 6(l) • C 6 H 6(l) + O 2(g) • 2 C 6 H 6(l) + 15 O 2(g) CO 2(g) + H 2 O(g) 12 CO 2(g) + 6 H 2 O(g)

Predicting the Products of a Chemical Reaction • The number of elements and/or compounds reacting is a good indicator of possible reaction type and thus possible products

Net Ionic Equations • Complete ionic equation is an equation that shows dissolved ionic compounds as dissociated free ions

Net Ionic Equations • Spectator ion - ion that appears on both sides of an equation and is not directly involved in the reaction • Net ionic equation- is an equation for a reaction in solution that shows only those particles that are directly involved in the chemical change - balanced with respect to both mass and charge

Net Ionic Equations • Sodium ions and nitrate ions are not changed during the chemical reaction of silver nitrate and sodium chloride so the net ionic equation is:

Writing Net Ionic Equations • Aqueous solutions of iron(III) chloride and potassium hydroxide are mixed. A precipitate of iron(III) hydroxide forms. Identify the spectator ions and write a balanced net ionic equation. Fe 3+(aq) + 3 Cl-(aq) + 3 K+(aq) + 3 OH-(aq) Fe(OH)3(s) + 3 K+(aq) + 3 Cl-(aq) Spectators = K+ and Cl • Fe 3+(aq) + 3 OH-(aq) Fe(OH)3(s)

Predicting the Formation of a Precipitate • You can predict the formation of a precipitate by using the general rules for solubility of ionic compounds