Bell Ringer 298 K A sample of nitrogen

Bell Ringer 298 K A sample of nitrogen occupies 10. 0 liters at 25ºC and 98. 7 k. Pa. What would be the volume at 20ºC and 102. 7 k. Pa? 293 K T 2 P 1 V 1 P 2 T 1 A 7. 87 L D 10. 6 L T 2 P 2 P 1 V 1 T 2 T 1 P 2 (98. 7 k. Pa) (10. 0 L) (293 K) V 2 = B 9. 45 L C 10. 2 L = P 2 V 2 = (298 K) (102. 7 k. Pa)

Gas Laws Quiz

For Next Class: • Homework: – Gas Laws Packet #2, problems 1 -10 • Quiz next class on Ideal Gas Law, Partial Pressures, and Density – 5 questions; 22 points total • 2 short answer/FITB (2 points each) • 3 math problems (6 points each)

The Ideal Gas Law & Co. Ms. Besal 3/14/2006

A Reminder… We that we live in an world where: • Gas particles have no mass • Gas particles have no volume • Gas particles have elastic collisions These assumptions are used when trying to calculate the AMOUNT of a gas we have!
Why are these assumptions important? PV = n. RT Image source: thefreedictionary. com

PV = n. RT P RESSURE V OLUME n MOLES OF GAS R GAS CONSTANT TEMPERATURE Image source: popartuk. com

The Myste. Rious R • R is a constant (doesn’t change). • Number value of R depends on other units. • Units of R are a combination of many units. 62. 4 mm. Hg · L mol · K 8. 31 k. Pa · L mol · K 0. 0821 atm · L mol · K Image source: toysrusemail. com
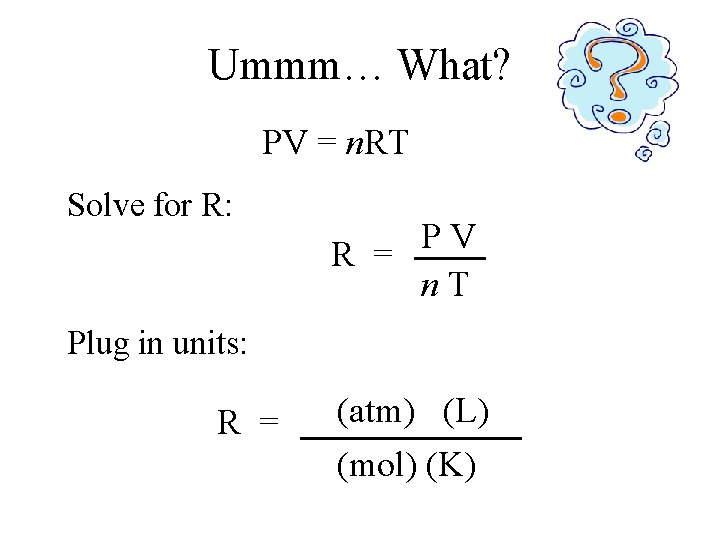
Ummm… What? PV = n. RT Solve for R: P V R = n. T Plug in units: (k. Pa) (atm) Hg) (L) R = (mm (mol) (K)

Gas Laws, Gas Laws Everywhere! V 1 T 1 = V 2 T 2 P 1 x V 1 = P 2 x V 2 P 1 V 1 P 2 V 2 = T 1 T 2 P VCONDITIONS =n. RT Used with CHANGING Used with only ONE SET OF CONDITIONS

When to Use PV = n. RT • Calculating amount of gas in moles • Calculating P, V, or T if moles of gas are known. – IMPORTANT! We must have 3 out of 4 pieces of information: • P • V • n • T

Practice with the Ideal Gas Law 1. A gas sample occupies 2. 62 L at 285ºC and 3. 42 atm. How many moles are present in this sample? PV = n. RT P V n = RT n = (3. 42 atm) (2. 62 L) = 0. 196 mol 0. 0821 L · atm (558 K) mol · K

But Let’s Be Practical… We don’t usually measure in moles! We usually measure quantities in GRAMS! PV = n. RT PVM = g. RT

PVM = g. RT P RESSURE V OLUME M OLAR MASS OF GAS (g/mol) g RAMS OF GAS R GAS CONSTANT TEMPERATURE Image source: popartuk. com

Practice with the Ideal Gas Law A balloon is filled with 0. 2494 g of helium to a pressure of 1. 26 atm. If the desired volume of the balloon is 1. 250 L, what must the temperature be in ºC? PVM = g. RT T = PVM g. R (1. 26 atm) (1. 250 L) 4. 00 g mol T = 0. 0821 L · atm mol · K (0. 2494 g) = 308 K - 273 35 ºC

PV=n. RT vs. PVM=g. RT • Use PV=n. RT when: – You are given moles in the problem. – You are searching for moles as an answer. • Use PVM=g. RT when: – You are given grams in the problem. – You are searching for grams as an answer.

What Else Happens Under Unchanging Conditions? At constant V and T, pressure is easy to calculate! “The sum of the individual pressures is equal to the total pressure. ” Total Pressure = Pressure of gas 1 + Pressure of gas 2 + Pressure of gas 3 + Pressure of gas 4 … Ptotal = P 1 + P 2 + P 3 + …

Partial Pressures Practice A sample of hydrogen gas is collected over water at 25ºC. The vapor pressure of water at 25ºC is 23. 8 mm. Hg. If the total pressure is 523. 8 mm. Hg, what is the partial pressure of the hydrogen? Ptotal = PH 2 + PH 2 O 523. 8 mm Hg PH 2 = = PH + 23. 8 mm Hg 2 500. 0 mm Hg Source: 2003 EOC Chemistry Exam

What do Changing Conditions Affect? We have learned that we can change 3 variables: Temperature, Volume, and Pressure. If MASS remains constant… …But VOLUME changes… Then DENSITY CHANGES! D = M V

Two Types of Density Problems: At STP: Not at STP: • molar volume of any gas at STP = • Determine new volume (V 2 ) using Combined Gas Law P 1 V 1 P 2 V 2 = T 1 T 2 • Density at STP = molar mass molar volume 22. 4 Liters • Density at non-STP = molar mass V 2

Practice with Density Problems: Determine the density of ethane (C 2 H 6) at STP: D (at STP) = Determine the density of C 2 H 6 at 3. 0 atm and 41ºC. molar mass V 2 P 1=V 8. 6 1 L P 2 V 2 molar volume D =V molar mass = P 1 V 1 T 2 2 V 2 T P 1 2 molar mass = 30. 08 g molar volume = 22. 4 L 30. 08 g D = = 1. 34 g/L 22. 4 L T 1 = T 2 P 1 = 1. 0 atm P 2 = 3. 0 atm V 2 D = (1. 0 atm) (22. 4 (314 K) = 30. 08 g =L) 3. 5 g/L V 1 = 22. 48. 6 L L V 2 = ? (273 K) (3. 0 atm) T 1 = 273 K V 2 = 8. 6 L T 2 = 314 K

For Next Class: • Homework: – Gas Laws Packet #2, problems 1 -10 • Quiz next class on Ideal Gas Law, Partial Pressures, and Density – 5 questions; 22 points total • 2 short answer/FITB (2 points each) • 3 math problems (6 points each)

What do Changing Conditions Affect? • Density • Stoichiometry problems So Far: Now…

Mass-Volume at Non-STP Two parts to solving these problems: • Use Stoichiometry • Use Gas Law
- Slides: 24