Enzymes Biochemistry Part II Enzymes are very important

Enzymes Biochemistry Part II

Enzymes are very important for chemical reactions. There are two general parts for every reaction: Reactants- the chemicals that participate in a chemical reaction Products- the substances that are formed as a result of a chemical reaction C 6 H 12 O 6 + O 2 CO 2 + H 2 O reactants products
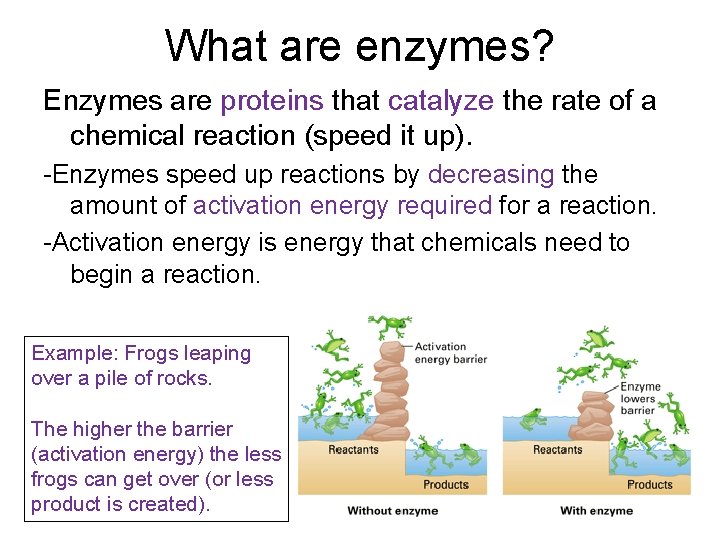
What are enzymes? Enzymes are proteins that catalyze the rate of a chemical reaction (speed it up). -Enzymes speed up reactions by decreasing the amount of activation energy required for a reaction. -Activation energy is energy that chemicals need to begin a reaction. Example: Frogs leaping over a pile of rocks. The higher the barrier (activation energy) the less frogs can get over (or less product is created).
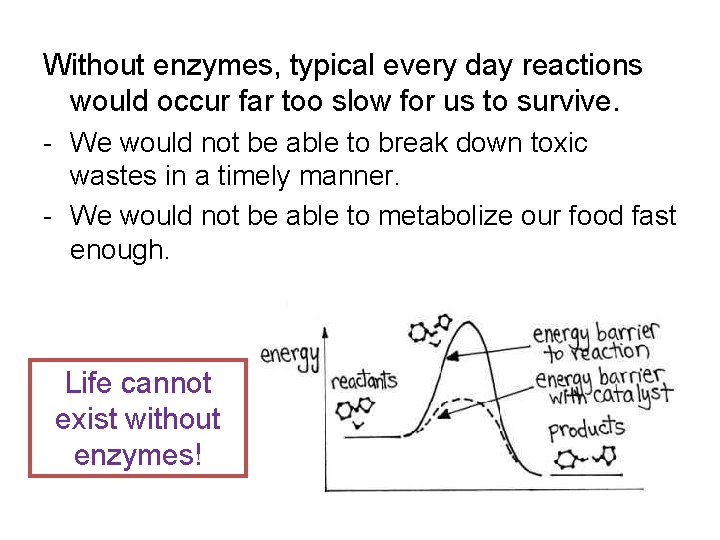
Without enzymes, typical every day reactions would occur far too slow for us to survive. - We would not be able to break down toxic wastes in a timely manner. - We would not be able to metabolize our food fast enough. Life cannot exist without enzymes!

Examples of Enzymes § Carbonic anhydrase: found in blood; speeds up the removal of carbon dioxide from our bodies. § Catalase: found in liver tissue; speeds up the decomposition of hydrogen peroxide in our bodies § Amylase; found in saliva; speeds up the breakdown of sugar for digestion. Enzyme Substrate (the Reactant) Anhydrase Carbon dioxide Catalase Hydrogen Peroxide Amylase Maltrose
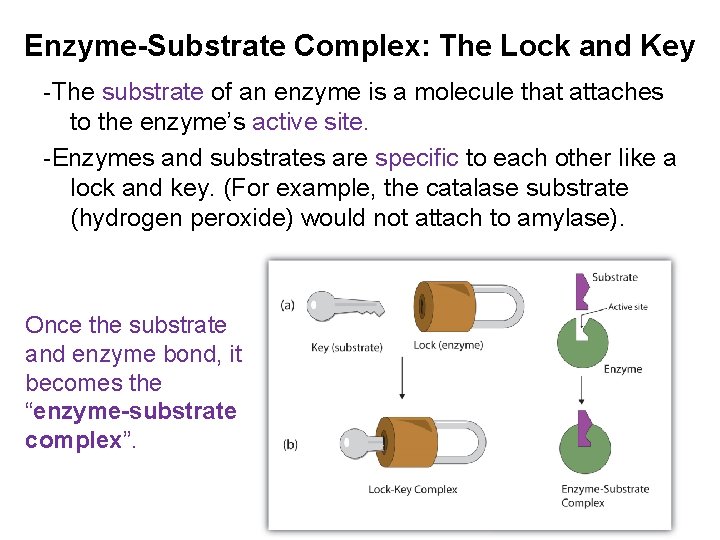
Enzyme-Substrate Complex: The Lock and Key -The substrate of an enzyme is a molecule that attaches to the enzyme’s active site. -Enzymes and substrates are specific to each other like a lock and key. (For example, the catalase substrate (hydrogen peroxide) would not attach to amylase). Once the substrate and enzyme bond, it becomes the “enzyme-substrate complex”.

Enzyme-Substrate Complex Once the enzyme-substrate complex is formed, the enzyme changes its shape slightly to create a more secure bond, and then the chemical reaction follows. The active site is simply where the substrate bonds to the enzyme.
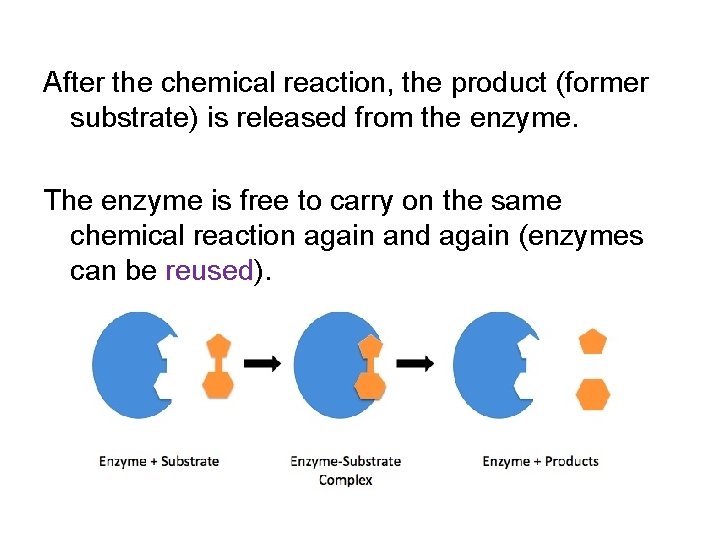
After the chemical reaction, the product (former substrate) is released from the enzyme. The enzyme is free to carry on the same chemical reaction again and again (enzymes can be reused).

Factors that Affect Enzymes -Two factors that affect enzyme productivity are p. H and temperature. -Enzymes thrive best within certain temperature and p. H ranges. -If the environment is altered, the enzyme activity may decrease. Example) people die of fevers not because of the high temperatures, but because the enzymes in their body do not function properly once the temperature gets too high.
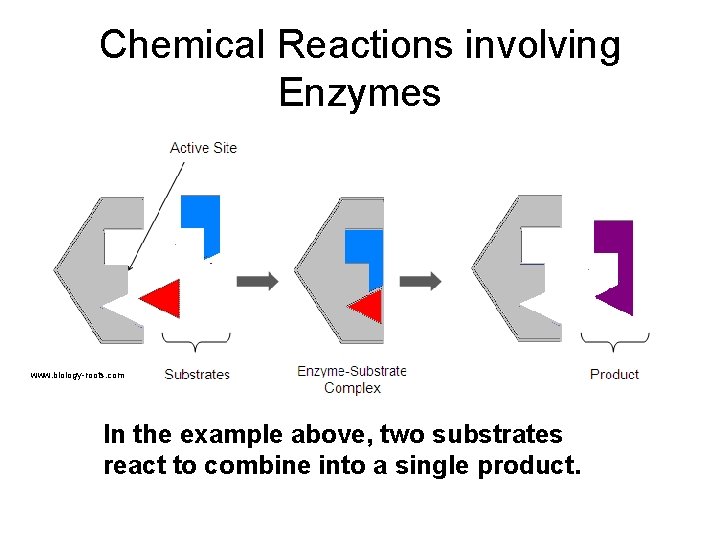
Chemical Reactions involving Enzymes www. biology-roots. com In the example above, two substrates react to combine into a single product.
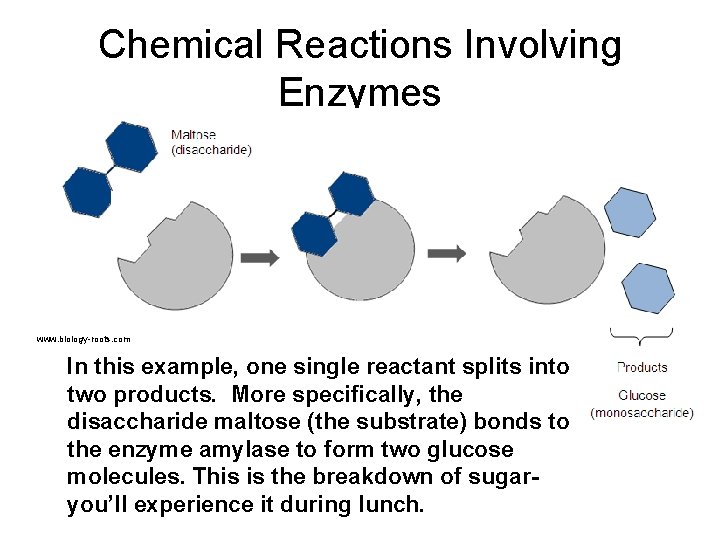
Chemical Reactions Involving Enzymes www. biology-roots. com In this example, one single reactant splits into two products. More specifically, the disaccharide maltose (the substrate) bonds to the enzyme amylase to form two glucose molecules. This is the breakdown of sugaryou’ll experience it during lunch.

Enzymes
- Slides: 12