Indicators of chemical reactions Emission of light or














- Slides: 31


Indicators of chemical reactions Emission of light or heat Formation of a gas Formation of a precipitate Color change Emission of odor

All chemical reactions: Two parts: Reactants – starting substances Products- ending substances The reactants turn into the products

Describing chemical reaction Combination of atoms changes Atoms are neither created nor destroyed. Can be described several ways Copper reacts with chlorine to form copper (II) chloride. Copper + chlorine copper (II) chloride Cu(s) + Cl 2(g) Cu. Cl 2(aq)

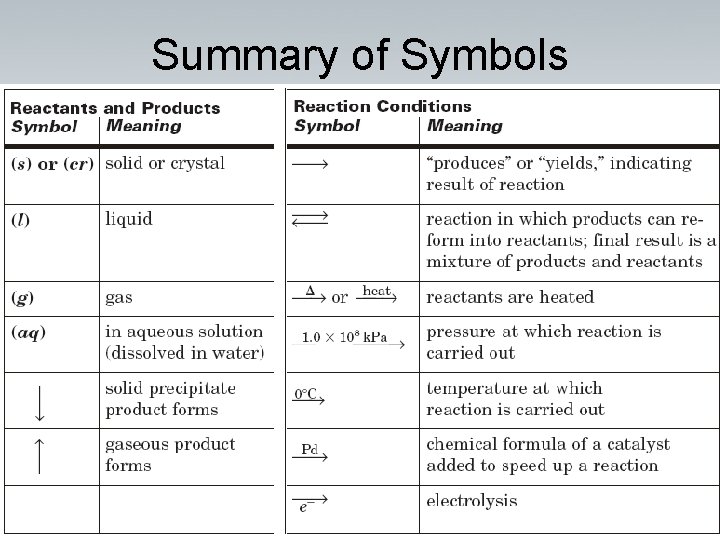
Symbols used in equations (s) after the formula – solid Cu(s) (g) after the formula – gas H 2 (g) (l) after the formula – liquid H 2 O(l) (aq) after the formula – dissolved in water, an aqueous solution. Ca. Cl 2 (aq)

Summary of Symbols

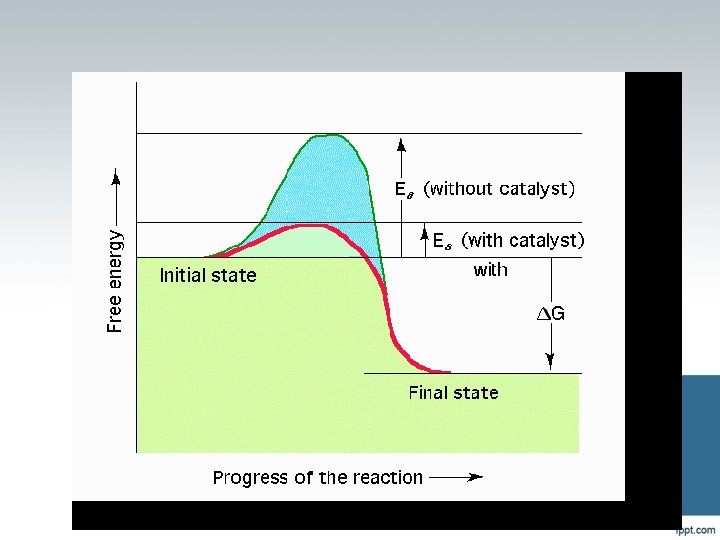
What is a catalyst? A substance that speeds up a reaction without being changed by the reaction. Enzymes are biological or protein catalysts.

Reaction Energy All chemical reactions are accompanied by a change in energy. Exothermic - reactions that release energy to their surroundings (usually in the form of heat) ΔH (enthalpy) is negative – energy leaving system Endothermic - reactions that need to absorb heat from their surroundings to proceed. ΔH (enthalpy) is positive – energy coming into the system

Reaction Energy Spontaneous Reactions - Reactions that proceed immediately when two substances are mixed together. Not all reactions proceed spontaneously. Activation Energy – the amount of energy that is required to start a chemical reaction. Once activation energy is reached the reaction continues until you run out of material to react.


Formula Equation Use formulas and symbols to describe a reaction. Doesn’t indicate how many All chemical equations are sentences that describe reaction.

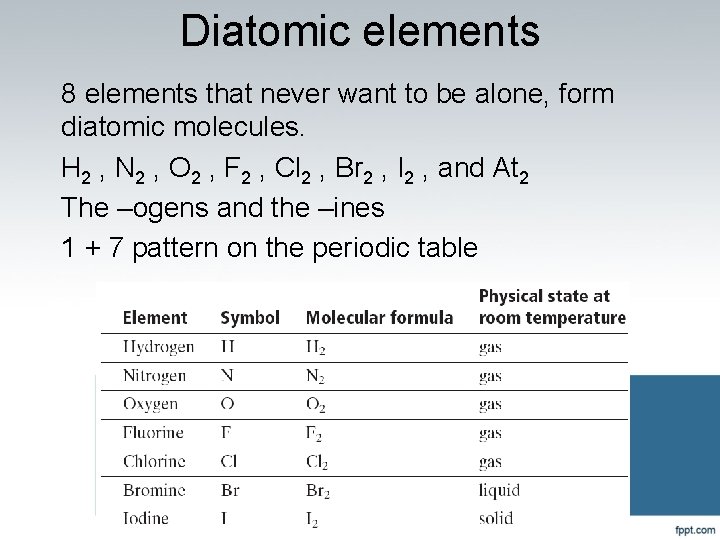
Diatomic elements 8 elements that never want to be alone, form diatomic molecules. H 2 , N 2 , O 2 , F 2 , Cl 2 , Br 2 , I 2 , and At 2 The –ogens and the –ines 1 + 7 pattern on the periodic table


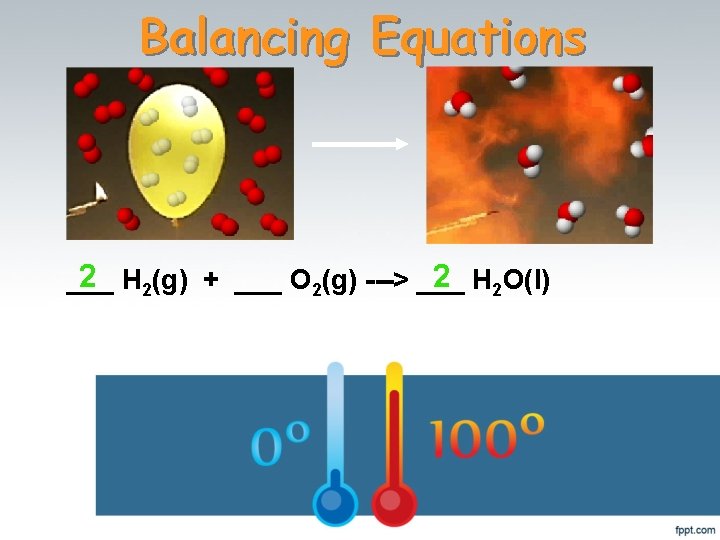
Balancing Equations 2 H 2(g) + ___ O 2(g) ---> ___ 2 H 2 O(l) ___

What Happened to the Other Oxygen Atom? This equation is not balanced! Two hydrogen atoms from a hydrogen molecule (H 2) combines with one of the oxygen atoms from an oxygen molecule (O 2) to form H 2 O. Then, the remaining oxygen atom combines with two more hydrogen atoms (from another H 2 molecule) to make a second H 2 O molecule. __2_ H 2(g) + ___ O 2(g) ---> __2_ H 2 O(l)

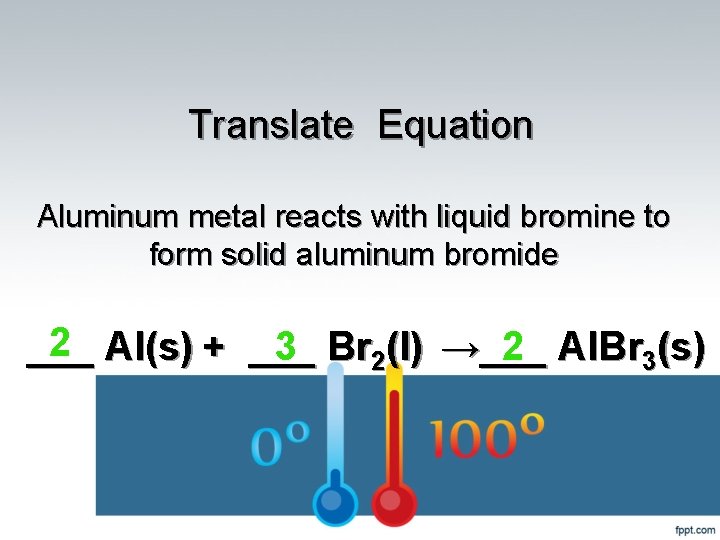
Translate Equation Aluminum metal reacts with liquid bromine to form solid aluminum bromide 2 Al(s) + ___ 2 Al. Br 3(s) ___ 3 Br 2(l) →___

Types of Reactions There are millions of reactions. Fall into several categories. Focus on 6 types. Be able to predict the products. Be able to predict if they will happen at all.

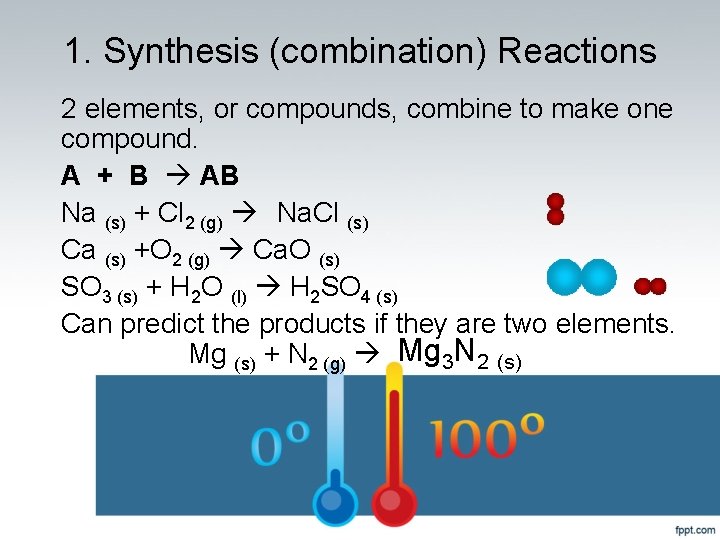
1. Synthesis (combination) Reactions 2 elements, or compounds, combine to make one compound. A + B AB Na (s) + Cl 2 (g) Na. Cl (s) Ca (s) +O 2 (g) Ca. O (s) SO 3 (s) + H 2 O (l) H 2 SO 4 (s) Can predict the products if they are two elements. Mg (s) + N 2 (g) Mg 3 N 2 (s)

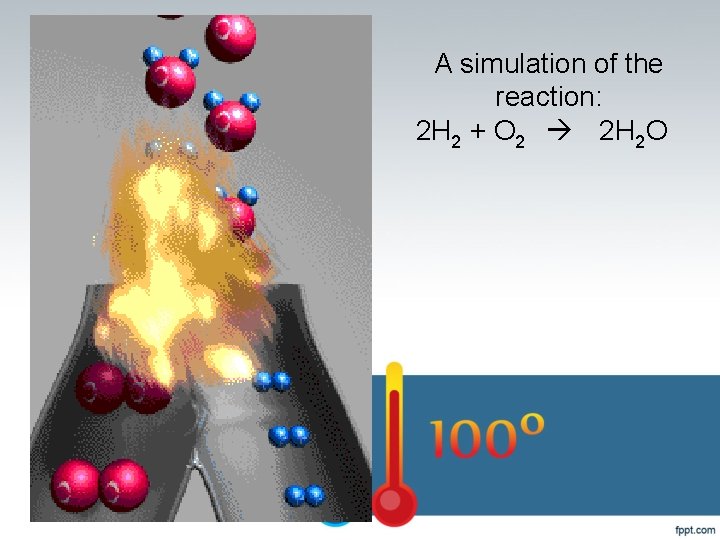
A simulation of the reaction: 2 H 2 + O 2 2 H 2 O

2. Decomposition Reactions decompose = fall apart one compound (reactant) falls apart into two or more elements or compounds. Usually requires energy AB A + B 2 Na. Cl 2 Na + Cl 2 Ca. CO 3 Ca. O + CO 2

Decomposition Reactions Can predict the products if it is a binary compound Made up of only two elements Falls apart into its elements H 2 O H 2 (g) + O 2 (g) 2 Hg. O 2 Hg(s) + O 2 (g)

Decomposition Reactions If the compound has more than two elements you must be given one of the products The other product will be from the missing pieces Ni. CO 3 (aq) CO 2 (g) + 2 Ni (s) H 2 CO 3(aq) H 2 (g) + CO 2 (g)

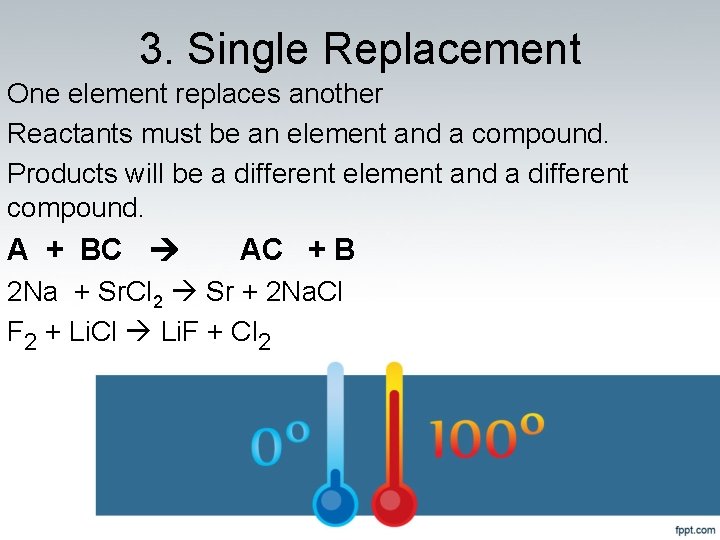
3. Single Replacement One element replaces another Reactants must be an element and a compound. Products will be a different element and a different compound. A + BC AC + B 2 Na + Sr. Cl 2 Sr + 2 Na. Cl F 2 + Li. Cl Li. F + Cl 2

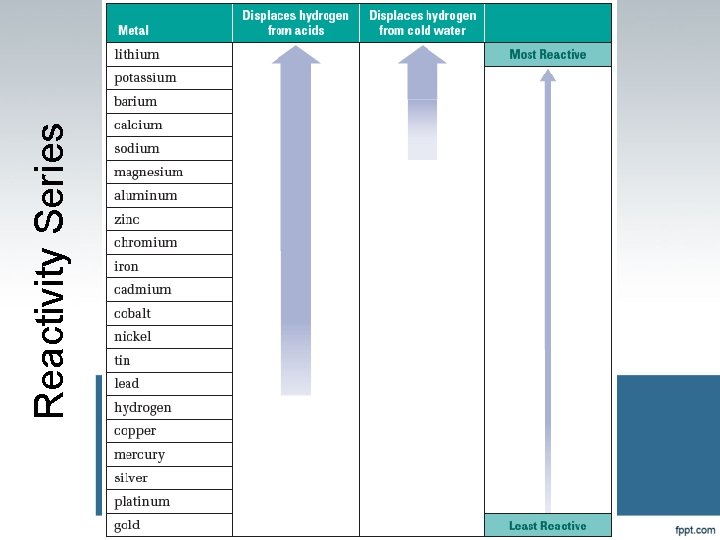
Single Replacement We can tell whether a reaction will happen Some are more active than others More active replaces less active

Reactivity Series

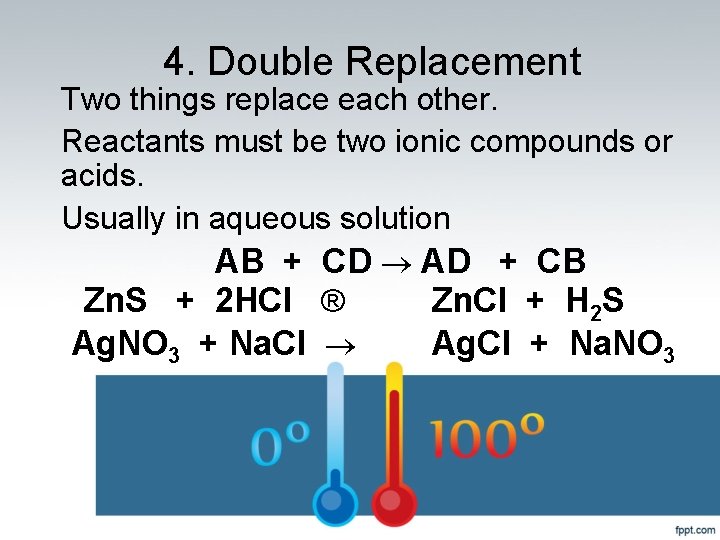
4. Double Replacement Two things replace each other. Reactants must be two ionic compounds or acids. Usually in aqueous solution AB + CD ® AD + CB Zn. S + 2 HCl ® Zn. Cl + H 2 S Ag. NO 3 + Na. Cl ® Ag. Cl + Na. NO 3

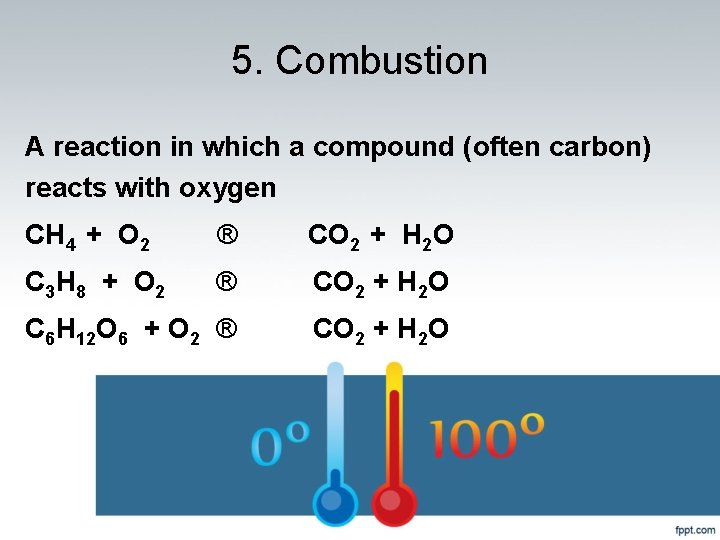
5. Combustion A reaction in which a compound (often carbon) reacts with oxygen CH 4 + O 2 ® CO 2 + H 2 O C 3 H 8 + O 2 ® CO 2 + H 2 O C 6 H 12 O 6 + O 2 ® CO 2 + H 2 O

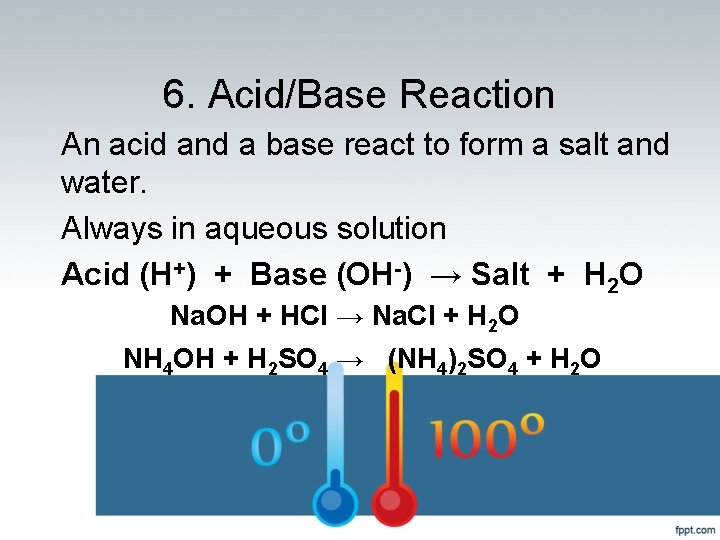
6. Acid/Base Reaction An acid and a base react to form a salt and water. Always in aqueous solution Acid (H+) + Base (OH-) → Salt + H 2 O Na. OH + HCl → Na. Cl + H 2 O NH 4 OH + H 2 SO 4 → (NH 4)2 SO 4 + H 2 O

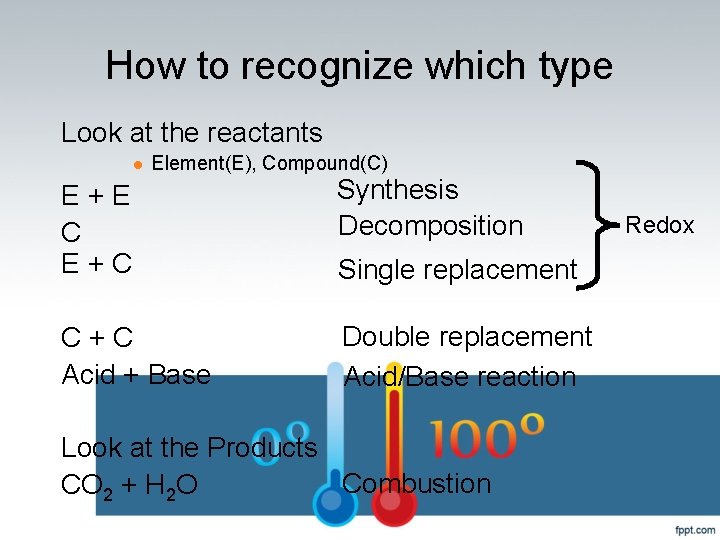
How to recognize which type Look at the reactants l Element(E), Compound(C) E + E C E + C Synthesis Decomposition C + C Acid + Base Double replacement Acid/Base reaction Single replacement Look at the Products Combustion CO 2 + H 2 O Redox

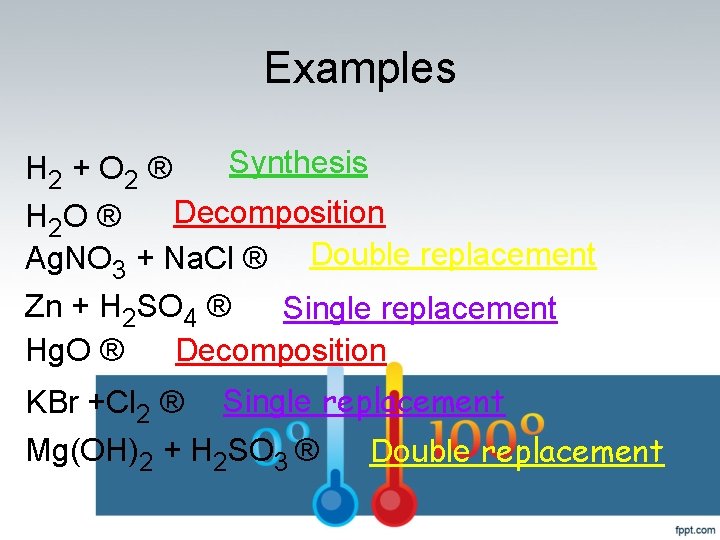
Examples Synthesis H 2 + O 2 ® H 2 O ® Decomposition Ag. NO 3 + Na. Cl ® Double replacement Zn + H 2 SO 4 ® Single replacement Hg. O ® Decomposition KBr +Cl 2 ® Single replacement Mg(OH)2 + H 2 SO 3 ® Double replacement

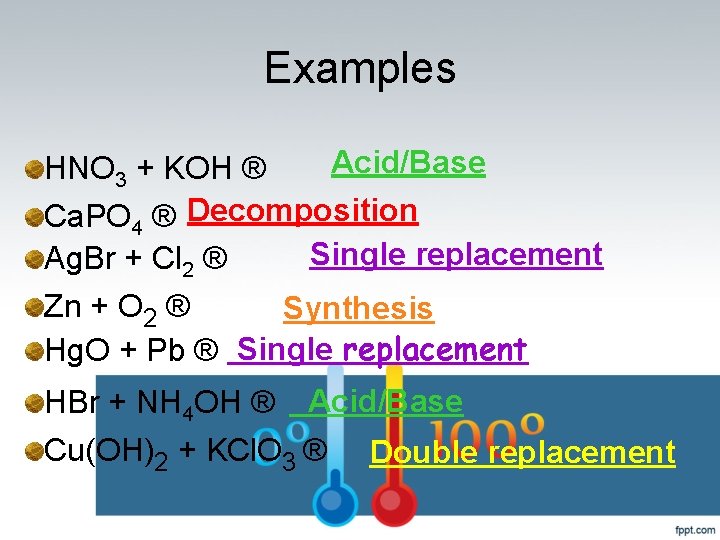
Examples Acid/Base HNO 3 + KOH ® Ca. PO 4 ® Decomposition Single replacement Ag. Br + Cl 2 ® Zn + O 2 ® Synthesis Hg. O + Pb ® Single replacement HBr + NH 4 OH ® Acid/Base Cu(OH)2 + KCl. O 3 ® Double replacement