Chemical Reactions Types of Chemical Reactions 5 common






















- Slides: 24

Chemical Reactions

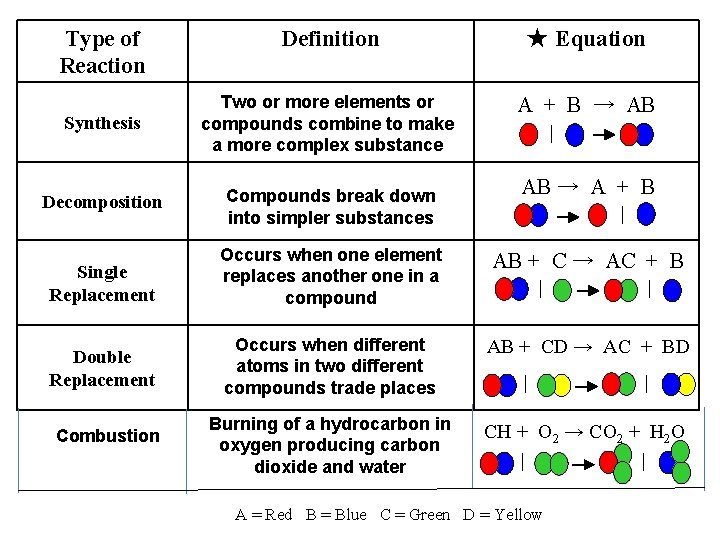
Types of Chemical Reactions 5 common types of chemical reactions 1. Synthesis 2. Decomposition 3. Single replacement 4. Double replacement 5. Combustion


The 5 Signs of a Chemical Reaction There are five main signs that a chemical reaction has taken place: Light released Temperature change Production of a gas Formation of a precipitate Color change

● Are used to represent chemical reactions ● 2 Mg + O 2 Reactants ● ● An 2 Mg. O Products arrow is placed between them to show the direction of reaction.

Synthesis • Means “putting together” • Two or more substances (elements or compounds) combine to form a single new compound General Form: A + B → AB Examples: 2 H 2 + O 2 → 2 H 2 O 2 Na + Cl 2 → 2 Na. Cl


Decomposition • Opposite of synthesis reactions • a single compound reacts to give two or more products General Form: AB → A + B Examples: 2 H 2 O → 2 H 2 + O 2 2 Na. Cl → 2 Na + Cl 2

Hydrogen Peroxide 2 H 2 O 2 —> 2 H 2 O + O 2


Single Replacement Reactions A reaction in which one element replaces or displaces another element in a compound. General Form: A + BC → B + AC A and B switch places. Example: Zn + Cu. SO 4 → Cu + Zn. SO 4

Single Replacement Reactions Use the activity series of metals to determine whether the reaction will work or not.

Double Replacement Reactions A reaction in which two soluble ionic compounds react to form a precipitate (or water). General Form: AC + BD→ AD + BC A and B switch places. Example: Ag. NO 3 + Na. Cl → Na. NO 3 + Ag. Cl (ppt)

Double Replacement Reactions Ag. NO 3(aq) + KCl(aq) ‑‑‑‑> Ag. Cl(s) + KNO 3(aq) Use a solubility table to determine if a precipitate is formed.


Combustion Reactions The burning of a hydrocarbon in the presence of oxygen. Hydrocarbon: organic compound that is composed only of carbon and hydrogen. Examples: gasoline, kerosene, propane

Combustion Reactions General Form: Cx. Hy + O 2 → CO 2 + H 2 O + heat The products are usually carbon dioxide, water, and energy in the form of heat.

Combustion Reactions • This is also called • burning!!! In order to burn something you need the 3 things in the “fire triangle”: 1) A Fuel (hydrocarbon) 2) Oxygen to burn it with 3) Something to ignite the reaction (spark)

Combustion Reactions • In general: Cx. Hy + O 2 → CO 2 + H 2 O • Products in combustion are ALWAYS carbon dioxide and water. • Combustion is used to heat homes and run automobiles (gasoline is octane, C 8 H 18) • (Note: incomplete burning causes some by-products like carbon monoxide = CO)

Type of Reaction Definition ★ Equation Synthesis Two or more elements or compounds combine to make a more complex substance A + B → AB Decomposition Compounds break down into simpler substances Single Replacement Occurs when one element replaces another one in a compound AB + C → AC + B Double Replacement Occurs when different atoms in two different compounds trade places AB + CD → AC + BD Burning of a hydrocarbon in oxygen producing carbon dioxide and water CH + O 2 → CO 2 + H 2 O Combustion AB → A + B A = Red B = Blue C = Green D = Yellow

Identifying Chemical Reactions S = Synthesis D = Decomposition SR = Single Replacement DR = Double Replacement C = Combustion ____ Hg. O → Hg + O 2 ____ Mg + O 2 → Mg. O ____ P + Cl 2 → P 4 Cl 10 ____ Al 2 O 3 → Al + O 2 ____ Cl 2 + Na. Br → Na. Cl + Br 2 ____ H 2 + N 2 → NH 3

Identifying Chemical Reactions ____ Na + Br 2 → Na. Br ____ CH 4 + O 2 → CO 2 + H 2 O ____ Cu. Cl 2 + H 2 S → Cu. S + HCl ____ C + H 2 → CH 4 ____ KCl. O 3 → KCl + O 2 ____ C 8 H 18 + O 2 → CO 2 + H 2 O ____ Ba. Cl 2 + Na 2 SO 4 → Na. Cl + Ba. SO 4 ____ S 8 + F 2 → SF 6

What types of reaction is it? • • • 1) Na. Br + H 3(PO 4) → Na 3(PO 4) + HBr 2) Mg + Fe 2 O 3 → Fe + Mg. O 3) C 2 H 4 + O 2 → CO 2 + H 2 O 4) Pb(SO 4) → Pb(SO 3) + O 2 5) NH 3 + I 2 → N 2 I 6 + H 2 7) H 2 O + SO 3 → H 2(SO 4)
