Types of Redox Reactions Balancing Redox Reactions Mr

-Types of Redox Reactions - Balancing Redox Reactions Mr. Shields Regents Chemistry U 14 L 02 1

Types of Redox Reactions We’ve talked about 5 different types of chemical reactions Before: 1) Synthesis 2) Decomposition 3) Combustion 4) Single Replacement 5) Double Repalcement The first 4 of these reaction types are examples of Redox Reactions. Why isn’t double replacement an example of a Redox Reaction? 2

In double replacement two ions simply replace each other In the compounds undergoing reaction. There is no REDOX Occuring. For example: Ba. Cl 2 + Ag. NO 3 Ag. Cl + Ba(NO 3)2 What’s the oxidation nos. of the reactants and Products In the equation above? Ba+2 Cl-1 Ag+1 N+5 O-2 Has any oxidation or reduction taken place? 3

Synthesis In Synthesis, elements or compounds combine to form One product. Example: N 2 + 3 H 2 2 NH 3 This is a Redox Reaction. What is being Oxidized and What’s being Reduced? N 0 and H 0 change to N-3 and H+1. Nitrogen has been reduced And Hydrogen has been oxidized 4

Decomposition In Decomposition a compound is broken down into simpler Compounds and/or elements. Example: 2 Hg. O 2 Hg + O 2 This too is a Redox reaction. What is being Oxidized & What’s Reduced? Hg+2 and O-2 become Hg 0 and O 0. Hg+2 has been reduced And O-2 has been oxidized. 5

Combustion In Combustion a compound reacts with oxygen to form several compounds, if combustion is complete the products Are always water and carbon dioxide. Example: CH 4 + 2 O 2 2 H 20 + CO 2 + Heat Again, this is a Redox reaction. What’s being Oxidized & What is being Reduced? C-4 and O 0 become C+4 and O-2. Carbon-4 has been oxidized And O 0 has been reduced. 6

Single Replacement In Single Repalcement rxns a free element replaces another Element that is combined in a compound. Example: Zn + Cu(NO 3)2 Zn(NO 3)2 + Cu Since this is a Redox rxn what is being Oxidized & Reduced? Zn 0 and Cu+2 become Zn+2 and Cu 0. Zinc (0) has been Oxidized and Cu (+2) has been reduced. 7

Remember when we’re discussingle replacement rxns We need to consider what their position is relative to one Another on TABLE J Metals higher on the List will replace metals Lower on the list. Most easily oxidized Metals higher on the List are more reactive And lastly, metals Higher on the list are More easily oxidized. Most easily reduced Less easily oxidized Leasteasily reduced Notice that halogens accept electrons so higher on the list 8 means more easily reduced!

Problem: in the following reaction Sr + Fe. CL 2 Sr. Cl 2 + Fe What gets oxidized? Sro What gets reduced? Iron +2 What is the oxidizing agent? Iron +2 What is the reducing agent? Sro Write the Oxidation & Reduction half cell rxns 9

Balancing Redox Eqs We’re now going to return to a topic we’ve discussed Before… BALANCING CEHEMICAL EQUATIONS For example: Fe + O 2 Fe 2 O 3 1) 1 st we count atoms on the reactant side and then on the Product side #R 1 2 atom #P Fe 2 O 3 10

Balancing Redox Eqs 2) We then pick one atom to balance, let’s say oxygen 3 x #R 1 2 atom #P Fe 2 O 3 Then see what we’ve changed: x 2 Fe + 3 O 2 2 Fe 2 O 3 3) After we’ve adjusted our table we then balance the next atom, Fe 4 x #R 1 6 atom #P Fe 4 O 6 balanced 4 Fe + 3 O 2 Fe O 11

Balancing Redox Rxns Many reactions can be balanced “visually” or using the RAP Method we just reviewed. However, many redox reactions are difficult to balance Using these methods. Consider the difficulty of balancing this REDOX equation: S + HNO 3 SO 2 + NO + H 2 O We could balance it with RAP tables but it would be difficult To more easily balance this equation we need to apply A method that will utilize oxidation numbers. 12

Balancing Redox Rxns There are some rules you need to follow to balance Redox reactions. By balancing reactions this way we will not only Balance the equation but we will BALANCE THE HALF CELLS 1) The number of e- lost in an oxidation process must equal the number of electrons gained in the reduction process 2) Assign oxidation numbers to all atoms in the chem. Eq. 3) Identify which reactants are oxidized & which are reduced 0 +1 +5 -2 +4 -2 +2 -2 +1 -2 S + HNO 3 SO 2 + NO + H 2 O oxidized reduced 13

Balancing Redox Rxns 4) Visually balance any atoms that change from ions to atoms or vice versa EX: Cr + Cl 2 Cr. Cl 3 becomes Cr + 3 Cl 2 2 Cr. Cl 3 And NH 3 + O 2 N 2 +H 20 becomes 2 NH 3 + O 2 N 2 + 2 H 2 O 14

Balancing Redox Rxns 5) Connect the atoms that change oxidation number by a line 6) Write the change in electrons on the line Ox. -4 (e-) 0 +1 +5 -2 S + HNO 3 +4 -2 +2 -2 +1 -2 SO 2 + NO + H 2 O Red +3 (e-) Note that these brackets define the 2 half cell reactions S 0 S+4 + 4 e- and N+5 + 3 e- N+2 15
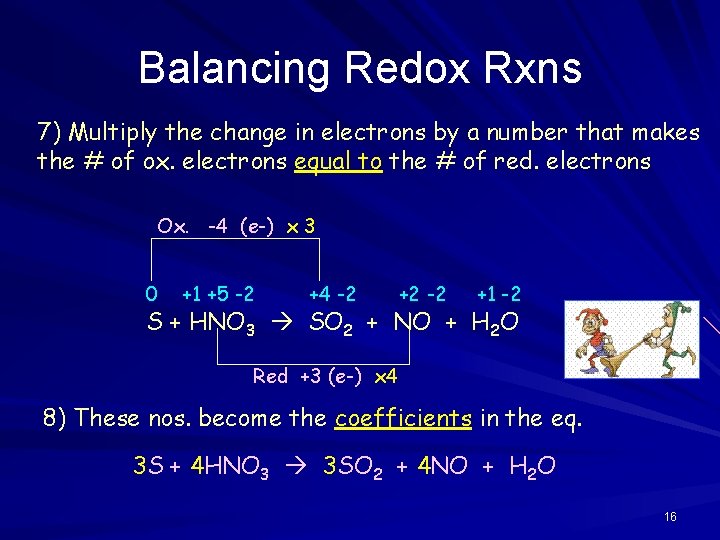
Balancing Redox Rxns 7) Multiply the change in electrons by a number that makes the # of ox. electrons equal to the # of red. electrons Ox. -4 (e-) x 3 0 +1 +5 -2 +4 -2 +2 -2 +1 -2 S + HNO 3 SO 2 + NO + H 2 O Red +3 (e-) x 4 8) These nos. become the coefficients in the eq. 3 S + 4 HNO 3 3 SO 2 + 4 NO + H 2 O 16

Balancing Redox Rxns 8) Balance the rest of the equation by visual inspection or RAP tables 3 S + 4 HNO 3 3 SO 2 + 4 NO + 2 H 2 O Need 4 H on product side Increase H to 4 Is oxygen also balanced? Yes. There are 12 O on each side of the Eq. This equation is now balanced. 17

Problem: Balance the following equation! Ag + HNO 3 Ag. NO 3 + NO + H 20 Solution: 1) Ox -1 e- x 3 0 +1 +5 -2 +2 -2 +1 -2 Ag + HNO 3 Ag. NO 3 + NO + H 20 Red. +3 e- x 1 2) 3 Ag + HNO 3 3 Ag. NO 3 + NO + H 20 Visually or using RAP tables Balance the Rest of the Eq. 3) 3 Ag + 4 HNO 3 3 Ag. NO 3 + NO + 2 H 20 1) I Need 4 N Is “O” OK? 2) SO… I need 4 H 18

Notice that the half cell reactions are now also = balanced Ox -1 e- x 3 0 +1 +5 -2 +2 -2 +1 -2 Ag + HNO 3 Ag. NO 3 + NO + H 20 Red. +3 e- x 1 3 Ag + 4 HNO 3 3 Ag. NO 3 + NO + 2 H 20 Unbalanced half cell reactions: Ag 0 Ag+1 + 1 e. N+5 + 3 e- N+2 Balanced half cell reactions: 3 Ag 0 3 Ag+1 + 3 e 1 N+5 + 3 e- 1 N+2 # of e- lost And gained Are equal 19

Conservation of Mass & Charge Remember !! In ALL chemical reactions there is not only conservation of mass - Equal number of atoms on both sides There must also be conservation of charge - Number of e- lost = Number of e- gained 20
- Slides: 20