Chapter 4 B Balancing Redox Reactions LEO SAYS

Chapter 4 B: Balancing Redox Reactions LEO SAYS GER West Valley High School AP Chemistry Mr. Mata

Oxidation and Reduction (Redox) q Electrons are transferred q Spontaneous redox rxns can transfer energy q Electrons (electricity) q Heat q Non-spontaneous redox rxns can be made to happen with electricity

Oxidation Reduction Reactions (Redox) Each sodium atom loses one electron: Each chlorine atom gains one electron:
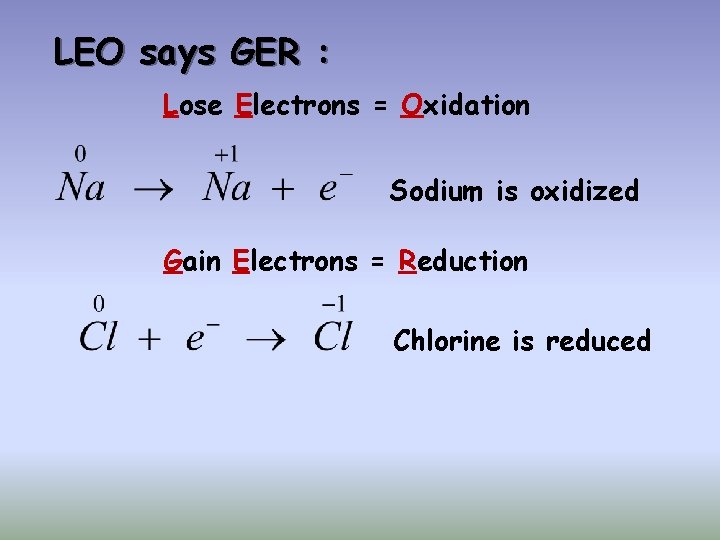
LEO says GER : Lose Electrons = Oxidation Sodium is oxidized Gain Electrons = Reduction Chlorine is reduced

Not All Reactions are Redox Reactions in which there has been no change in oxidation number are not redox rxns. Examples:

Rules for Assigning Oxidation Numbers Rules 1 & 2 1. The oxidation number of any uncombined element is zero 2. The oxidation number of a monatomic ion equals its charge

Rules for Assigning Oxidation Numbers Rules 3 & 4 3. The oxidation number of oxygen in compounds is -2 4. The oxidation number of hydrogen in compounds is +1

Rules for Assigning Oxidation Number Rule 5 5. The sum of the oxidation numbers in the formula of a compound is 0 2(+1) + (-2) = 0 H O (+2) + 2(-2) + 2(+1) = 0 Ca O H

Rules for Assigning Oxidation Numbers Rule 6 6. The sum of the oxidation numbers in the formula of a polyatomic ion is equal to its charge X + 3(-2) = -1 N O X + 4(-2) = -2 S O X = +5 X = +6

The Oxidation Number Rules SIMPLIFIED 1. The sum of the oxidation numbers in ANYTHING is equal to its charge 2. Hydrogen in compounds is +1 3. Oxygen in compounds is -2

Reducing Agents and Oxidizing Agents q The substance reduced is the oxidizing agent q The substance oxidized is the reducing agent Sodium is oxidized – it is the reducing agent Chlorine is reduced – it is the oxidizing agent

Trends in Oxidation and Reduction Active metals: Lose electrons easily Are easily oxidized Are strong reducing agents Active nonmetals: Gain electrons easily Are easily reduced Are strong oxidizing agents
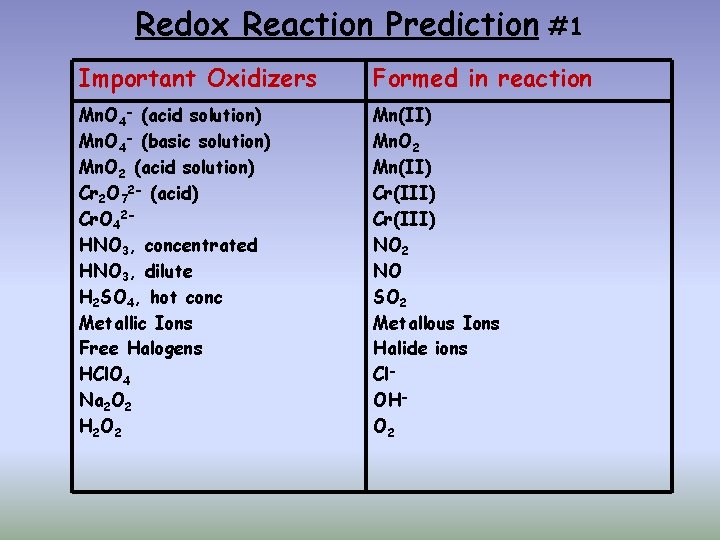
Redox Reaction Prediction #1 Important Oxidizers Formed in reaction Mn. O 4 - (acid solution) Mn. O 4 - (basic solution) Mn. O 2 (acid solution) Cr 2 O 72 - (acid) Cr. O 42 HNO 3, concentrated HNO 3, dilute H 2 SO 4, hot conc Metallic Ions Free Halogens HCl. O 4 Na 2 O 2 H 2 O 2 Mn(II) Mn. O 2 Mn(II) Cr(III) NO 2 NO SO 2 Metallous Ions Halide ions Cl. OHO 2

Redox Reaction Prediction #2 Important Reducers Formed in reaction Halide Ions Free Metals Metalous Ions Nitrite Ions Sulfite Ions Free Halogens (dil, basic sol) Free Halogens (conc, basic sol) C 2 O 42 - Halogens Metal Ions Metallic ions Nitrate Ions SO 42 Hypohalite ions Halate ions CO 2

Oxidation Reduction • Oxidation means an increase in oxidation state - lose electrons. • Reduction means a decrease in oxidation state - gain electrons. • The substance that is oxidized is called the reducing agent. • The substance that is reduced is called the oxidizing agent.

Agents • • • Oxidizing agent gets reduced. Gains electrons. More negative oxidation state. Reducing agent gets oxidized. Loses electrons. More positive oxidation state.

Half-Reactions • All redox reactions can be thought of as happening in two halves. • One produces e-’s = Oxidation half. • One requires e-’s = Reduction half.

Balancing Redox Reactions • In aqueous solutions the key is the number of electrons produced must be the same as those required. • For reactions in acidic solution an 8 step procedure. 1 Write separate half reactions 2 For each half reaction balance all reactants except H and O 3 Balance O using H 2 O

Acidic Solution 4 Balance H using H+ 5 Balance charge using e 6 Multiply equations to make electrons equal 7 Add equations and cancel identical species 8 Check that charges and elements are balanced.

Balancing Redox Reactions Al + __ Cu 2+ --> __ Cu + __ Al 3+ Start by writing half reactions (Oxidation and reduction) (Electrons go on the more positive side) Oxidation: Al --> Al 3+ + 3 e. Reduction: 2 e- + Cu 2+ --> Cu • __ • •

Balancing Redox Reactions 2. Balance the electrons by finding the common multiple and multiply the half reactions accordingly. • The common multiple of the electrons is 6 so • Oxidation: 2 x (Al --> Al 3+ + 3 e-) • Reduction: 3 x ( 2 e- + Cu 2+ --> Cu)

Balancing Redox Reactions • Oxidation: 2 x (Al --> Al 3+ + 3 e-) • Reduction: 3 x ( 2 e- + Cu 2+ --> Cu) • _________________ • Recombine: 6 e-+2 Al + 3 Cu 2+-->2 Al 3++ 3 Cu + 6 e- • The electrons must cancel. • 2 Al + 3 Cu 2+--> 2 Al 3+ + 3 Cu • Atoms and charges must be conserved.

Balancing Redox Reactions (Acidic Conditions) • Mn. O 4 - + I- --> I 2 + Mn 2+ (acidic) • Step 1 Half Reactions: • Mn. O 4 - --> Mn 2+ • I- --> I 2

Lets balance the reduction one first: • for every Oxygen add a water on the other side: • Mn. O 4 - --> Mn 2+ + 4 H 2 O • For every hydrogen add a H+ to the other side: • 8 H+ + Mn. O 4 - --> Mn 2+ + 4 H 2 O • Balance the imbalance of charge with electrons (+7 vs. +2): • 5 e- + 8 H+ + Mn. O 4 - --> Mn 2+ + 4 H 2 O
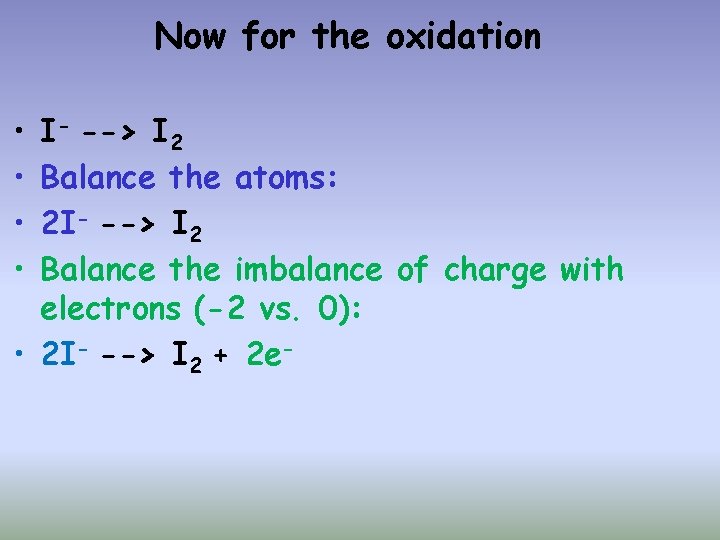
Now for the oxidation • • I- --> I 2 Balance the atoms: 2 I- --> I 2 Balance the imbalance of charge with electrons (-2 vs. 0): • 2 I- --> I 2 + 2 e-

Balancing Redox Reactions • Balance the electrons by finding the common multiple and multiply the half reactions accordingly. • Common Multiple here is 10.

Balancing Redox Reactions • 2( 5 e- + 8 H+ + Mn. O 4 - --> Mn 2+ + 4 H 2 O ) • 5( 2 I- --> I 2 + 2 e- ) • Step 3 Check electrons, atoms and charge. Clean it up. • 10 e- + 16 H+ + 2 Mn. O 4 - + 10 I--->5 I 2 + 2 Mn 2+ + 8 H 2 O + 10 e • 16 H+ +2 Mn. O 4 - +10 I-->5 I 2 +2 Mn 2+ + 8 H 2 O

Basic Solution • Do everything you would with acid, but add one more step. • Add enough OH- to both sides to neutralize the H+ • Makes water

Basic Solution • Cr(OH)3 +Cl. O 3 - ->Cr. O 42 - + Cl- (basic) • Step 1 Half Reactions: • Lets balance the reduction one first: • Cl. O 3 - --> Cl • for every Oxygen add a water on the other side: • Cl. O 3 - --> Cl- + 3 H 2 O • For every hydrogen add a H+ to the other side: • 6 H+ + Cl. O 3 - --> Cl- + 3 H 2 O

Basic Solution • Each H+ will react with an OH- on both sides: • 6 OH- + 6 H++Cl. O 3 - -> Cl- +3 H 2 O + 6 OH • H+ and OH- make water: • 6 H 2 O + Cl. O 3 - --> Cl- + 3 H 2 O + 6 OH • cancel the waters: • 3 H 2 O + Cl. O 3 - --> Cl- + 6 OH • Balance the imbalance of charge with e-’s (-1 vs. -7): • 6 e- + 3 H 2 O + Cl. O 3 - --> Cl- + 6 OH-
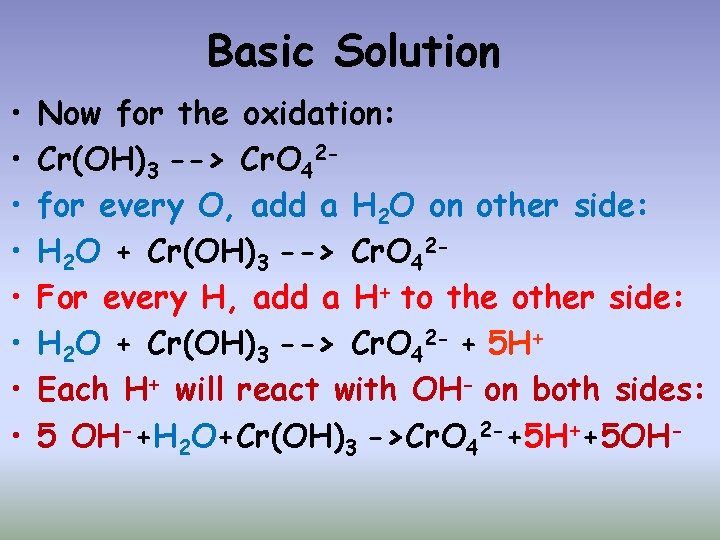
Basic Solution • • Now for the oxidation: Cr(OH)3 --> Cr. O 42 for every O, add a H 2 O on other side: H 2 O + Cr(OH)3 --> Cr. O 42 For every H, add a H+ to the other side: H 2 O + Cr(OH)3 --> Cr. O 42 - + 5 H+ Each H+ will react with OH- on both sides: 5 OH-+H 2 O+Cr(OH)3 ->Cr. O 42 -+5 H++5 OH-

Basic Solution • • • H+ and OH- make water: 5 OH- +H 2 O + Cr(OH)3 -> Cr. O 42 - + 5 H 2 O cancel the waters: 5 OH- + Cr(OH)3 --> Cr. O 42 - + 4 H 2 O Balance the imbalance of charge with electrons (-5 vs. -2): • 5 OH- + Cr(OH)3 -> Cr. O 42 - + 4 H 2 O + 3 e-

Basic Solution • Balance the electrons by finding the common multiple and multiply the half reactions accordingly. • Common Multiple here is 6: • 1(6 e- + 3 H 2 O + Cl. O 3 - -> Cl- + 6 OH-) • 2(5 OH-+Cr(OH)3 -> Cr. O 42 -+4 H 2 O + 3 e-)
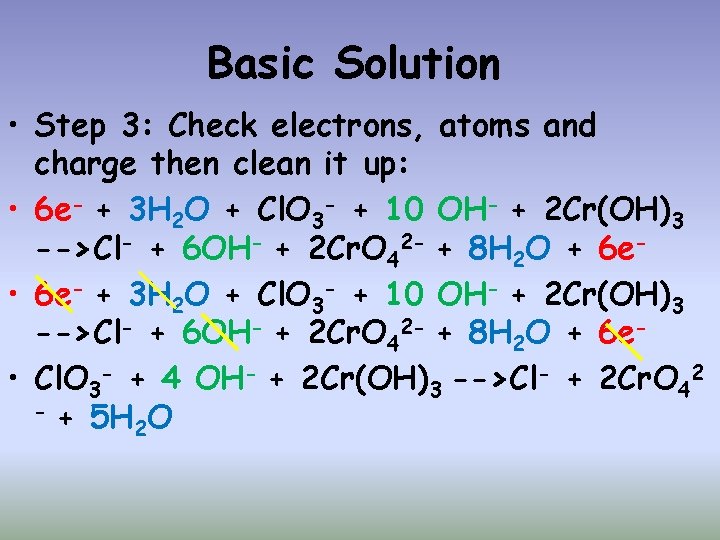
Basic Solution • Step 3: Check electrons, atoms and charge then clean it up: • 6 e- + 3 H 2 O + Cl. O 3 - + 10 OH- + 2 Cr(OH)3 -->Cl- + 6 OH- + 2 Cr. O 42 - + 8 H 2 O + 6 e • Cl. O 3 - + 4 OH- + 2 Cr(OH)3 -->Cl- + 2 Cr. O 42 - + 5 H O 2

Practice Redox Reaction #1 Mn 2+ + Bi. O 3 - => Mn. O 4 - + Bi 3+ 2 (4 H 2 O + Mn 2+ => Mn. O 4 - + 8 H + 5 e) 5 ( 2 e + 6 H + + Bi. O 3 - => Bi 3+ + 3 H 2 O) 14 H + + 2 Mn 2+ + 5 Bi. O 3 - => 2 Mn. O 4 - + 5 Bi 3+ + 7 H 2 O

Practice Redox Reaction #2 Cl. O 3 - + Cl - => Cl 2 + Cl. O 2 2 (e + 2 H + + Cl. O 3 - => Cl. O 2 + H 2 O) 2 Cl - => Cl 2 + 2 e) 4 H + + 2 Cl. O 3 - + 2 Cl - => 2 Cl. O 2 + 2 H 2 O + Cl 2

Practice Redox Reaction #3 P + Cu 2+ => Cu + H 2 PO 4 2 (4 H 2 O + P => H 2 PO 4 - + 6 H + + 5 e) 5 (2 e + Cu 2+ => Cu) 8 H 2 O + 2 P + 5 Cu 2+ - + 12 H + + 5 Cu) => 2 H 2 PO 4

Practice Redox Reaction #4 Mn. O 4 - + C 2 O 4 2 - => Mn. O 2 + CO 2 2 (3 e + 4 H + + Mn. O 4 - => Mn. O 2 + 2 H 2 O) 3 (C 2 O 4 2 - => 2 CO 2 + 2 e) 4 H 2 O + 2 Mn. O 4 - + 3 C 2 O 4 2 - => + 8 OH - + 6 CO 2 2 Mn. O 2

Practice Redox Reaction #5 Cl. O 2 => Cl. O 2 - + Cl. O 3 e + Cl. O 2 => Cl. O 2 H 2 O + Cl. O 2 => Cl. O 3 - + 2 H + + e 2 OH - + 2 Cl. O 2 => Cl. O 2 - + Cl. O 3 - + H 2 O

Practice Redox Reaction #6 H 2 O + Zn + NO 3 - => Zn(OH)4 2 - + NH 3 8 e + 9 H + + NO 3 - => NH 3 + 3 H 2 O 4 (4 H 2 O + Zn => Zn(OH)4 2 - + 4 H + + 2 e 6 H 2 O + NO 3 - + 7 OH - + 4 Zn Zn(OH)4 2 - => NH 3 + 4
- Slides: 40