Redox and Electrochemistry Redox Reactions Reduction Oxidation reactions
























- Slides: 25

Redox and Electrochemistry

Redox Reactions • Reduction – Oxidation reactions • Involve the transfer of electrons from one substance to another + The oxidation numbers of the atoms will change…. one goes up (oxidation) and one goes down (reduction)

Oxidation Number (Oxidation State) • Used to keep track of the transfer of electrons • Number is assigned to every atom in a chemical formula, in accordance with certain rules • NOT an ionic charge, but is often the same as the ionic charge – Possible oxidation states are given on the periodic table (upper right hand corner)

Rules for assigning Oxidation Numbers 1. For a neutral compound, the sum of the oxidation states must be zero 2. The oxidation state of any atom in an uncombined element is zero • • Element not in chemical combination with another element Examples: Na, Mg, H 2, Cl 2

Rules for assigning Oxidation Numbers 3. The oxidation state of a monatomic ion is equal to its charge – Examples: Na+ = 4. In an ionic salt, the oxidation number of each ion is equal to its charge – Examples: Ca. Cl 2 5. For a polyatomic ion, the sum of the oxidation states must equal the overall charge – Example: SO 42 -

Rules for assigning Oxidation Numbers 6. Metals of group 1 always have an oxidation number of +1 7. Metals of groups 2 always have an oxidation number of +2 8. Fluorine is always -1, other halogens are usually -1 9. Aluminum is always +3

Rules for assigning Oxidation Numbers 10. Oxygen is usually -2 Exceptions: – When paired with F (OF 2), oxygen will be +2 – Peroxides (H 2 O 2), oxygen will be -1 11. Hydrogen is usually +1 Exceptions: – Metal hydrides (Group 1 or 2 metals paired with hydrogen), Li. H, Ca. H 2, hydrogen will be -1

Examples Assign an oxidation state to each element in the following: 1. H 2 SO 4 2. SO 323. K 2 Cr. O 4 4. Cr. Cl 3

• Reduction – Reduction of charge by gaining electrons Na+ + e- Na O + 2 e- → O 2 - • Oxidation – Increase in charge by loss of electrons Fe 3+ + 3 e. Cl- Cl + e-

LEO the lion says GER Losing Electrons Oxidation Gaining Electrons Reduction

Conservation of Matter/ Conservation of Charge • Mass must be conserved – Mass on both sides must be the same (balanced) • Charge must be conserved – Net charge on both sides must be the same (balanced) – add electrons to the higher side • Reduction and Oxidation reactions must occur together (REDOX reactions)

Half Reactions • Every Redox reaction consists of a reduction and oxidation reaction • Each reaction is called a ½ reaction • A separate equation can be written for each ½ reaction

Half Reactions • Net charge and mass must be the same on both sides of the equation • The number of electrons must balance out, electrons do not appear in the net equation • One ½ reaction is reduction and the other is oxidation

Spectator Ion • Does not change oxidation states in the reaction, same oxidation state on both sides of the equation • Not every species in an equation is oxidized or reduced, some are spectator ions

1. Assign oxidation states to each element in the reaction 2. Identify the 2 substances that are changing oxidation states 3. Write the half reactions • • Balance the mass Balance the charge (add electrons to the higher side)

Examples 1. H 2 + Cl 2 2 HCl 2. Fe + Zn. O Zn + Fe. O

Reducing Agent • Substance which is oxidized – Serves as a source of electrons to make the reduction reaction occur – Good reducing agents are substances that lose (donate) electrons easily – elements with low ionization energies Examples: group 1 and 2 metals

Oxidizing Agent • Substance which is reduced – Accepts (gains electrons) – Good oxidizing agents are substances that gain electrons (highly electronegative elements) Examples: Group 17 elements

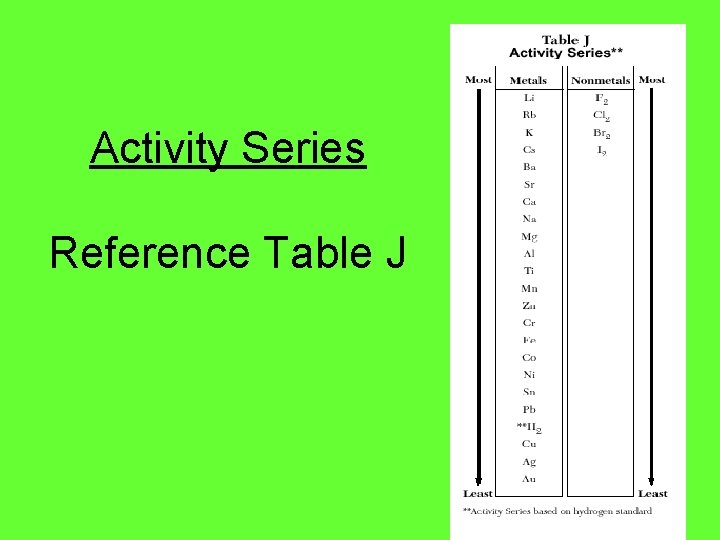
Activity Series Reference Table J

Metals • The most reactive metals are listed at the top • A reaction will occur spontaneously if the metal is higher than the metal ion that it is trying to replace • Reactive metals lose electrons easily (low ionization energy) • Higher on the table = More likely to be oxidized

Examples Ba + Zn. Cl 2 → Zn + Ba. Cl 2 • Ba will replace Zn because Ba is above Zn – Ba is more reactive than Zn • More reactive means that it loses electrons easier

Nonmetals • For the halogen nonmetals listed in Table J, the most reactive ones are at the top • For nonmetals, high reactivity means that they are likely to gain electrons (high electronegativity) • Higher on the table = More likely to be reduced Example: F 2 will replace any other halogen (it is the most reactive)

Examples 1. Which metal is most reactive? a. Fe b. Zn Cu 2. Will Ba react with Mn 2+? 3. Will Na+ react with Cr? 4. Will this reaction occur spontaneously? Mg + Co(NO 3)2 → 5. If this reaction does occur, what products would be made?

Balancing Equations 1. Assign oxidation numbers to all substances in the equation 2. Write the oxidation and reduction ½ reactions 3. Balance (cancel out) the electrons in the ½ reaction 4. Balance the rest of the equation 5. Check

Examples 1. Fe + Cl 2 → Fe. Cl 3 2. Fe + Cu 2+ Cu + Fe 3+ 3. KMn. O 4 + HCl → KCl + Mn. Cl 2 + H 2 O + Cl 2