Balancing Chemical Equations A chemical reaction is a

Balancing Chemical Equations A chemical reaction is a process by which one set of chemicals is transformed into a new set of chemicals. A chemical equation uses standard chemical symbols to describe the changes occurring during a reaction General form: reactants products Example: Water Skeletal equation H 2 + O 2 H 2 O Hydrogen and oxygen combine to form water. But the equation is not balanced. There are 2 oxygen atoms on the reactant side of the equation, but only one on the product side. If we place a 2 in front of the water on the product side, we will balance the oxygens. H 2 + O 2 2 H 2 O

We now need to balance the hydrogens Place a 2 before the hydrogens on the reactant side of the equation 2 H 2 + O 2 2 H 2 O Balanced Note: Diatomic molecules Equation formation of water included hydrogens and oxygens with subscript 2. Other elements which occur in this way are Nitrogen, Fluorine, Chlorine, Bromine and Iodine. These elements occur naturally as diatomic (meaning 2 atoms) molecules The stoichiometric coefficients multiplying the chemical formulas tell you the relative numbers of moles of each substance that reacts or is produced in a chemical reaction. Therefore, we can conclude from the balanced equation for water that 2 moles of hydrogen and one mol of oxygen combine to form water.

A chemical equation may also tell you what physical state the reactants and products are in. The following state symbols are used: • s (solid) • l (liquid) • g (gas) • aq (aqueous solution) These are found as subscripts after each reactant and product. Example Balance the following equation: Na(s) + H 2 O(l) Na. OH(aq) + H 2(g) Step 1: How many atoms of each element are present on the reactant and product side? Reactants Products Na H O 1 2 1 1 3 1
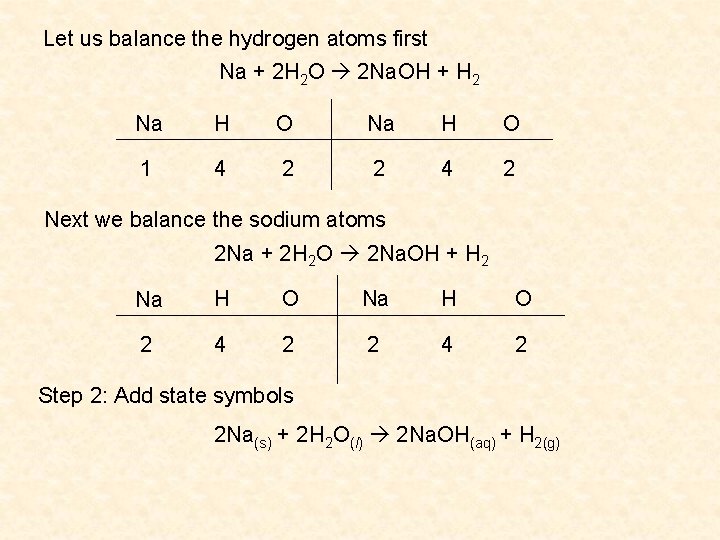
Let us balance the hydrogen atoms first Na + 2 H 2 O 2 Na. OH + H 2 Na H O 1 4 2 2 4 2 Next we balance the sodium atoms 2 Na + 2 H 2 O 2 Na. OH + H 2 Na H O 2 4 2 Step 2: Add state symbols 2 Na(s) + 2 H 2 O(l) 2 Na. OH(aq) + H 2(g)

Questions Balance the following equations: (a) P 2 O 5(s) + H 2 O(l) H 3 PO 4(aq) Answer: P 2 O 5(s) + 3 H 2 O(l) 2 H 3 PO 4(aq) (b) NO 2(g) + H 2(g) NH 3(g) + H 2 O(g) Answer: NO 2(g) + 3 ½ H 2(g) NH 3(g) + 2 H 2 O(g) (c) SO 2(g) + O 2(g) SO 3(g) Answer: 2 SO 2(g) + O 2(g) 2 SO 3(g)

Balancing redox reactions • Redox (meaning reduction-oxidation) reactions involves transfer of electrons • Involves two processes oxidation reduction OIL RIG is is loss gain Whenever oxidation occurs, reduction must also occur Can split a redox reaction into two half reactions: The oxidation reaction which involves a loss of electrons The reduction reaction which involves a gain of electrons To keep track of electrons in redox reactions, use oxidation numbers Oxidation number: the charge an atom has or appears to have when electrons are distributed according to certain rules

Rules for assigning oxidation numbers (O. N. ) 1. 2. 3. 4. 5. 6. 7. 8. A free element (or uncombined element) is assigned O. N. of zero The sum of O. N. s of atoms in a neutral compound must equal zero The charge of a monoatomic ion is same as its oxidation number, e. g. Li+ has O. N. of +1 Fe+3 has O. N. of +3 In their compounds, group 1 elements have an O. N. of +1, group 2 elements have an O. N. of +2. The sum of O. N. of atoms in polyatomic ion equals the charge on the ion The O. N. of halogens is usually -1. Exception: when they are bonded to a more electonegative element The O. N. of oxygen in most compounds is -2. Exceptions: H 2 O 2 and peroxide ion, O 2 -2 (O. N. of -1), and the compound OF 2 (O. N. +2) The O. N. of hydrogen is +1, Exception: when bonded to a less electronegative element, i. e. a metal, where it’s O. N. is -1, e. g. Li. H, Na. H

Examples Assign oxidation numbers to all the elements in the following: (a) Li 2 O Lithium: Group 1 element Oxidation number +1 Oxygen: Oxidation number -2 (b) HNO 3 Hydrogen: +1 Oxygen: -2 HNO 3 is neutral adding the O. N. s of the atoms together must equal zero (+1) + N + 3(-2) = 0 N = +5 (c) Cr 2 O 72 Sum of O. N. s equals charge on ion here the sum equals -2 Oxygen: Oxidation number -2 2 Cr + 7(-2) = -2 2 Cr -14 = -2 Cr = +6

Questions Assign oxidation numbers to each of the following: (a) Zn. Cl 2 Answer: Zn: +2 Cl: -1 Answer: O: -2 S: +6 Answer: O: -2 Mn: +7 (b) SO 3 (c) Mn. O 4 -

Defining Oxidising and Reducing agents • Remember that for an oxidation to occur, a reduction must also occur • OIL RIG • Oxidation can be defined (in terms of electon transfer) as the loss of electrons from a species • Reduction can be defined as the gain of electrons by a species • An oxidising agent (or electron acceptor) must itself be reduced • A reducing agent (or electron donor) must itself be oxidised • The action of both oxidising and reducing agents can be represented in the form of a half-equation

Identifying Oxidising and Reducing Agents Example Formation of calcium oxide (Ca. O) 2 Ca(s) + O 2(g) 2 Ca. O(s) Ca and O 2 (reactants): neutral atoms Calcium oxide (product): ionic compound Ca 2+ O 2 ions Can write the reaction as two half-reactions: 2 Ca 2+ + 4 e. O 2 + 4 e- 2 O 2 - Here, calcium is oxidised; i. e. it loses electrons to oxygen OIL RIG It reduces the oxygen it acts as a reducing agent Oxygen is reduced; i. e. it gains electrons from calcium It oxidises the calcium it acts as an oxidising agent
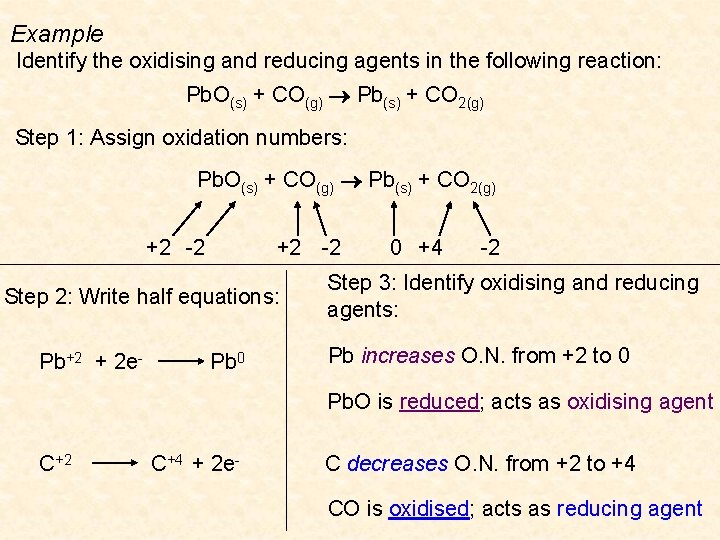
Example Identify the oxidising and reducing agents in the following reaction: Pb. O(s) + CO(g) Pb(s) + CO 2(g) Step 1: Assign oxidation numbers: Pb. O(s) + CO(g) Pb(s) + CO 2(g) +2 -2 Step 2: Write half equations: Pb+2 + 2 e- Pb 0 0 +4 -2 Step 3: Identify oxidising and reducing agents: Pb increases O. N. from +2 to 0 Pb. O is reduced; acts as oxidising agent C+2 C+4 + 2 e- C decreases O. N. from +2 to +4 CO is oxidised; acts as reducing agent

Questions Identify the oxidising and reducing agents and write the half reactions in the following: (a) Cl 2 + 2 Na. Br 2 Na. Cl + Br 2 Answer: 2 Br-- Br 2 + 2 e. Cl 2 + 2 e- 2 Cl. Cl 2 : oxidising agent Br - : reducing agent (b) Si + 2 F 2 Si. F 4 Answer: Si 4+ + 4 e. F 2 + 2 e- 2 FF 2 : oxidising agent Si : reducing agent (c) 4 Fe +3 O 2 2 Fe 2 O 3 Answer: Fe 3+ + 3 e. O 2 + 4 e- 2 O 2 O 2 : oxidising agent Fe: reducing agent

Example Balancing redox equations Balance the equation showing the oxidation of Fe 2+ ions to Fe 3+ ions by dichromate ions (Cr 2 O 72 -) in an acidic medium Fe 2+ + Cr 2 O 72 - Fe 3+ + Cr 3+ Step 1: Identify oxidising and reducing agents and write half reactions Fe 2+ : +2 Cr 2 O 72 - : 2 Cr + 7(-2) = -2 Fe 3+ : +3 2 Cr = 12 Cr 3+ : +3 Cr = +6 Fe 2+ Fe 3+ Iron loses one electron, i. e. it is oxidised. It donates one electron to dichromate, i. e. it is the reducing agent. Half equation: Oxidation reaction: Fe 2+ Fe 3+ + e- Cr 26+ Cr 3+ Chromium gains three electrons, i. e. it is reduced. It gains its electrons from iron, i. e. it is the oxidising agent. Half equation: Reduction reaction: Cr 26+ + 3 e- Cr 3+

Fe 2+ + Cr 2 O 72 - Fe 3+ + Cr 3+ Step 2: Balance each kind of atom other that H and O Fe 2+ Fe 3+ + e. Cr 2 O 72 - + 3 e- 2 Cr 3+ Step 3: Balance oxygen atoms by using H 2 O Fe 2+ Fe 3+ + e- Cr 2 O 72 - + 3 e- 2 Cr 3+ + 7 H 2 O Step 4: Balance H atoms by using H+ ions Fe 2+ Fe 3+ + e- 14 H+ + Cr 2 O 72 - + 3 e- 2 Cr 3+ + 7 H 2 O Step 5: Use electrons as needed to obtain a charge that is balanced Fe 2+ Fe 3+ + e+2 +3 -1 14 H+ + Cr 2 O 72 - +3 e- 2 Cr 3+ + 7 H 2 O +14 -2 -3 +6 0 +2 +2 +9 +6 Add three electrons to the reactant side to balance the charges Fe 2+ Fe 3+ + e- 14 H+ + Cr 2 O 72 - + 6 e- 2 Cr 3+ + 7 H 2 O

Step 6: Ensure the number of electrons gained equals the number of electrons lost and add the 2 half reactions together Fe 2+ Fe 3+ + e- × 6 14 H+ + Cr 2 O 72 - + 6 e- 2 Cr 3+ + 7 H 2 O × 1 6 Fe 2+ 6 Fe 3+ + 6 e 14 H+ + Cr 2 O 72 - + 6 e- 2 Cr 3+ + 7 H 2 O 14 H+ + Cr 2 O 72 - + 6 Fe 2+ + 6 e- 2 Cr 3+ + 7 H 2 O + 6 Fe 3+ + 6 e 14 H+ + Cr 2 O 72 - + 6 Fe 2+ 2 Cr 3+ +7 H 2 O + 6 Fe 3+ Balanced Check to ensure that all atoms and charges are balanced.

Question Balance the equation for the redox reaction of Mn. O 4 - with Fe 2+ to produce Mn 2+ and Fe 3+ in an acidic medium. Procedure: • Step 1: Identify the oxidising and reducing agents, then write the half equations • Step 2: Balance each atom, except H and O • Step 3: Balance the O atoms (using H 2 O) • Step 4: Balance the H atoms (using H+) • Step 5: Check the charges are balanced for each half equation • Step 6: Ensure the numbers of electrons are equal (i. e. there are the same numbers lost and gained) • Step 7: Add the two equations together • Finally: check that all the charges and the numbers of atoms balance!

Question Balance the equation for the redox reaction of Mn. O 4 - with Fe 2+ to produce Mn 2+ and Fe 3+ in an acidic medium. Answer Step 1: Identify oxidising and reducing agents and write half reactions Mn. O 4 - Mn 2+ Mn + 4(-2) = -1 Mn = +7 Mn : +2 Mn. O 4 - gains 5 electrons. It acts as the oxidising agent as it is reduced. Mn. O 4 - + 5 e- Mn 2+ Reduction reaction Fe 2+ Fe 3+ Fe loses an electron. It acts as the reducing agent as it is oxidised. Fe 2+ Fe 3+ + e- Oxidation reaction

Step 2: Balance each kind of atom other than H and O. Balanced in this case Step 3: Balance O atoms by adding H 2 O Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O Fe 2+ Fe 3+ + e. Step 4: Balance H atoms by using H+ ions 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O Fe 2+ Fe 3+ + e. Step 5: Use electrons as needed to obtain a charge that is balanced Fe 2+ Fe 3+ + e+2 +2 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O +2 Already balanced! +2

Step 6: Ensure the number of electrons gained equals the number of electrons lost and add the two half reactions together Fe 2+ Fe 3+ + e- × 5 × 1 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O 5 Fe 2+ 5 Fe 3+ + 5 e 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O 8 H+ + Mn. O 4 - + 5 Fe 2+ + 5 e- Mn 2+ + 5 Fe 3+ + 4 H 2 O + 5 e 8 H+ + Mn. O 4 - + 5 Fe 2+ Mn 2+ + 5 Fe 3+ + 4 H 2 O Balanced Check to ensure that all atoms and charges are balanced.

Question Balance the following equation, which takes place in an acidic medium: Mn. O 4 - + SO 2 Mn 2+ + SO 42 Step 1: Identify oxidising and reducing agents and write half reactions Mn. O 4 -: Mn +4(-2) = -1 Mn - 8 = -1 Mn = +7 Mn 2+ : +2 Mn. O 4 - gains 5 electrons: it acts as the oxidising agent as it is reduced Mn. O 4 - + 5 e- Mn 2+ SO 2: S + 2(-2) = 0 S-4=0 S = +4 Reduction reaction SO 42 -: S + 4(-2) = -2 S - 8 = -2 S = +6 SO 2 loses 2 electrons: it acts as the reducing agent as it is oxidised SO 2 SO 42 - + 2 e- Oxidation reaction
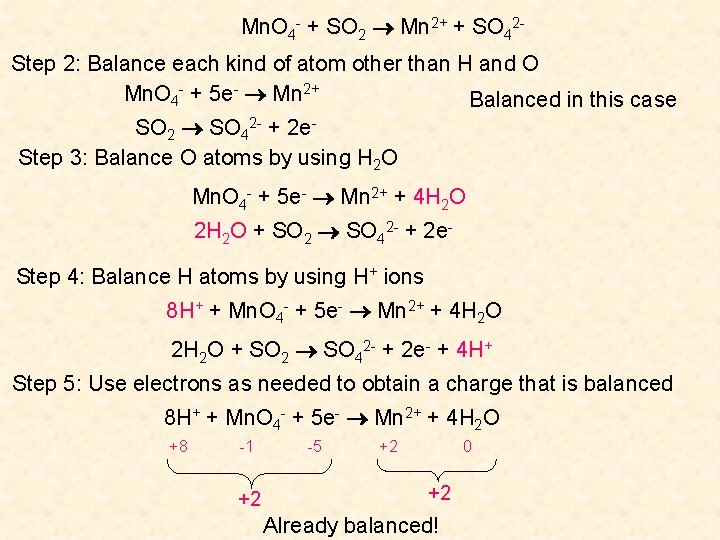
Mn. O 4 - + SO 2 Mn 2+ + SO 42 Step 2: Balance each kind of atom other than H and O Mn. O 4 - + 5 e- Mn 2+ Balanced in this case SO 2 SO 42 - + 2 e. Step 3: Balance O atoms by using H 2 O Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O 2 H 2 O + SO 2 SO 42 - + 2 e. Step 4: Balance H atoms by using H+ ions 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O 2 H 2 O + SO 2 SO 42 - + 2 e- + 4 H+ Step 5: Use electrons as needed to obtain a charge that is balanced 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O +8 -1 +2 -5 +2 0 +2 Already balanced!

2 H 2 O + SO 2 SO 42 - + 2 e- + 4 H+ 0 0 0 -2 -2 +4 0 Already balanced Step 6: Ensure number of electrons gained equals number of electrons lost and add the two half reactions together 8 H+ + Mn. O 4 - + 5 e- Mn 2+ + 4 H 2 O × 2 2 H 2 O + SO 2 SO 42 - + 2 e- + 4 H+ × 5 16 H+ + 2 Mn. O 4 - + 10 e- 2 Mn 2+ + 8 H 2 O 10 H 2 O + 5 SO 2 5 SO 42 - + 10 e- + 20 H+ 16 H+ + 2 Mn. O 4 - + 10 H 2 O + 5 SO 2 2 Mn 2+ + 8 H 2 O + 5 SO 42 - + 20 H+ 2 Mn. O 4 - + 2 H 2 O + 5 SO 2 2 Mn 2+ + 5 SO 42 - + 4 H+ Balanced Check to ensure that all atoms and charges are balanced.

Questions Write balanced equations to represent the following reactions in an acidic solution: (a) I- + SO 42 - I 2 + S Answer: 6 I- + 8 H+ + SO 42 - 3 I 2 + S + 4 H 2 O (b) Mn. O 4 - + H 2 C 2 O 4 Mn 2+ + CO 2 Answer: 2 Mn. O 4 - + 5 H 2 C 2 O 4 + 6 H+ 2 Mn 2+ + 10 CO 2 + 8 H 2 O (c) Cl. O 3 - + Cl- Cl 2 + Cl. O 2 Answer: 2 Cl- + 2 Cl. O 3 - + 4 H+ Cl 2 + 2 Cl. O 2 + 2 H 2 O

Example In basic solution Write a balanced equation to represent the oxidation of iodide ion (I-) by permanganate ion (Mn. O 4 -) in basic solution to yield molecular iodine (I 2) and manganese (IV) oxide (Mn. O 2) Skeletal equation: I- + Mn. O 4 - I 2 + Mn. O 2 Step 1: Identify oxidising and reducing agents and write half equations I- I 2 O. N. of I- is -1 O. N of I 2 is 0 I- loses an electron: it acts as the reducing agent as it is oxidised I- I 2 + e Mn. O 4 - Mn. O 2 Oxidation reaction Mn. O 4 -: Mn + 4(-2) = -1 Mn = +7 Mn. O 2 : Mn + 2(-2) = 0 Mn = +4 Mn. O 4 - gains 3 electrons: it acts as the oxidising agent as it is reduced Mn. O 4 - + 3 e- Mn. O 2 Reduction reaction

Step 2: Balance each kind of atom other than H and O 2 I- I 2 + e. Mn. O 4 - + 3 e- Mn. O 2 Step 3: Balance the O atoms by using H 2 O 2 I- I 2 + e. Mn. O 4 - + 3 e- Mn. O 2 + 2 H 2 O Step 4: Balance H atoms by using H+ ions 2 I- I 2 + e 4 H+ + Mn. O 4 - + 3 e- Mn. O 2 + 2 H 2 O

Step 5: Since the reaction occurs in a basic medium, for each H+ ion we add an equal number of OH- ions to both sides of the equation. Where H+ and OH- ions appear on the same side of the equation, they may be combined to form H 2 O. 4 H+ + 4 OH- + Mn. O 4 - + 3 e- Mn. O 2 + 2 H 2 O + 4 OH- 4 H 2 O + Mn. O 4 - + 3 e- Mn. O 2 + 2 H 2 O + 4 OH 2 H 2 O + Mn. O 4 - + 3 e- Mn. O 2 + 4 OHStep 6: Use electrons as needed to obtain a charge that is balanced 2 I- I 2 + e-2 0 -2 -1 -1 Add one electron to the product side to balance charges 2 I- I 2 + 2 e 2 H 2 O + Mn. O 4 - + 3 e- Mn. O 2 + 4 OH 0 -1 -3 -4 0 -4 -4 Already balanced!

Step 7: Ensure the number of electrons gained equals the number of electrons lost and add the two half reactions together 2 I- I 2 + 2 e 2 H 2 O + Mn. O 4 - + 3 e- Mn. O 2 + 4 OH- × 3 × 2 6 I- 3 I 2 + 6 e 4 H 2 O + 2 Mn. O 4 - + 6 e- 2 Mn. O 2 + 8 OH 2 Mn. O 4 - + 6 I- + 4 H 2 O 2 Mn. O 2 + 3 I 2 + 8 OHBalanced Check to ensure that all atoms and charges are balanced.

Question Balance the following redox equation which occurs in basic solution Mn 2+ + H 2 O 2 Mn. O 2 + H 2 O Step 1: Identify the oxidising and reducing agents and write half reactions Mn 2+ Mn. O 2 Mn 2+ : +2 Mn. O 2: Mn +2(-2) = 0 Mn = +4 Mn 2+ loses two electrons: it acts as the reducing agent as it is oxidised Mn 2+ Mn. O 2 + 2 e. H 2 O 2 H 2 O Oxidation reaction H 2 O 2: 2(+1) + 2 O = 0 O = -1 H 2 O: 2(+1) + O = 0 O = -2 H 2 O 2 gains one electron: it acts as the oxidising agent as it is reduced H 2 O 2 + e - H 2 O Reduction reaction

Step 2: Balance each kind of atom other than H and O. Mn 2+ Mn. O 2 + 2 e. Already balanced in this case H 2 O 2 + e - H 2 O Step 3: Balance the O atoms by using H 2 O 2 H 2 O + Mn 2+ Mn. O 2 + 2 e. H 2 O 2 + e- 2 H 2 O Step 4: Balance the H atoms by using H+ 2 H 2 O + Mn 2+ Mn. O 2 + 2 e- + 4 H+ 2 H+ + H 2 O 2 + e- 2 H 2 O Step 5: For each H+ ion, add equal no. of OH- to both sides of equation 2 H 2 O + Mn 2+ + 4 OH- Mn. O 2 + 2 e- + 4 H+ + 4 OH 2 H 2 O + Mn 2+ + 4 OH- Mn. O 2 + 2 e- + 4 H 2 O Mn 2+ + 4 OH- Mn. O 2 + 2 e- + 2 H 2 O
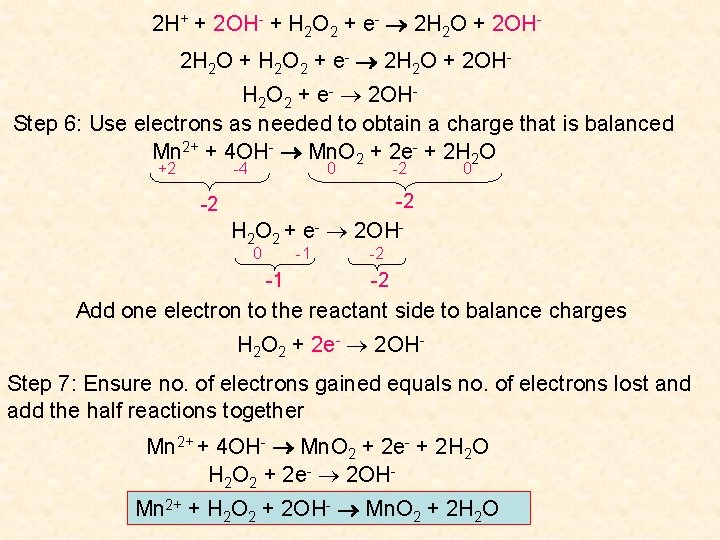
2 H+ + 2 OH- + H 2 O 2 + e- 2 H 2 O + 2 OH 2 H 2 O + H 2 O 2 + e- 2 H 2 O + 2 OHH 2 O 2 + e- 2 OHStep 6: Use electrons as needed to obtain a charge that is balanced Mn 2+ + 4 OH- Mn. O 2 + 2 e- + 2 H 2 O +2 -4 0 -2 -2 H 2 O 2 + e- 2 OH-1 0 -1 -2 -2 Add one electron to the reactant side to balance charges H 2 O 2 + 2 e- 2 OHStep 7: Ensure no. of electrons gained equals no. of electrons lost and add the half reactions together Mn 2+ + 4 OH- Mn. O 2 + 2 e- + 2 H 2 O 2 + 2 e- 2 OHMn 2+ + H 2 O 2 + 2 OH- Mn. O 2 + 2 H 2 O

Questions Write balanced equations to represent the following reactions in a basic solution (a) Fe(OH)2 + Mn. O 4 - Mn. O 2 + Fe(OH)3 Answer: 3 Fe(OH)2 + Mn. O 4 - + 2 H 2 O Mn. O 2 + 3 Fe(OH)3 + OH- (b) Bi(OH)3 + Sn. O 22 - Sn. O 32 - + Bi Answer: 2 Bi(OH)3 + 3 Sn. O 22 - 2 Bi + 3 H 2 O + 3 Sn. O 32 -
- Slides: 32