Electrochemistry Lesson 6 Balancing Redox Reactions Balancing Redox

Electrochemistry Lesson 6 Balancing Redox Reactions

Balancing Redox Full Reactions in Acid Solution 4. 3. 5. 2. 1. Simplify Add the two half reactions Check Separate Balance each into half reactions Mn. O 4 - + C 2 O 42 - → 2( Mn. O 4 - + 4 H+ + 3 e- → Mn. O 2 + CO 32+ 2 H 2 O ) 3(C 2 O 42 - + 2 H 2 O → 2 CO 32 - + 4 H+ + 2 e-) 2 4 2 Mn. O 4 - + 3 C 2 O 42 - + 6 H 2 O + 8 H+ → 2 Mn. O 2 + 6 CO 32 - + 12 H+ + 4 H 2 O -8 -8 2 Mn. O 4 - + 3 C 2 O 42 - + 2 H 2 O → 2 Mn. O 2 + 6 CO 32 - + 4 H+

Balancing Redox Full Reactions in Basic Solution 1. 2. - toacid Balance Add OHin neutralize solutionthe H+ 2 Mn. O 4 - + 3 C 2 O 42 - + 2 H 2 O → 2 Mn. O 2 + 6 CO 32 - + 4 OH- 4 H+ 4 OH 2 4 H 2 O

Balancing Redox Full Reactions in Basic Solution 2 Mn. O 4 - + 3 C 2 O 42 - + 4 OH- → 2 Mn. O 2 + 6 CO 32 - + 2 H 2 O -12

Balancing Redox Full Reactions in Acid Solution Fe + O 2 4( Fe + → 3 H 2 O 3( 4 e- + O 2 + H 2 O → 4 H+ + 3 H+ Fe(OH)3 + 3 e -) → H 20 + H 2 O) 6 4 Fe + 12 H 2 O + 3 O 2 +12 H+ → 12 H+ + 4 Fe(OH)3 + 6 H 2 O 4 Fe + 6 H 2 O + 3 O 2 → 4 Fe(OH)3 0 0

Balance the redox reaction As → H 2 As. O 4+ As. H 3 You must separate into two half reactions! 3(As + 5( 3 H+ 4 H 2 O + As → H 2 As. O 4 - + 3 e- → + (alkaline) 6 H+ + As. H 3 ) 3 As + 12 H 2 O + 15 H+ + 5 As → 3 H 2 As. O 4 - 5 e-) 3 + 18 H+ + 5 As. H 3 8 As + 12 H 2 O → 3 H 2 As. O 4 - + 3 H+ + 5 As. H 3 3 OH 3 OH- + 8 As + 9 H 2 O → 3 H 2 As. O 4 - + 5 As. H 3 -3 -3
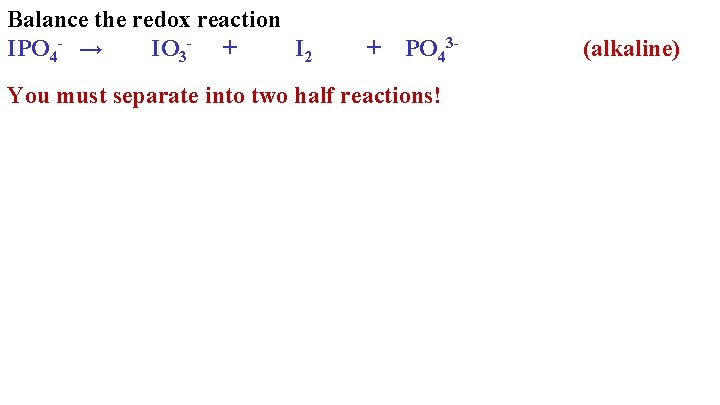
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - You must separate into two half reactions! (alkaline)
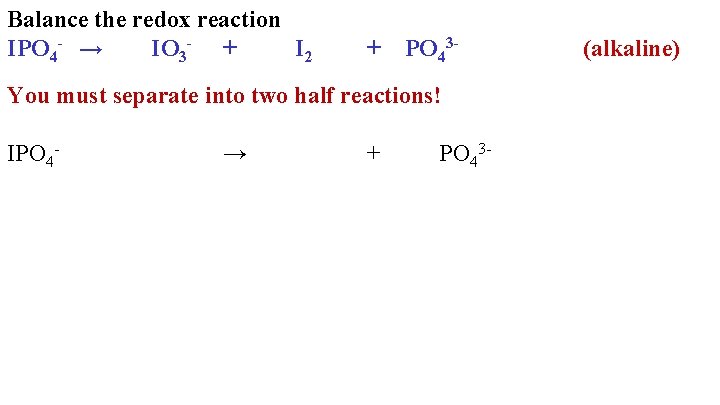
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - You must separate into two half reactions! IPO 4 - → + PO 43 - (alkaline)
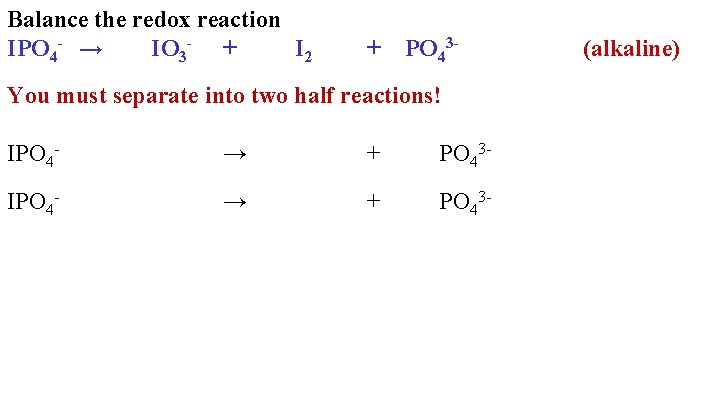
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - You must separate into two half reactions! IPO 4 - → + PO 43 - (alkaline)
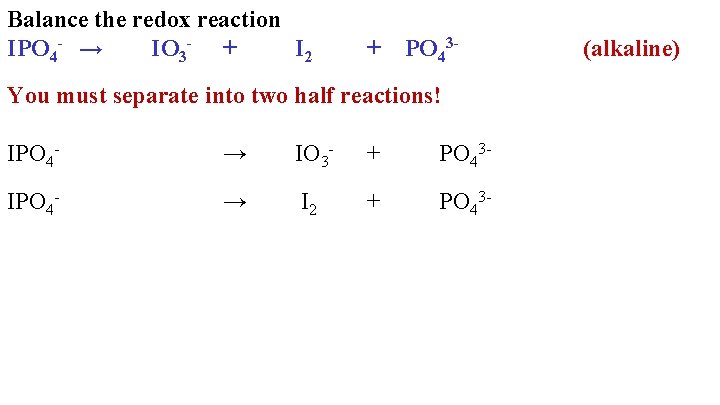
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - You must separate into two half reactions! IPO 4 - → IO 3 - + PO 43 - IPO 4 - → I 2 + PO 43 - (alkaline)
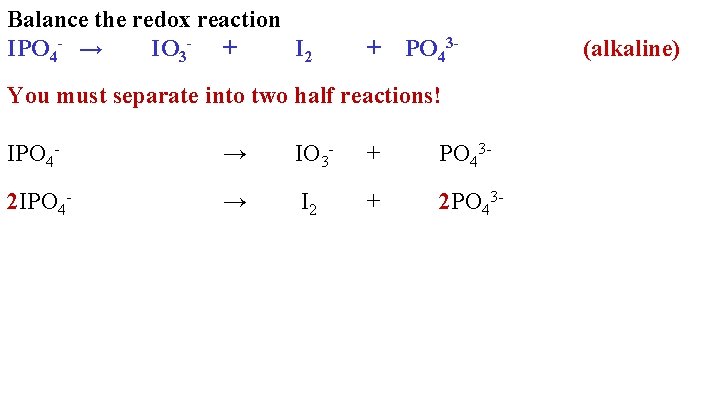
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - You must separate into two half reactions! IPO 4 - → IO 3 - + PO 43 - 2 IPO 4 - → I 2 + 2 PO 43 - (alkaline)
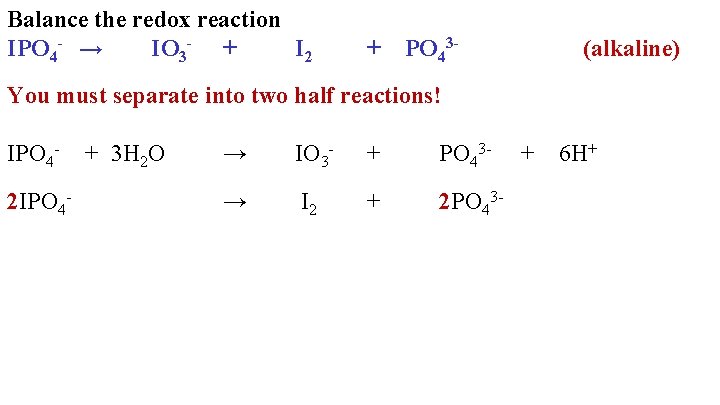
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - (alkaline) You must separate into two half reactions! IPO 42 IPO 4 - + 3 H 2 O → IO 3 - + PO 43 - → I 2 + 2 PO 43 - + 6 H+
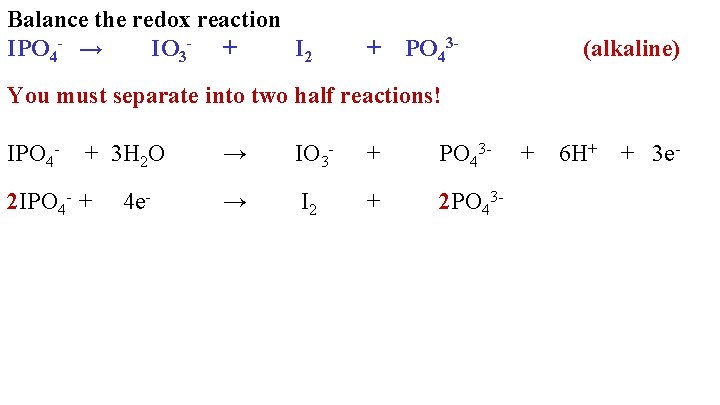
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - (alkaline) You must separate into two half reactions! IPO 4 - + 3 H 2 O 2 IPO 4 - + 4 e- → IO 3 - + PO 43 - → I 2 + 2 PO 43 - + 6 H+ + 3 e-
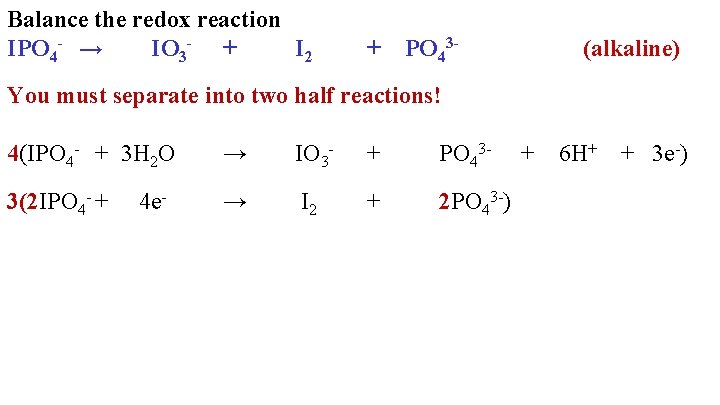
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - (alkaline) You must separate into two half reactions! 4(IPO 4 - + 3 H 2 O → IO 3 - + PO 43 - 3(2 IPO 4 - + → I 2 + 2 PO 43 -) 4 e- + 6 H+ + 3 e-)
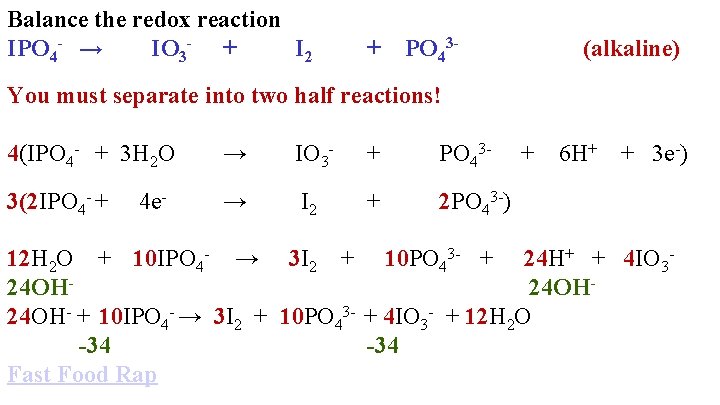
Balance the redox reaction IPO 4 - → IO 3 - + I 2 + PO 43 - (alkaline) You must separate into two half reactions! 4(IPO 4 - + 3 H 2 O → IO 3 - + PO 43 - 3(2 IPO 4 - + → I 2 + 2 PO 43 -) 4 e- + 6 H+ + 3 e-) 12 H 2 O + 10 IPO 4 - → 3 I 2 + 10 PO 43 - + 24 H+ + 4 IO 324 OH- + 10 IPO 4 - → 3 I 2 + 10 PO 43 - + 4 IO 3 - + 12 H 2 O -34 Fast Food Rap
- Slides: 15