Redox Reactions Electrochemical Cells I Balancing Redox Reactions

Redox Reactions & Electrochemical Cells I. Balancing Redox Reactions

I. Balancing Redox Reactions §STEP 1. Split Reaction into 2 Half-Reactions §STEP 2. Balance Elements Other than H & O §STEP 3. Balance O by Inserting H 2 O into eqns. as necessary §STEP 4. Balance H with H+ or H 2 O (see 4 a, 4 b) §STEP 5. Balance Charge by Inserting Electrons as needed §STEP 6. Multiply Each 1/2 Reaction by Factor needed to make no. of Electrons in each 1/2 Reaction Equal §STEP 7. Add Eqns. & Cancel Out Duplicate

I. Balancing Redox Reactions (continued) §STEP 4 a. In ACID: Balance H by Inserting H+, as needed §STEP 4 b. In BASE: Balance H by (i) inserting 1 H 2 O for each missing H & (ii) inserting same no. of OH- on OTHER SIDE OF REACTION as H 2 Os added in (i)

I. Balancing Redox Reactions (continued) §Example §Complete and Balance Following Reaction: Cu. S (s) + NO 3 - (aq) Cu 2+(aq) + SO 42 - (aq) + NO (g) §STEP 1. Split into 2 Half-Reactions a. 1 Cu. S b. 1 NO 3 - Cu 2+ + SO 42 NO

I. Balancing Redox Reactions (continued) §STEP 2. Balance Elements Other than H & O Already O. K. !

I. Balancing Redox Reactions (continued) §STEP 3. Balance O by inserting H 2 O into equations as necessary a. 3 Cu. S + 4 H 2 O b. 3 NO 3 - Cu 2+ + SO 42 - NO + 2 H 2 O

I. Balancing Redox Reactions (continued) §STEP 4. ACIDIC, so Balance H by inserting H+ as needed a 4. Cu. S + 4 H 2 O b 4. NO 3 - + 4 H+ Cu 2+ + SO 42 - + 8 H+ NO + 2 H 2 O

I. Balancing Redox Reactions (continued) §STEP 5. Balance Charge by inserting Electrons, where necessary a 5. Cu. S + 4 H 2 O Cu 2+ + SO 42 - + 8 H+ + 8 e- b 5. NO 3 - + 4 H+ + 3 e- NO + 2 H 2 O

I. Balancing Redox Reactions (continued) §STEP 6. Multiply each Eqn. by factor to make No. of Electrons in Each 1/2 Reaction the Same a 6. Multiply by 3 x 3 Cu. S + 12 H 2 O 3 Cu 2+ + 3 SO 42 - + 24 H+ + 24 eb 6. Multiply by 8 x 8 NO 3 - + 32 H+ + 24 e 8 NO + 16 H+ + 24 e-

I. Balancing Redox Reactions (continued) §STEP 7. Add Eqns. and Cancel Out Duplicated Terms (a 7 + b 7) 8 H+ 3 Cu. S + 12 H 2 O + 8 NO 3 - + 32 H+ + 24 e 3 Cu 2+ + 3 SO 42 - + 24 H+ + 8 NO +16 H 2 O +24 e- 4 H 2 O

I. Balancing Redox Reactions (continued) So, the final, balanced reaction is: 3 Cu. S(s) + 8 NO 3 -(aq) + 8 H+ (aq) 3 Cu 2+(aq) + 3 SO 42 -(aq) + 8 NO(g) + + 4 H 2 O(l)

Checking mass balance and charge balance in Equation §L. H. S §R. H. S. Ø 3 x Cu Ø 3 x S Ø 8 x N Ø 24 x O Ø 8 x H (8 x 1 -) + (8 x H+) = 0 (3 x 2+ )+(3 x 2 - ) = 0

Redox Reactions in Electrochemistry §Two Types of Electrochemical Cells: 1. Galvanic 2. Electrolytic Galvanic Cell - Converts a Chemical Potential Energy into an Electrical Potential to Perform Work Electrolytic Cell- Uses Electrical Energy to

Anode and Cathode in Electrochemistry §ANODE - Where OXIDATION takes place (-e-) §CATHODE - Where REDUCTION takes place (+e-)

Electrochemistry and the Metals Industry §Many Electrochemical Processes are used Commercially for Production of Pure Metals: e. g. Al Manufacture (by electrolysis of Al 2 O 3) Mg Manufacture (by electrolysis of Mg. Cl 2) Na Manufacture (by electrolysis of Na. Cl)
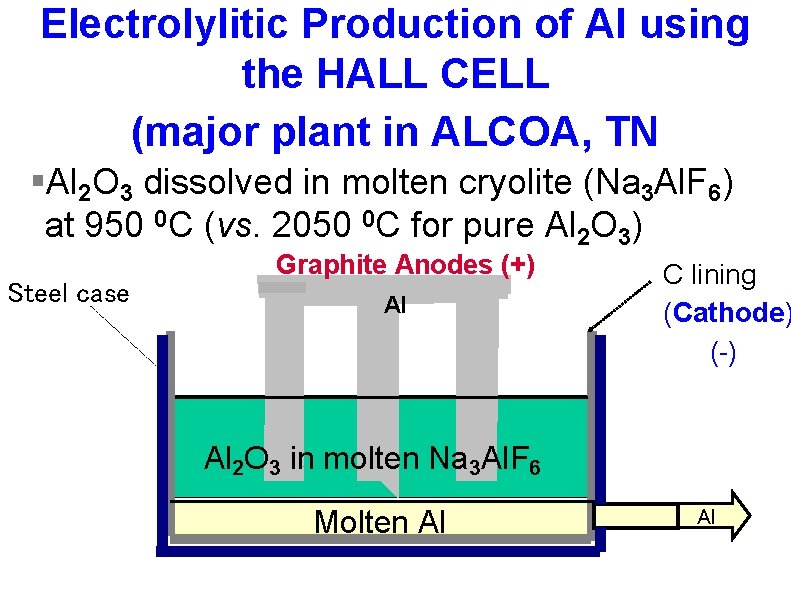
Electrolylitic Production of Al using the HALL CELL (major plant in ALCOA, TN §Al 2 O 3 dissolved in molten cryolite (Na 3 Al. F 6) at 950 0 C (vs. 2050 0 C for pure Al 2 O 3) Steel case Graphite Anodes (+) Al C lining (Cathode) (-) Al 2 O 3 in molten Na 3 Al. F 6 Molten Al Al
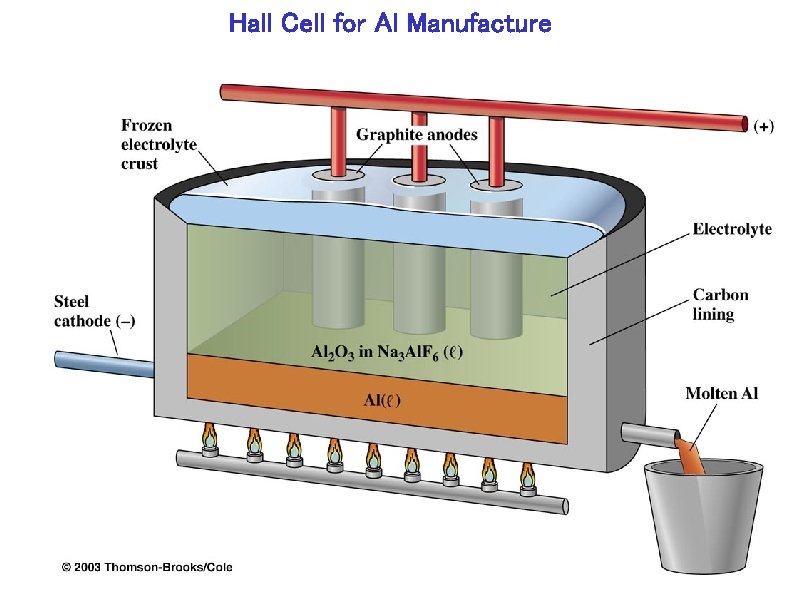
Hall Cell for Al Manufacture

Hall Cell Process Reaction: 2 Al 2 O 3 (sln) + 3 C (s) 4 Al (l) + 3 CO 2 (g) Location of Hall cell plant in E. Tennessee through availability of inexpensive Hydroelectric power. Process uses 50, 000 – 100, 000 A.
- Slides: 18