Electrochemistry Redox Day 3 Utilizing Redox Reactions Balancing

Electrochemistry / Redox Day 3: Utilizing Redox Reactions

Balancing Practice Balance the following redox reaction: Au+3(aq) + I-1(aq) Au(s) + I 2(s) Balance the following redox reaction, which takes place in an acidic solution: Mn. O 4 - + S 2 O 3 -2 Mn+2 + S 4 O 6 -2 Balance the following redox reaction, which takes place in a basic solution: Zn + NO 3 - Zn(OH)4 -2 + NH 3
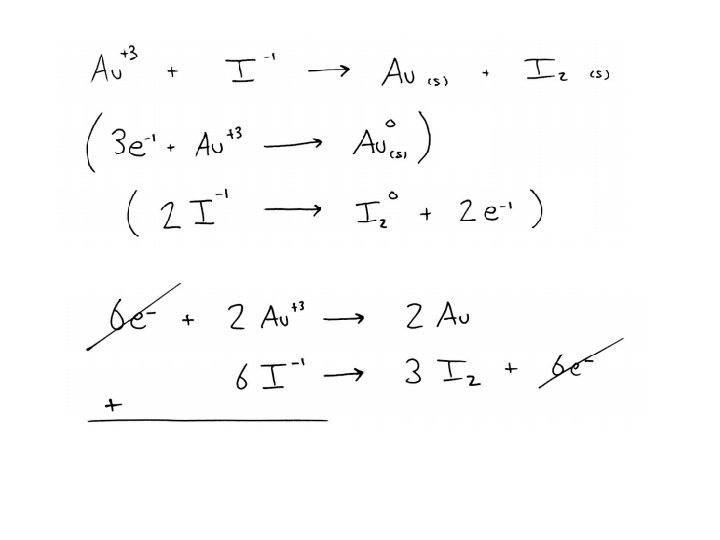
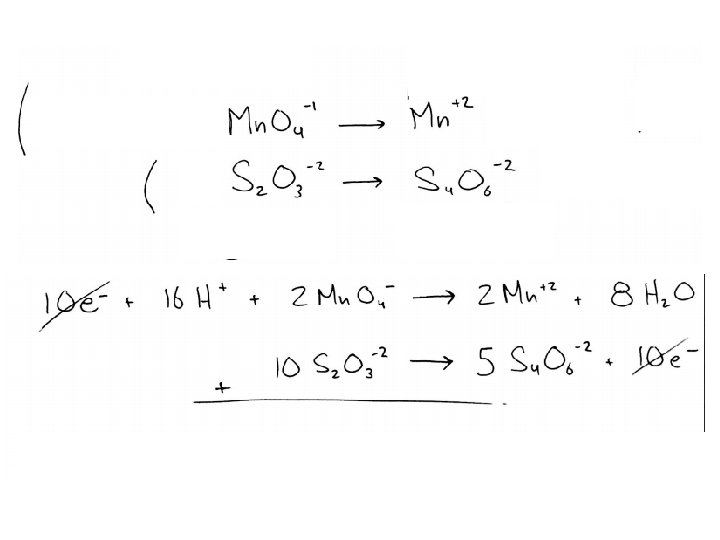
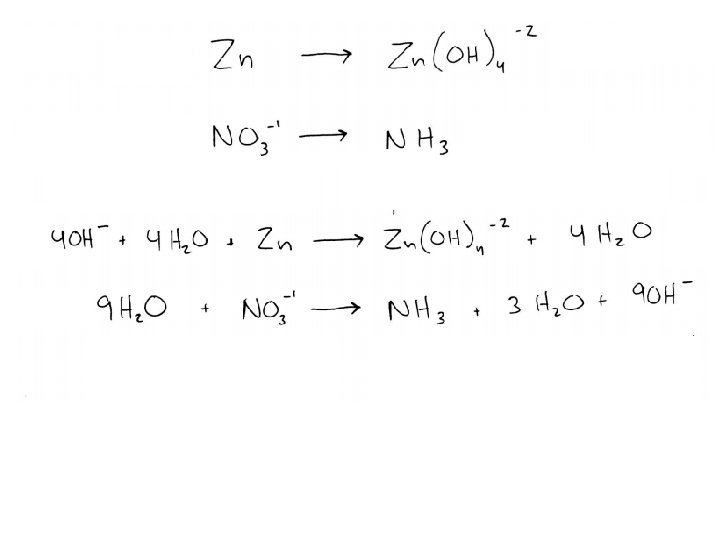
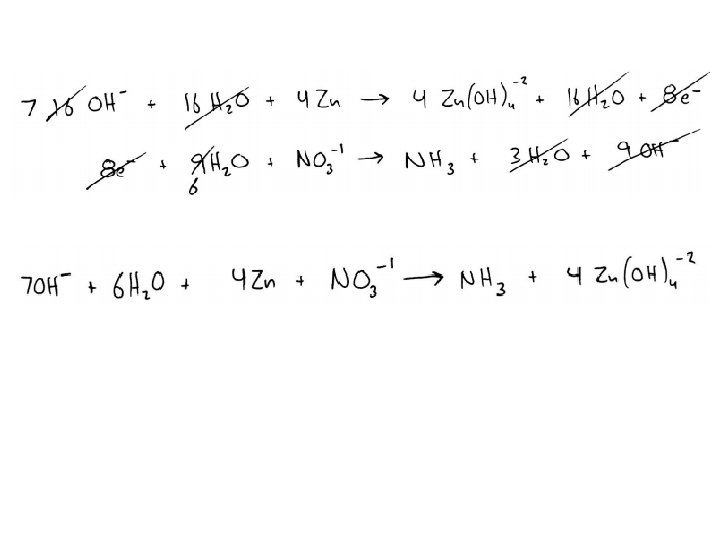
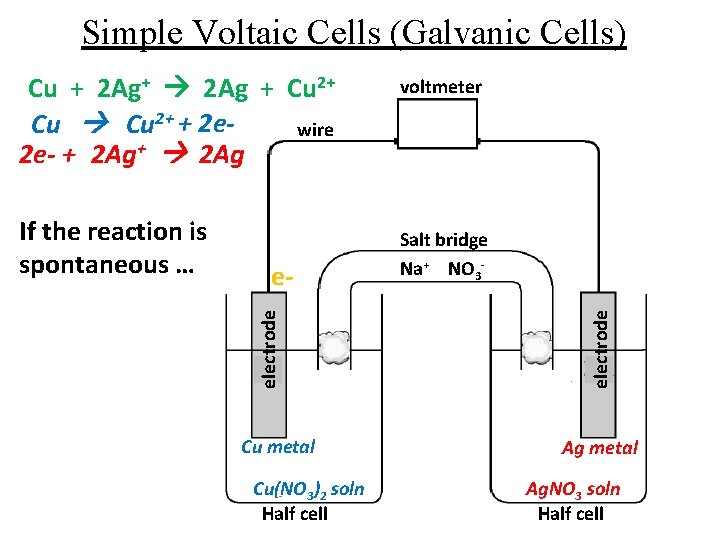
Simple Voltaic Cells (Galvanic Cells) eelectrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
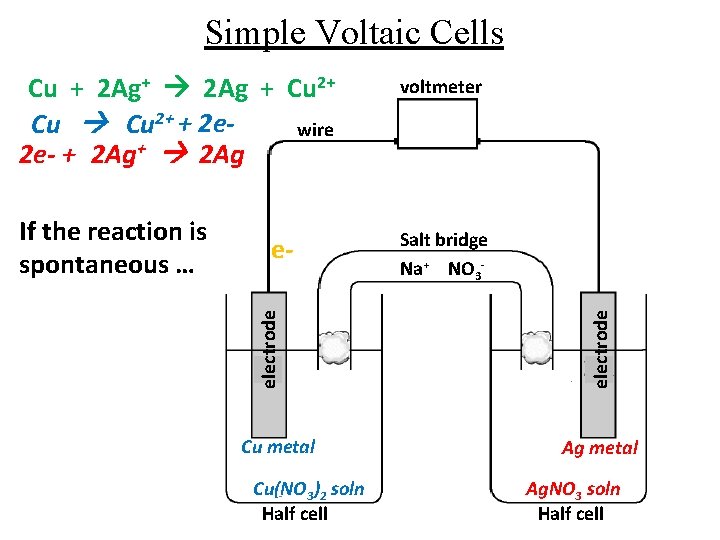
Simple Voltaic Cells eelectrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
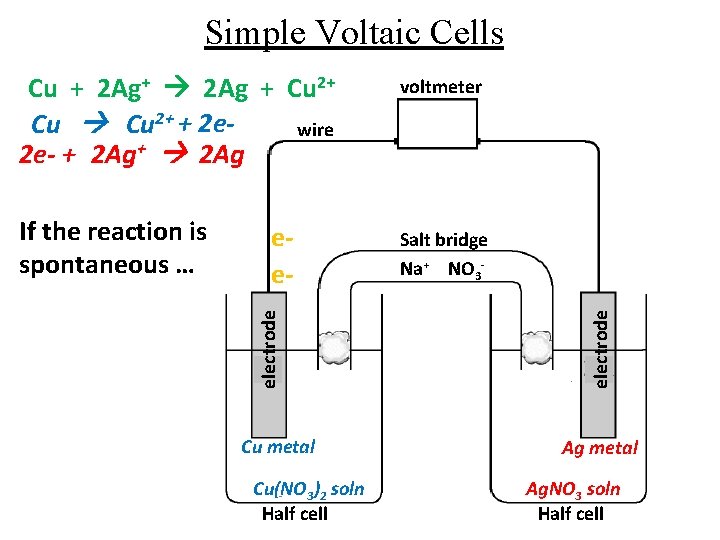
Simple Voltaic Cells eeelectrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
![Simple Voltaic Cells Mass of electrode decreases, [Cu 2+] increases Cu metal Cu(NO 3)2 Simple Voltaic Cells Mass of electrode decreases, [Cu 2+] increases Cu metal Cu(NO 3)2](http://slidetodoc.com/presentation_image_h2/40d6aedd4dbc8a0f2f7a3c393c520edc/image-10.jpg)
Simple Voltaic Cells Mass of electrode decreases, [Cu 2+] increases Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag e. If the reaction is espontaneous … e- Ag metal Ag. NO 3 soln Half cell
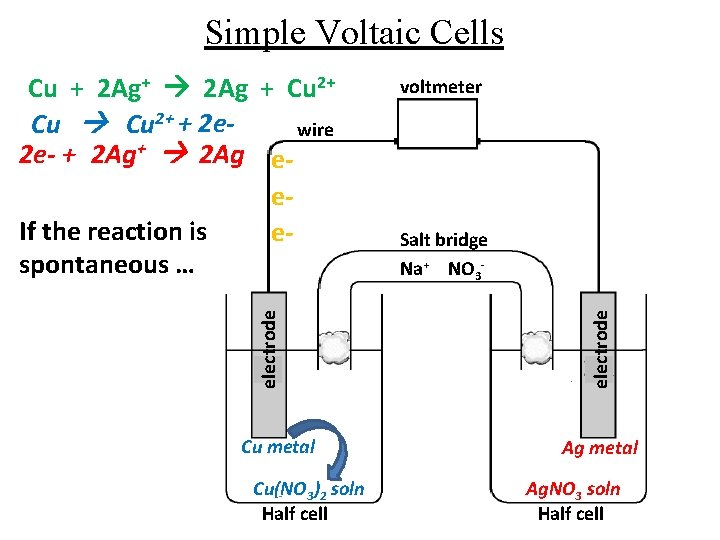
Simple Voltaic Cells Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag ee. If the reaction is espontaneous … Ag metal Ag. NO 3 soln Half cell
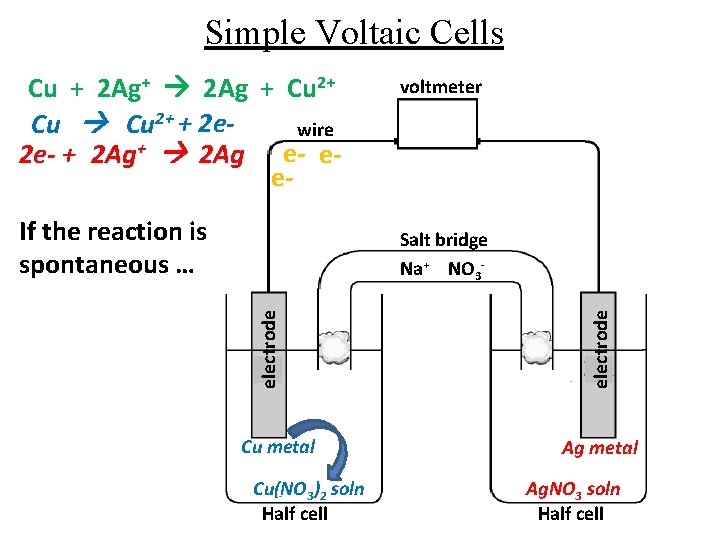
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag e- ee. If the reaction is spontaneous … voltmeter Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
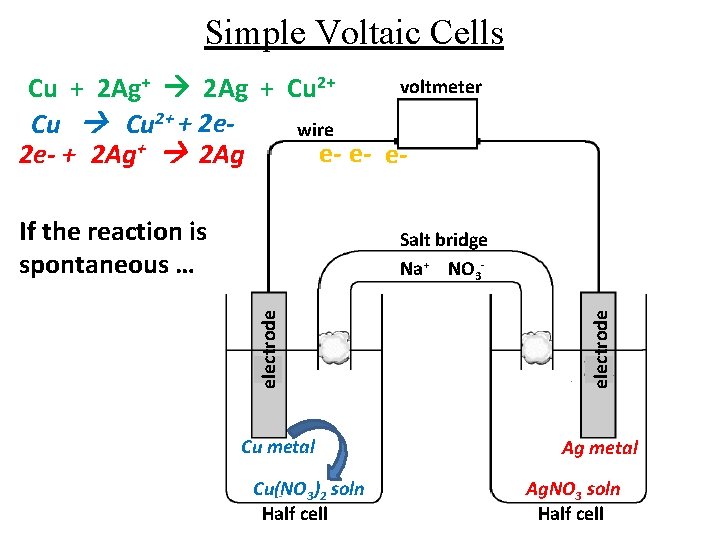
Simple Voltaic Cells voltmeter Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire e- e- e 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
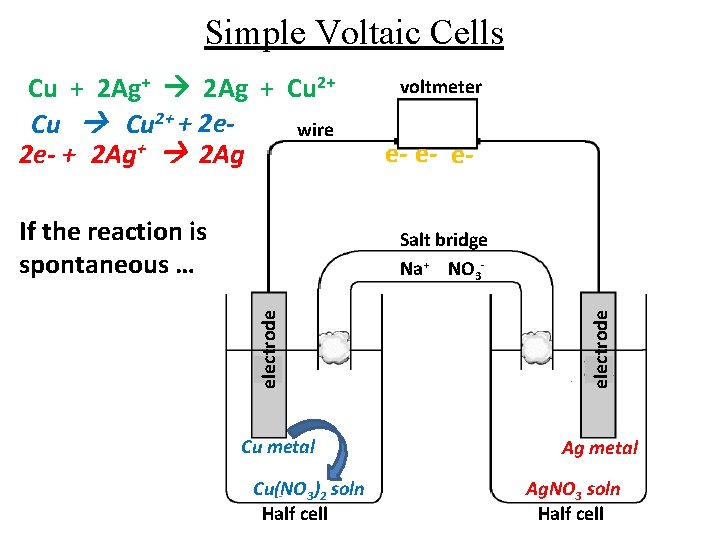
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter e- e- e- Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
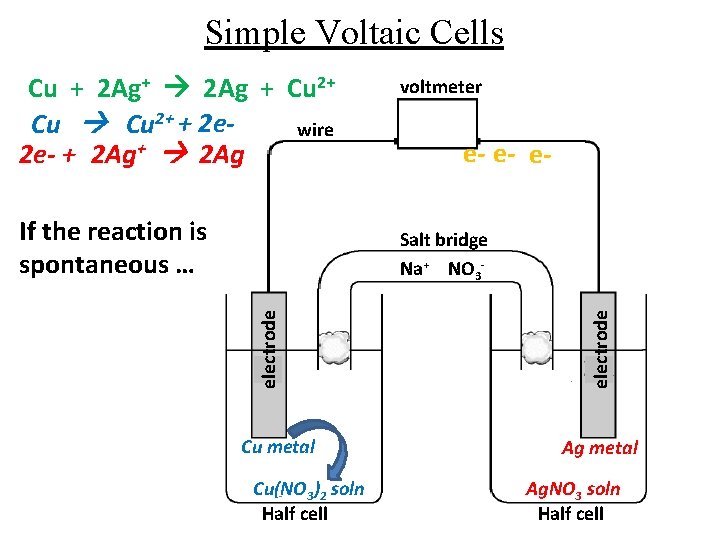
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter e- e- e- Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
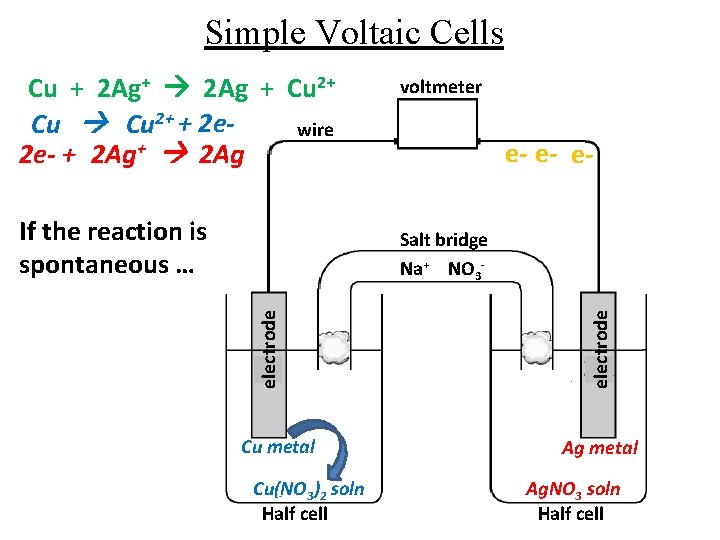
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter e- e- e- Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
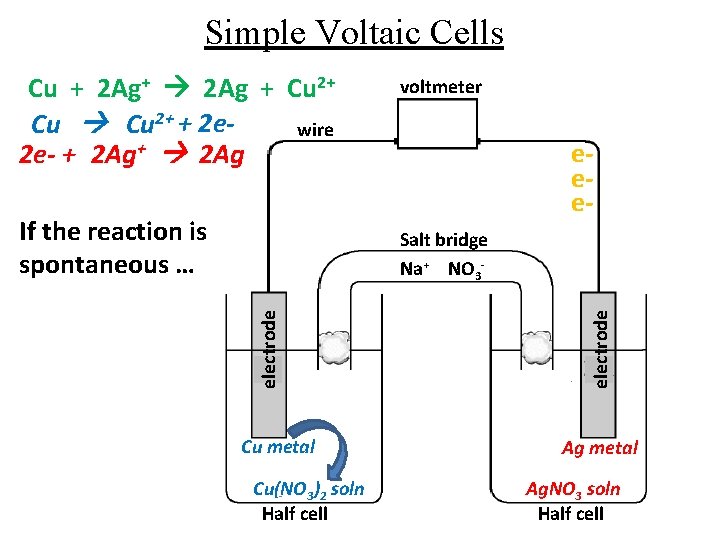
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter eee- Cu metal Cu(NO 3)2 soln Half cell electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
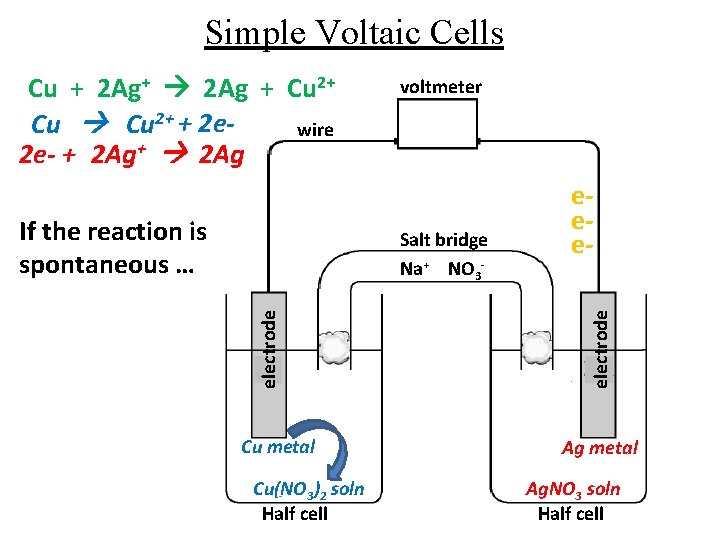
Simple Voltaic Cells If the reaction is spontaneous … voltmeter electrode Salt bridge Na+ NO 3 - Cu metal Cu(NO 3)2 soln Half cell eeeelectrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
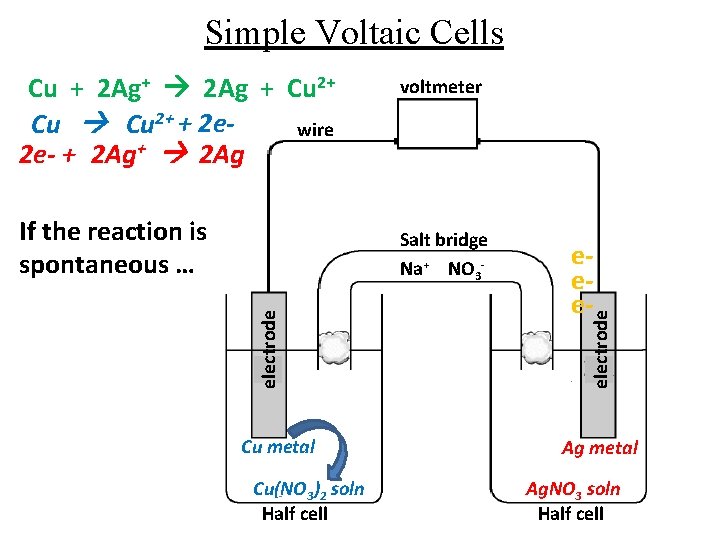
Simple Voltaic Cells If the reaction is spontaneous … voltmeter electrode Salt bridge Na+ NO 3 - Cu metal Cu(NO 3)2 soln Half cell eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
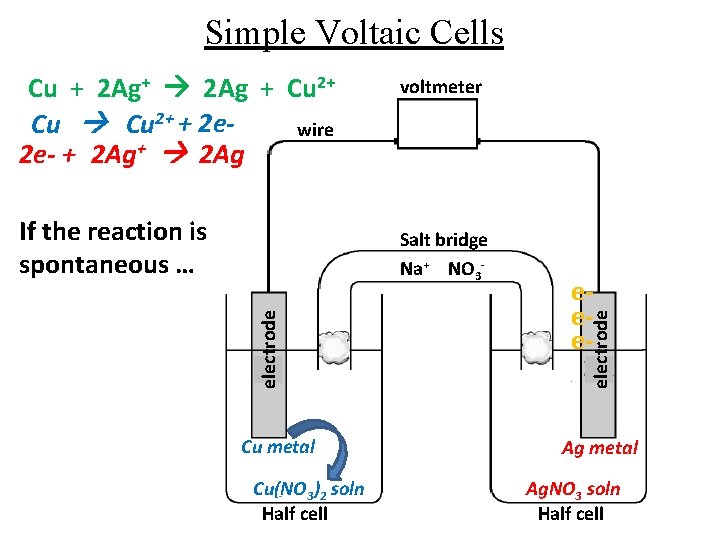
Simple Voltaic Cells If the reaction is spontaneous … voltmeter electrode Salt bridge Na+ NO 3 - Cu metal Cu(NO 3)2 soln Half cell eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
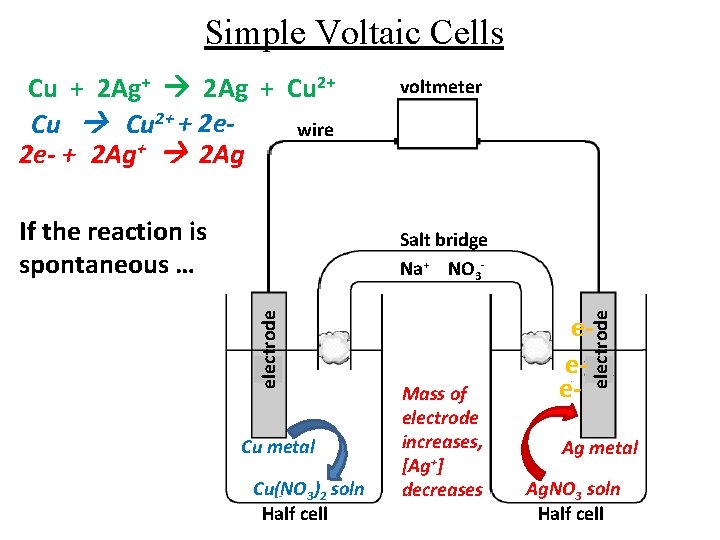
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter Cu metal Cu(NO 3)2 soln Half cell Mass of electrode increases, [Ag+] decreases eee- electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
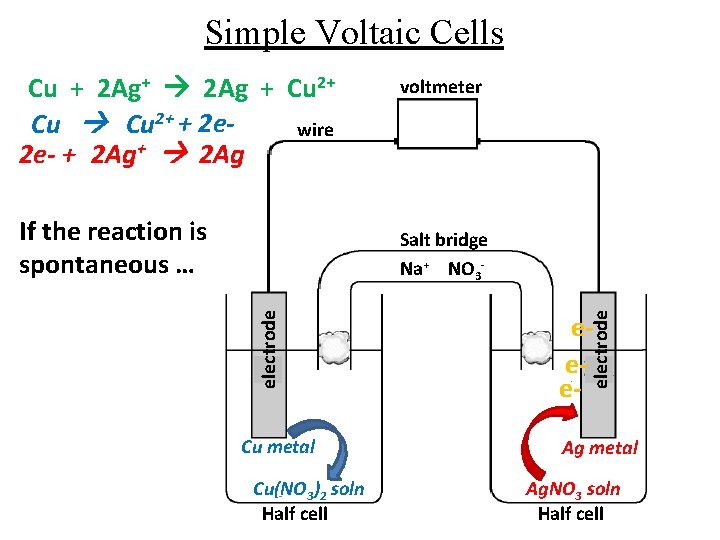
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag If the reaction is spontaneous … voltmeter Cu metal Cu(NO 3)2 soln Half cell eee- electrode Salt bridge Na+ NO 3 - Ag metal Ag. NO 3 soln Half cell
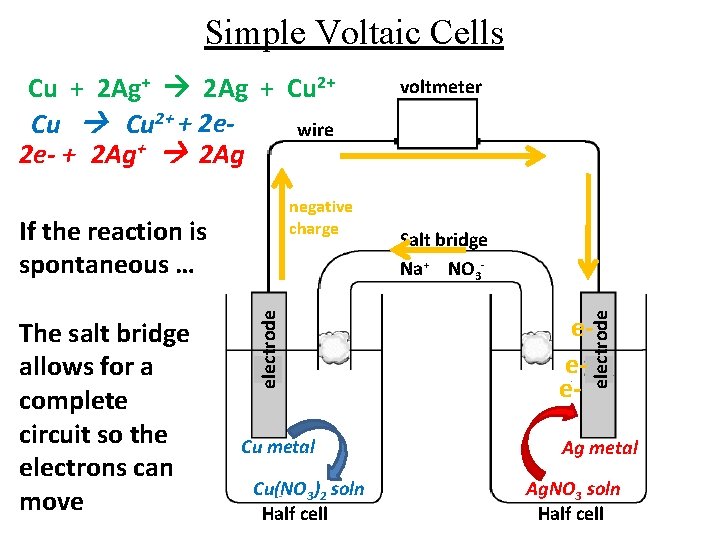
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 - eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
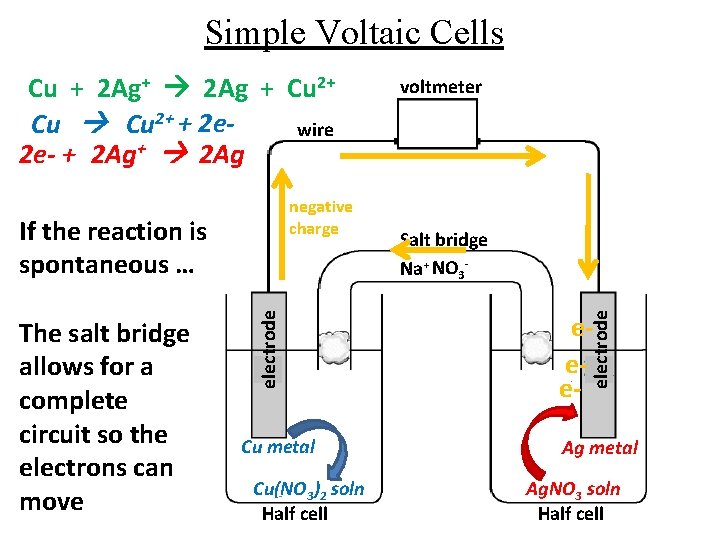
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ NO 3 - eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
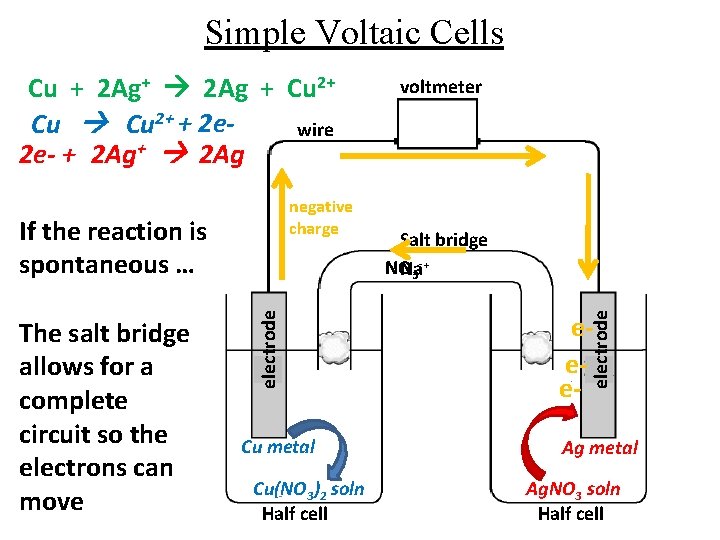
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge -+ NO Na 3 eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
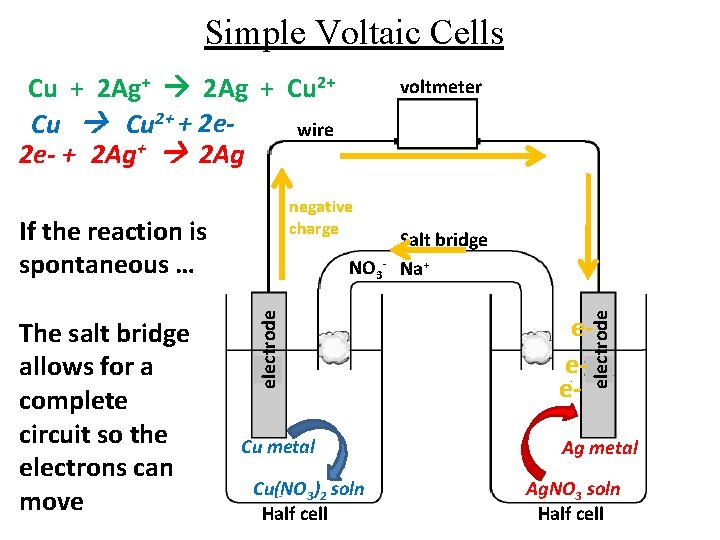
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag electrode NO 3 - Cu metal Cu(NO 3)2 soln Half cell Salt bridge Na+ eee- electrode negative charge If the reaction is spontaneous … The salt bridge allows for a complete circuit so the electrons can move voltmeter Ag metal Ag. NO 3 soln Half cell
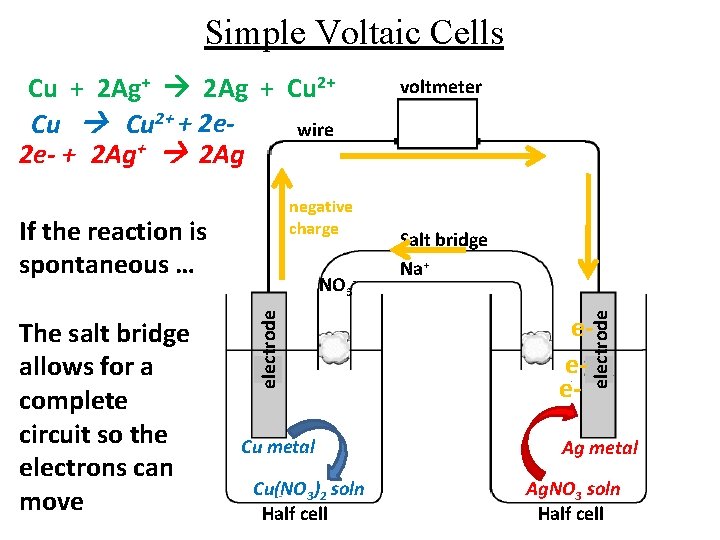
Simple Voltaic Cells negative charge If the reaction is spontaneous … electrode The salt bridge allows for a complete circuit so the electrons can move NO 3 - Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
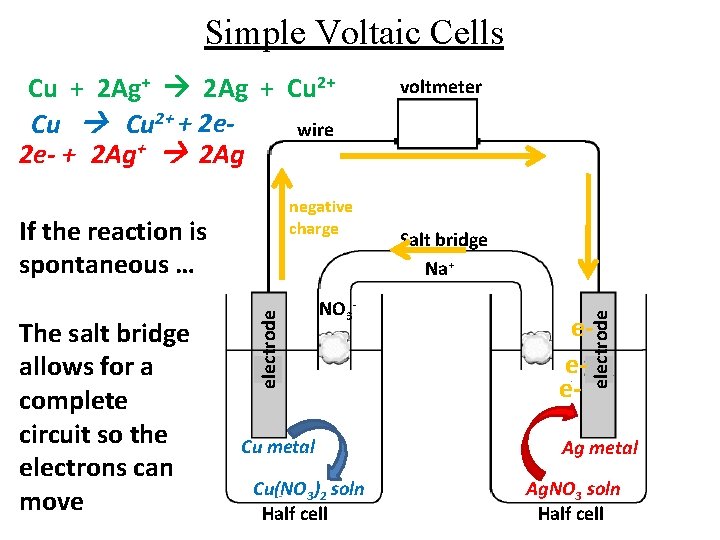
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … NO 3 - Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
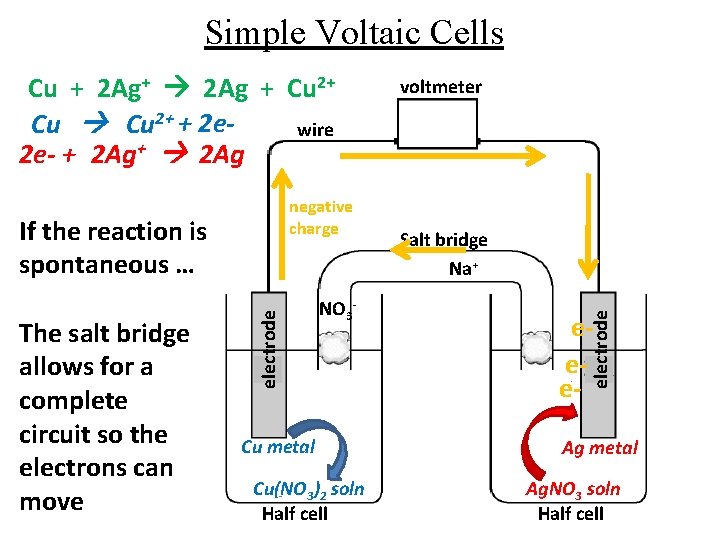
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … NO 3 - Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
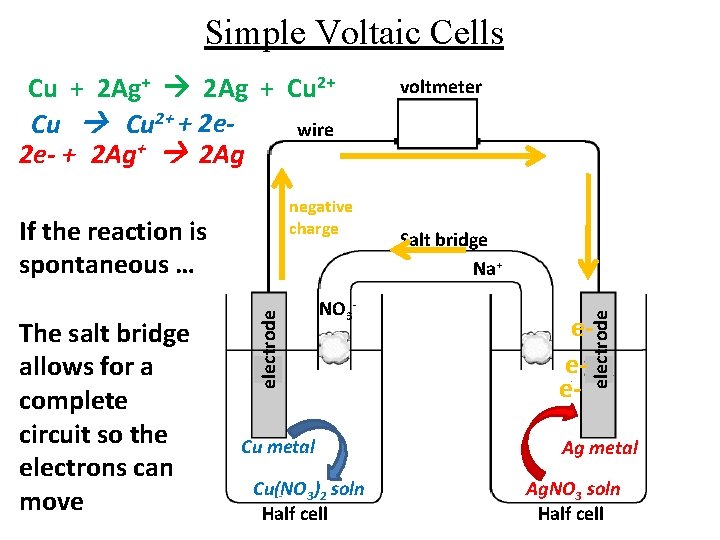
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … NO 3 - Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
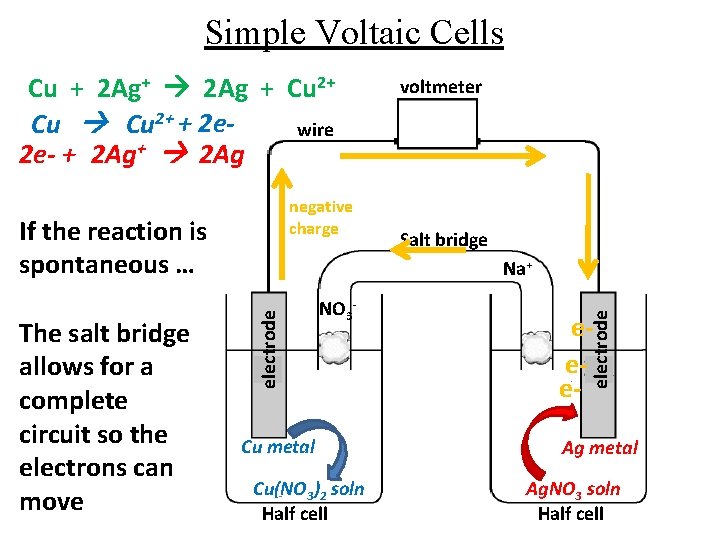
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag negative charge If the reaction is spontaneous … Salt bridge Na+ NO 3 - Cu metal Cu(NO 3)2 soln Half cell eee- electrode The salt bridge allows for a complete circuit so the electrons can move voltmeter Ag metal Ag. NO 3 soln Half cell
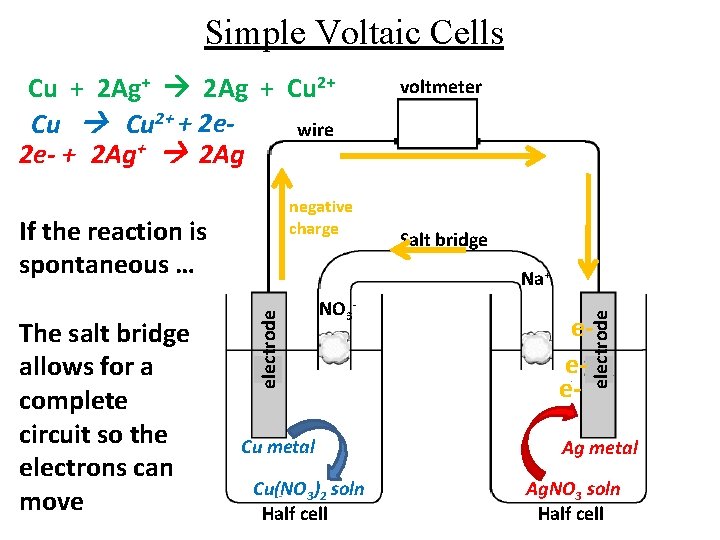
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag negative charge If the reaction is spontaneous … Salt bridge Na+ NO 3 - Cu metal Cu(NO 3)2 soln Half cell eee- electrode The salt bridge allows for a complete circuit so the electrons can move voltmeter Ag metal Ag. NO 3 soln Half cell
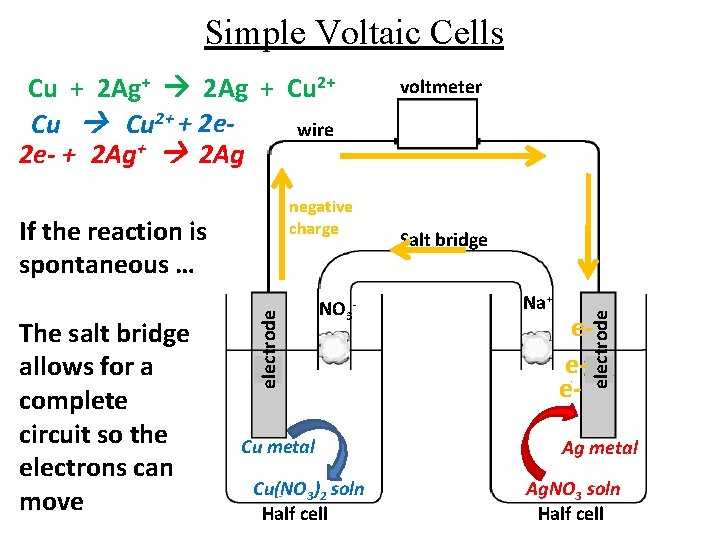
Simple Voltaic Cells negative charge The salt bridge allows for a complete circuit so the electrons can move electrode If the reaction is spontaneous … NO 3 - Cu metal Cu(NO 3)2 soln Half cell voltmeter Salt bridge Na+ eee- electrode Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 ewire 2 e- + 2 Ag+ 2 Ag Ag metal Ag. NO 3 soln Half cell
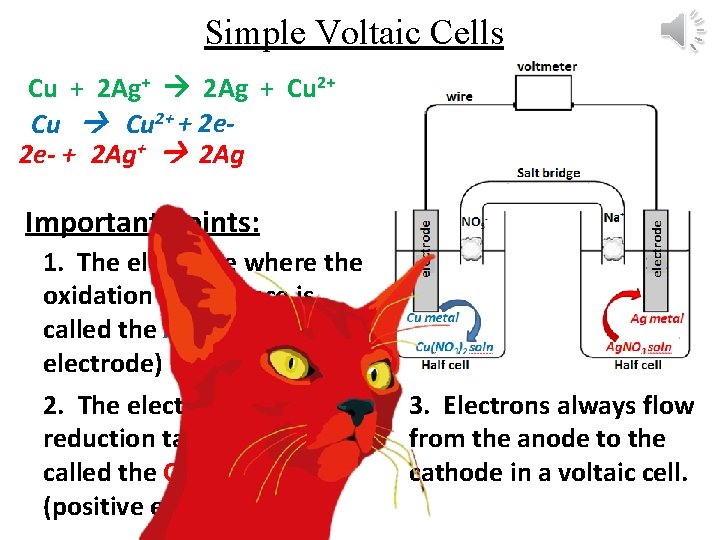
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 e 2 e- + 2 Ag+ 2 Ag Important Points: 1. The electrode where the oxidation takes place is called the ANODE (negative electrode) 2. The electrode where the reduction takes place is called the CATHODE (positive electrode) 3. Electrons always flow from the anode to the cathode in a voltaic cell.
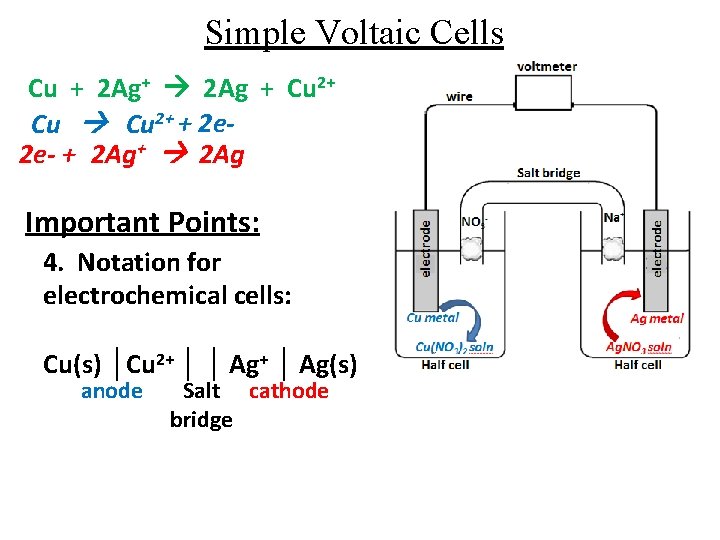
Simple Voltaic Cells Cu + 2 Ag+ 2 Ag + Cu 2+ + 2 e 2 e- + 2 Ag+ 2 Ag Important Points: 4. Notation for electrochemical cells: Cu(s) │Cu 2+ │ │ Ag+ │ Ag(s) anode Salt cathode bridge
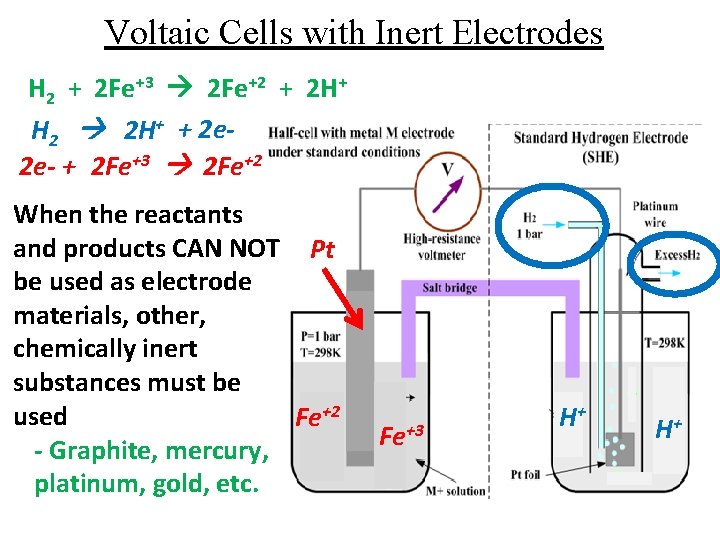
Voltaic Cells with Inert Electrodes H 2 + 2 Fe+3 2 Fe+2 + 2 H+ H 2 2 H+ + 2 e 2 e- + 2 Fe+3 2 Fe+2 When the reactants and products CAN NOT Pt be used as electrode materials, other, chemically inert substances must be used Fe+2 - Graphite, mercury, platinum, gold, etc. Fe+3 H+ H+
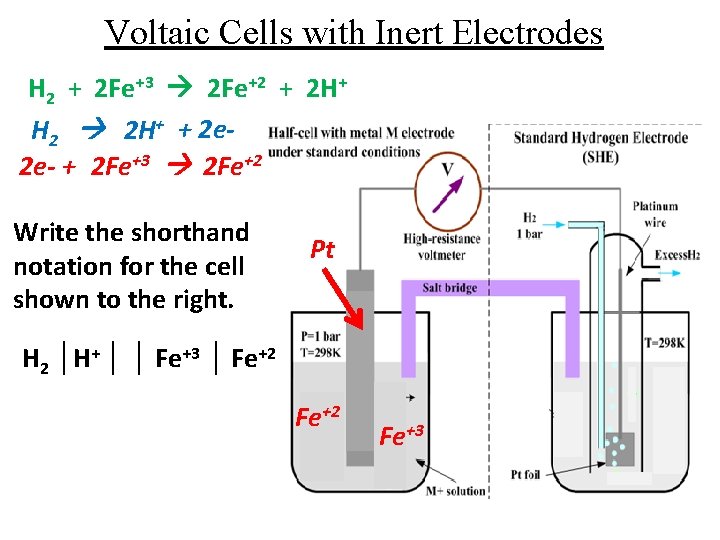
Voltaic Cells with Inert Electrodes H 2 + 2 Fe+3 2 Fe+2 + 2 H+ H 2 2 H+ + 2 e 2 e- + 2 Fe+3 2 Fe+2 Write the shorthand notation for the cell shown to the right. Pt H 2 │H+ │ │ Fe+3 │ Fe+2 Fe+3

Voltaic Cells with Inert Electrodes Suggested homework for this weekend: 1. 1 -2 hours of AP Test Prep 2. Practice several redox balancing problems (regular, acidic and basic solution problems)
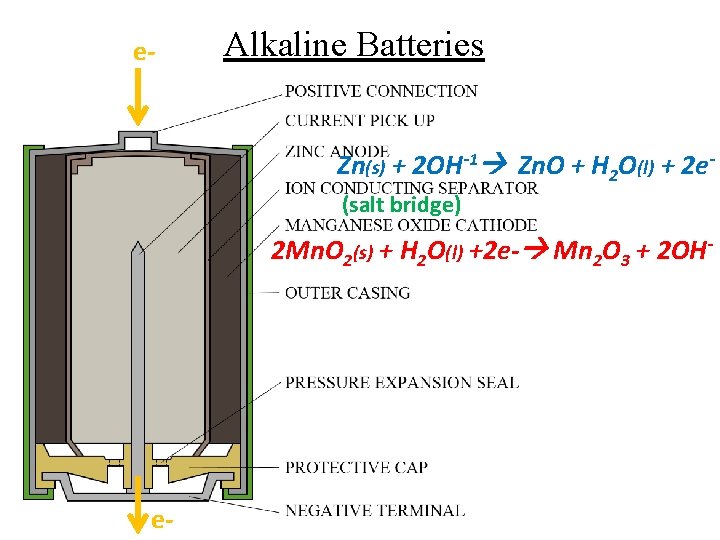
e- Alkaline Batteries Zn(s) + 2 OH-1 Zn. O + H 2 O(l) + 2 e(salt bridge) 2 Mn. O 2(s) + H 2 O(l) +2 e- Mn 2 O 3 + 2 OH- e-
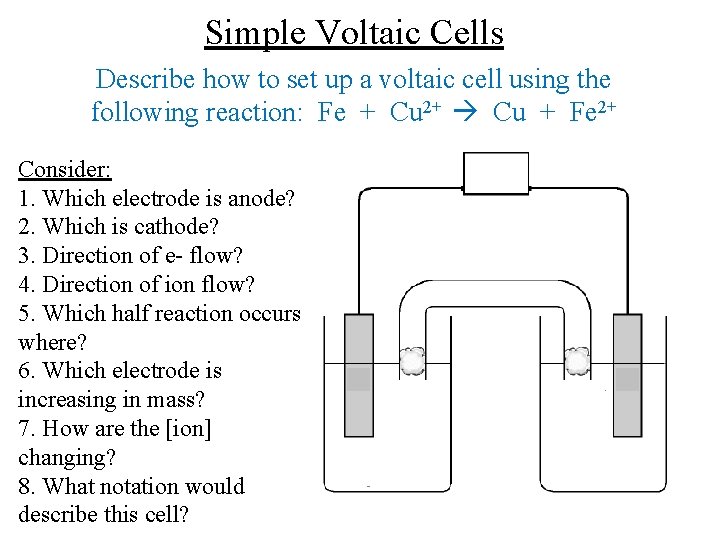
Simple Voltaic Cells Describe how to set up a voltaic cell using the following reaction: Fe + Cu 2+ Cu + Fe 2+ Consider: 1. Which electrode is anode? 2. Which is cathode? 3. Direction of e- flow? 4. Direction of ion flow? 5. Which half reaction occurs where? 6. Which electrode is increasing in mass? 7. How are the [ion] changing? 8. What notation would describe this cell?



- Slides: 43