Balancing Redox Equations ReductionOxidation Redox A new iron

Balancing Redox Equations Reduction/Oxidation = Redox A new iron bolt (Fe) A rusted (oxidized) bolt (rust is Fe 2 O 3) Iron (Fe) rusts (is oxidized) when it reacts with oxygen (O 2) in the air. 2 Fe + 3 O 2 Fe 2 O 3 [iron(III) oxide] 1

Redox Equations Rusting is a an example of a redox reaction. 2 Fe + 3 O 2 Fe 2 O 3 [iron(III) oxide] In redox reactions, one chemical is oxidized (e. g. , Fe is oxidized) and another chemical is reduced (e. g. , O 2 is reduced). Oxidation and reduction always occur together. One cannot occur without the other. In this reaction, when iron is oxidized, it gains oxygen. This definition of oxidation (‘gain of oxygen’), while correct in this case, is inadequate, because many redox reactions do not involve oxygen. A better definition follows, but do not disregard the example of rusting. It is something we all have observed and will help you understand remember the broader definitions. 2

Redox Equations 0 0 2 Fe + 3 O 2 +3 -2 Fe 2 O 3 lose 3 e’s, LEO [iron(III) oxide] gain 2 e’s, GER What are the oxidation numbers of iron and oxygen in the rust equation? Definitions to Memorize: Loss of Electrons = Oxidation LEO the Lion says GER Gain of Electrons = Reduction To do redox chemistry, you must be able to determine which atoms have lost e’s (have been oxidized) and which atoms have gained e’s (have been reduced). Thus, iron has been oxidized by oxygen. Oxygen is an ‘oxidizing agent’. and oxygen has been reduced by iron. Iron is a ‘reducing agent’. 3

Balancing Redox Equations by the Half-Reaction Method The following rules describe the most versatile method for balancing redox equations. . . 1. Break the eqn. into two ½-reactions, one for oxidation (oxid. ) and one for reduction (red. ) 2. Balance all atoms in each ½-reaction other than H and O 3. Balance O and H as follows. . . a) first balance O by adding H 2 O, b) then balance H’s by adding H+ c) finally, balance the charges by adding sufficient electrons e-'s are always added to the right side of the oxid. ½-reaction because oxid. = loss of e-'s are always added to the left side of the red. ½-reaction because red. = gain of e-'s 4. Multiply each ½-reaction by a coefficient so that the same number of e-'s are transferred in each ½-reaction. The number of e’s gained must equal the number of e’s lost. 5. Add the ½-reactions and cancel common terms (e. g. , H 2 O and/or H+) from each side. 6. If the reaction is carried out in neutral or basic solution, add enough OH- to neutralize all H+ (combine them to make H 2 O) and be sure to add the same quantity of OH- to both sides to keep mass and charge balanced. 7. Cancel common terms (H 2 O and/or OH-) from each side and check for mass and charge balance 4
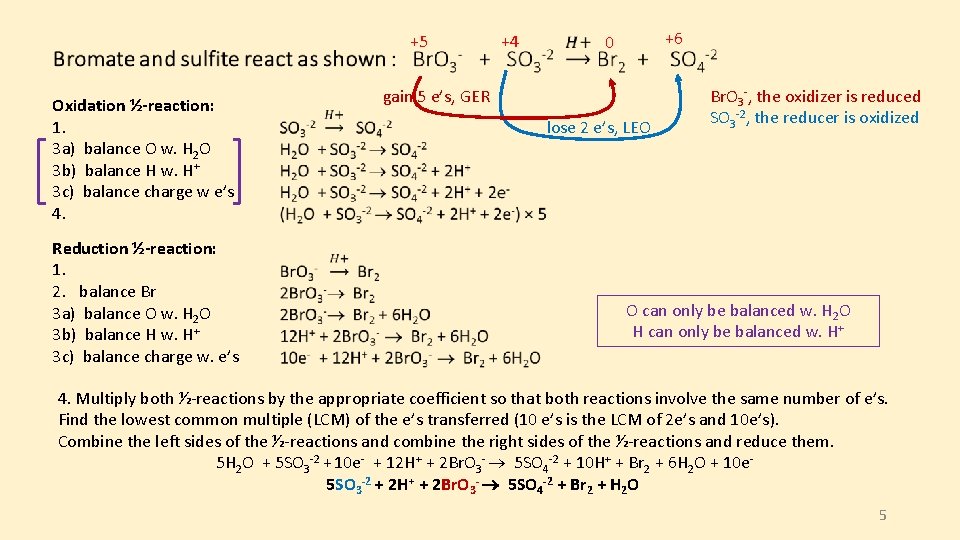
Oxidation ½-reaction: 1. 3 a) balance O w. H 2 O 3 b) balance H w. H+ 3 c) balance charge w e’s 4. Reduction ½-reaction: 1. 2. balance Br 3 a) balance O w. H 2 O 3 b) balance H w. H+ 3 c) balance charge w. e’s +5 +4 +6 0 gain 5 e’s, GER lose 2 e’s, LEO Br. O 3 -, the oxidizer is reduced SO 3 -2, the reducer is oxidized O can only be balanced w. H 2 O H can only be balanced w. H+ 4. Multiply both ½-reactions by the appropriate coefficient so that both reactions involve the same number of e’s. Find the lowest common multiple (LCM) of the e’s transferred (10 e’s is the LCM of 2 e’s and 10 e’s). Combine the left sides of the ½-reactions and combine the right sides of the ½-reactions and reduce them. 5 H 2 O + 5 SO 3 -2 + 10 e- + 12 H+ + 2 Br. O 3 - 5 SO 4 -2 + 10 H+ + Br 2 + 6 H 2 O + 10 e 5 SO 3 -2 + 2 H+ + 2 Br. O 3 - 5 SO 4 -2 + Br 2 + H 2 O 5

+7 Oxidation ½-reaction: 1. 3 b) balance H w. H+ 3 c) balance charge w e’s 6. neutralize H+ w. OHReduction ½-reaction: 1. 3 a) balance O w. H 2 O 3 b) balance H w. H+ 3 c) balance charge w. e’s 6. neutralize H+ w. OH 7. cancel H 2 O gain 3 e’s, GER -1 +4 0 lose 1 e’s, LEO e’s are always on the right side of an oxidation ½-reaction Mn. O 4 -, the oxidizer is reduced H 2 O 2, the reducer is oxidized e’s are always on the left side of a reduction ½-reaction Multiply both ½-reactions by the appropriate coefficient so that both reactions involve the same number of e’s. Find the lowest common multiple (LCM) of the e’s transferred (6 e’s is the LCM of 2 e’s and 3 e’s) 6 OH- + 3 H O + 6 e- + 4 H O + 2 Mn. O - 2 Mn. O + 8 OH- + 3 O + 6 H O + 6 e 2 2 2 4 2 2 2 3 H 2 O 2 + 2 Mn. O 4 2 Mn. O 2 + 2 OH + 3 O 2 + 2 H 2 O 6
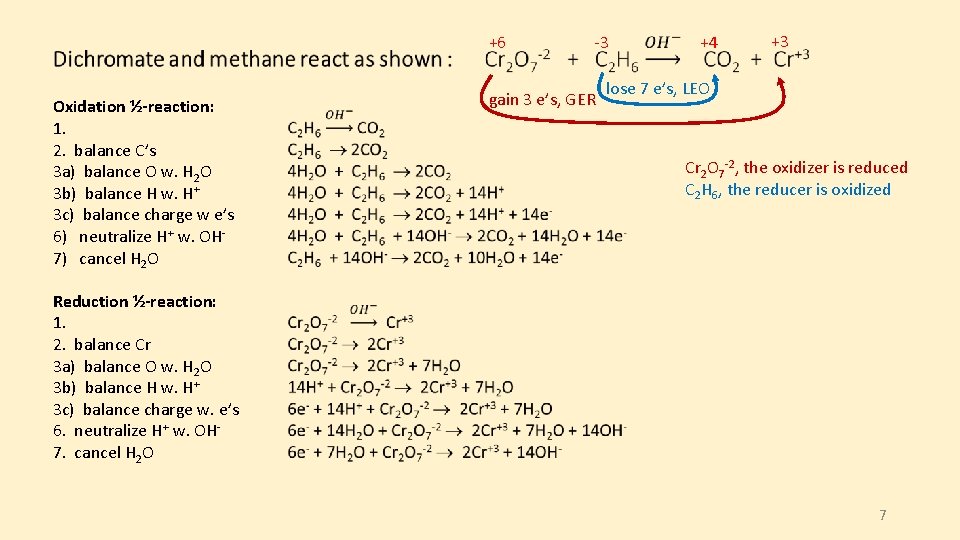
+6 Oxidation ½-reaction: 1. 2. balance C’s 3 a) balance O w. H 2 O 3 b) balance H w. H+ 3 c) balance charge w e’s 6) neutralize H+ w. OH 7) cancel H 2 O gain 3 e’s, GER -3 +4 +3 lose 7 e’s, LEO Cr 2 O 7 -2, the oxidizer is reduced C 2 H 6, the reducer is oxidized Reduction ½-reaction: 1. 2. balance Cr 3 a) balance O w. H 2 O 3 b) balance H w. H+ 3 c) balance charge w. e’s 6. neutralize H+ w. OH 7. cancel H 2 O 7

+6 -3 gain 3 e’s, GER Oxidation ½-reaction: C 2 H 6 + 14 OH- 2 CO 2 + 10 H 2 O + 14 e- +4 +3 lose 7 e’s, LEO Reduction ½-reaction: 7 H 2 O + Cr 2 O 7 -2 + 6 e- 2 Cr+3 + 14 OH- Multiply both ½-reactions by the appropriate coefficient so that both reactions involve the same number of e’s. Find the lowest common multiple (LCM) of the e’s transferred ( 42 is the LCM’s of 14 e’s and 6 e’s) (C 2 H 6 + 14 OH- 2 CO 2 + 10 H 2 O + 14 e-) × 3 (7 H 2 O + Cr 2 O 7 -2 + 6 e- 2 Cr+3 + 14 OH-) × 7 Combine the two ½-reactions: 3 C 2 H 6 + 42 OH- 49 H 2 O + 7 Cr 2 O 7 -2 + 42 e- 14 Cr+3 + 98 OH- + 6 CO 2 + 30 H 2 O + 42 e 3 C 2 H 6 + 19 H 2 O + 7 Cr 2 O 7 -2 14 Cr+3 + 56 OH- + 6 CO 2 8

Oxidation Number Method for Balancing Redox Equations +5 +4 each Br atom in Br. O 3 - gains 5 e’s. each S atom in SO 3 -2 loses 2 e’s, 10 e’s is the LCM 5 e’s × 2 Br. O 3 - = 10 e’s gained 2 e’s × 5 SO 3 -2 = 10 e’s lost 2 Br. O 3 - + 5 SO 3 -2 Br 2 + 5 SO 4 -2 +6 0 gain 5 e’s, GER lose 2 e’s, LEO +7 -1 +4 gain 3 e’s, GER +6 0 lose 1 e’s, LEO -3 gain 3 e’s, GER +4 lose 7 e’s, LEO +3 each Mn atom in Mn. O 4 - gains 3 e’s. each O atom in H 2 O 2 loses 1 e’s or each H 2 O 2 lose 2 e’s, 6 e’s is the LCM 3 e’s × 2 Mn. O 4 - = 6 e’s gained 2 e’s × 3 H 2 O 2 = 6 e’s lost 2 Mn. O 4 - + 3 H 2 O 2 2 Mn. O 2 + 3 O 2 each Cr atom in Cr 2 O 7 -2 gains 3 e’s or each Cr 2 O 7 -2 gains 6 e’s each C atom in C 2 H 6 loses 7 e’s or each C 2 H 6 lose 14 e’s, 42 e’s is the LCM 6 e’s × 7 Cr 2 O 7 -2 = 42 e’s gained 14 e’s × 3 C 2 H 6 = 42 e’s lost 7 Cr 2 O 7 -2+ 3 C 2 H 6 14 Cr+3 + 6 CO 2 9
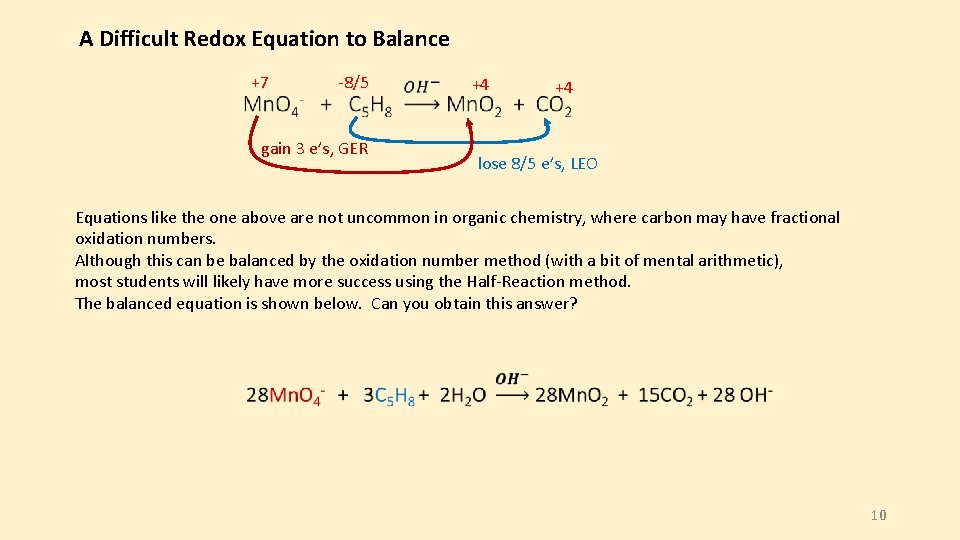
A Difficult Redox Equation to Balance +7 -8/5 gain 3 e’s, GER +4 +4 lose 8/5 e’s, LEO Equations like the one above are not uncommon in organic chemistry, where carbon may have fractional oxidation numbers. Although this can be balanced by the oxidation number method (with a bit of mental arithmetic), most students will likely have more success using the Half-Reaction method. The balanced equation is shown below. Can you obtain this answer? 10

Practice is the best teacher 11
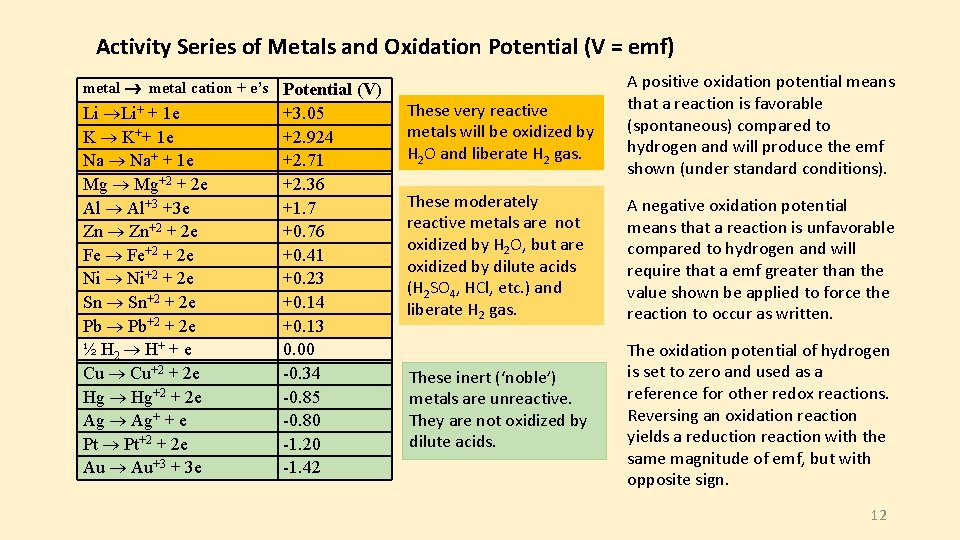
Activity Series of Metals and Oxidation Potential (V = emf) metal cation + e’s Potential (V) Li Li+ + 1 e K K++ 1 e Na Na+ + 1 e Mg Mg+2 + 2 e Al Al+3 +3 e Zn Zn+2 + 2 e Fe Fe+2 + 2 e Ni Ni+2 + 2 e Sn Sn+2 + 2 e Pb Pb+2 + 2 e ½ H 2 H+ + e Cu Cu+2 + 2 e Hg Hg+2 + 2 e Ag Ag+ + e Pt Pt+2 + 2 e Au Au+3 + 3 e +3. 05 +2. 924 +2. 71 +2. 36 +1. 7 +0. 76 +0. 41 +0. 23 +0. 14 +0. 13 0. 00 -0. 34 -0. 85 -0. 80 -1. 20 -1. 42 These very reactive metals will be oxidized by H 2 O and liberate H 2 gas. A positive oxidation potential means that a reaction is favorable (spontaneous) compared to hydrogen and will produce the emf shown (under standard conditions). These moderately reactive metals are not oxidized by H 2 O, but are oxidized by dilute acids (H 2 SO 4, HCl, etc. ) and liberate H 2 gas. A negative oxidation potential means that a reaction is unfavorable compared to hydrogen and will require that a emf greater than the value shown be applied to force the reaction to occur as written. These inert (‘noble’) metals are unreactive. They are not oxidized by dilute acids. The oxidation potential of hydrogen is set to zero and used as a reference for other redox reactions. Reversing an oxidation reaction yields a reduction reaction with the same magnitude of emf, but with opposite sign. 12
- Slides: 12