Chemical Reactions and Enzymes Chemical Reactions Chemical Reaction











- Slides: 16

Chemical Reactions and Enzymes

Chemical Reactions Chemical Reaction – the process that changes one set of chemicals into another. �Chemical reactions involve the breaking of bonds in the reactants and the formation of new bonds in the products CO 2 + H 2 O → H 2 CO 3 Carbon dioxide and water form carbonic acid

Chemical Reactions �Reactants – elements or compounds that you start with, found on the left hand side of the reaction arrow. �Products – elements or compounds produced from the reactants, found on the right hand side of the reaction arrow. CO 2 + H 2 O H 2 CO 3 Reactants Product

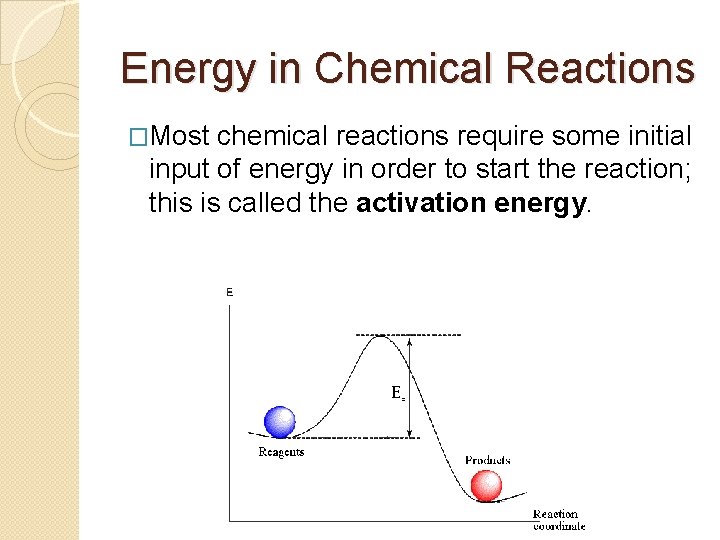
Energy in Chemical Reactions �Most chemical reactions require some initial input of energy in order to start the reaction; this is called the activation energy.

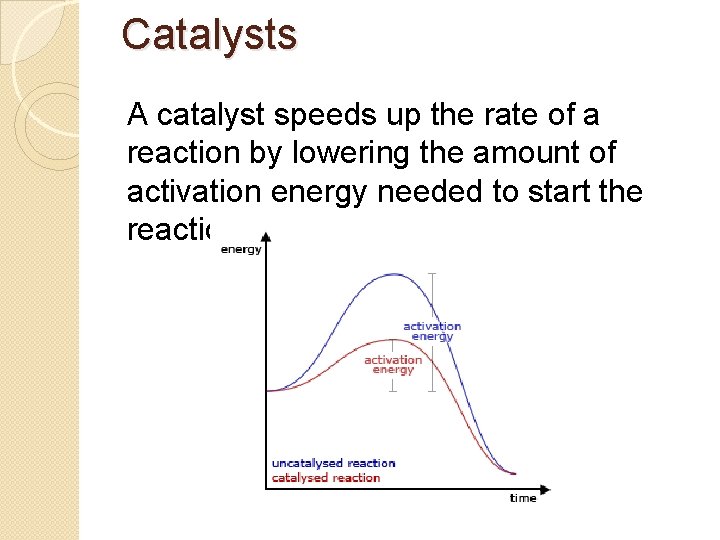
Catalysts A catalyst speeds up the rate of a reaction by lowering the amount of activation energy needed to start the reaction.

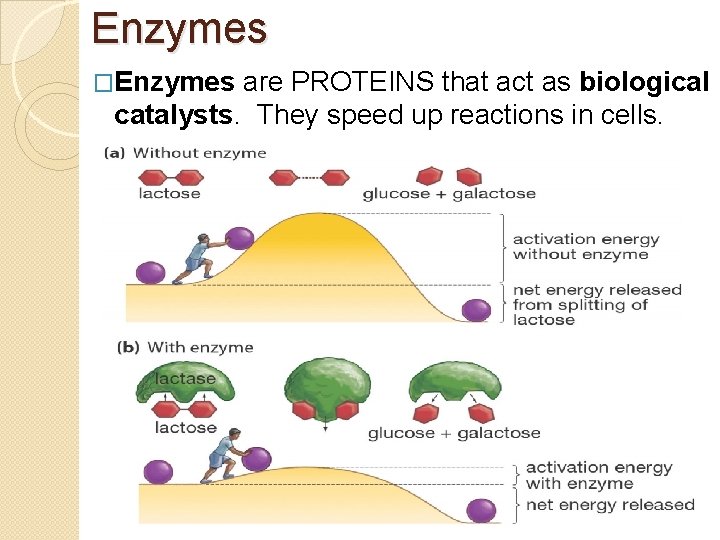
Enzymes �Enzymes are PROTEINS that act as biological catalysts. They speed up reactions in cells.

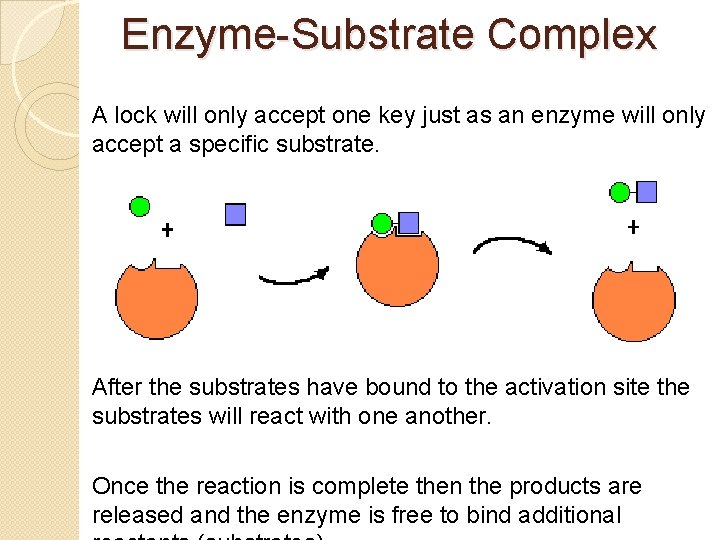
Enzyme-Substrate Complex �Enzymes provide a site for the reactants (substrates) to react. This site is called the activation site. �The activation site and the substrate have complementary shapes, similar to a lock & key.

Enzyme-Substrate Complex A lock will only accept one key just as an enzyme will only accept a specific substrate. After the substrates have bound to the activation site the substrates will react with one another. Once the reaction is complete then the products are released and the enzyme is free to bind additional

Enzyme-Substrate Complex If the substrates do not fit in the activation site then the reaction will not take place. Each enzyme is very specific

Rate of Enzyme Activity There are factors that can affect an enzyme: 1. Temperature Each enzyme has an optimal temperature range. At high temps they may denature (break down) and not work correctly.

Rate of Enzyme Activity 2. p. H �There is an optimal p. H for every enzyme to function at. (usually at 7, neutral p. H) �Many enzymes become denatured at low p. H levels (when acid is present).

Rate of Enzyme Activity 3. Concentration of Substrate or Enzyme • The more substrate available, the faster the reaction tends to go • The more enzyme that is available, the faster the reaction tends to go

4. Cofactors and Coenzymes � Help Enzymes work faster (like some minerals and vitamins) 13

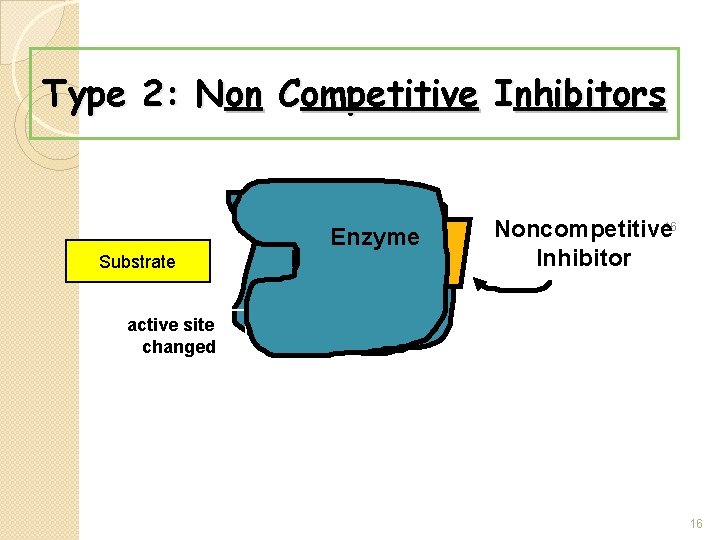
5. Inhibitors � Stop enzymes from doing their jobs � Two types 1. Competitive inhibitors: look like the substrate and block the active site 2. Noncompetitive inhibitors: bind to the enzyme causing its shape to change, changing the active site. 14

Type 1: Competitive Inhibitors Stop enzymes from doing their jobs. � Two types ◦ Competitive inhibitors: look like the substrate and block the Enzyme active site. Substrate � Competitive inhibitor 15

Type 2: Non Competitive Inhibitors Substrate Enzyme Noncompetitive 16 Inhibitor active site changed 16