1 of 40 Boardworks Ltd 2008 2 of

1 of 40 © Boardworks Ltd 2008
2 of 40 © Boardworks Ltd 2008

Safety precautions When scientists carry out experiments, they have to take safety precautions. This is to make sure that they minimize the risk of harming themselves, other people and the environment. What kind of safety precautions should you take in the lab? What kind of safety precautions should chemists working in industrial labs take? 3 of 40 © Boardworks Ltd 2008

What are hazard symbols? When working in a lab, how can you tell which chemicals are safe and which are dangerous? Hazard labels are used on containers and vehicles that contain dangerous chemicals. toxic irritant harmful corrosive flammable These hazard symbols show why the chemical is dangerous. Why is it important that symbols are used, rather than words? 4 of 40 © Boardworks Ltd 2008

What do hazard symbols mean? 5 of 40 © Boardworks Ltd 2008

What do hazard symbols mean? 6 of 40 © Boardworks Ltd 2008
7 of 40 © Boardworks Ltd 2008

Everyday reactions Chemical reactions happen all around us, not just in the lab. cooking materials sticking burning rusting living Can you think of more everyday chemical reactions? 8 of 40 © Boardworks Ltd 2008

How can you spot a chemical reaction? Lots of changes happen in chemical reactions. You might recognize that a chemical reaction is happening because the substances involved: l change colour l give off a gas l get hot l get cold l give out light l make smells l form solids. 9 of 40 © Boardworks Ltd 2008

Chemical reactions are hard to reverse Chemical reactions are usually difficult to reverse. For example, magnesium burns in oxygen to form magnesium oxide. It is not at all easy to ‘un-burn’ the magnesium once it has been burnt. magnesium + oxygen magnesium oxide Many reactions need an input of energy to get them started. This is called activation energy. Many reactions (like the burning of magnesium) also release energy once the reaction has started. 10 of 40 © Boardworks Ltd 2008
Odd-one-out 11 of 40 © Boardworks Ltd 2008

Non-useful chemical reactions Chemical reactions in food cause it to decay, making it unsafe to eat. Chemical reactions like these are a nuisance, but other reactions can even be dangerous. The reaction between iron and oxygen causes the metal to become rusty, which weakens the structure and makes it dangerous to use. Factories sometimes produce waste gases that pollute the atmosphere. These gases cause acid rain and might contribute to the greenhouse effect. 12 of 40 © Boardworks Ltd 2008

Useful chemical reactions 13 of 40 © Boardworks Ltd 2008

More useful chemical reactions Chemists carry out chemical reactions to produce new substances that improve people’s lives. 14 of 40 © Boardworks Ltd 2008

The chemistry of food There are many chemical reactions involved in our food: how many can you think of? l cooking l ripening fruit l food going mouldy l making fertilizers and pesticides l photosynthesis for plants to grow l digestion when you eat food l fermentation to make bread, beer and wine. 15 of 40 © Boardworks Ltd 2008

Cooking involves chemical reactions. Changes in colour, taste and texture are due to the molecules in food joining together in new ways. Cooking an egg changes its texture from runny to firm. Eggs contain a protein called albumen. The protein molecules are long chains of amino acids folded into a ball shape. When eggs are heated, some of the proteins break apart and the molecules unfold. These molecules then join to other nearby protein molecules until they are all linked in a network. 16 of 40 © Boardworks Ltd 2008

Making bread and alcohol Yeast is a living organism that carries out the chemical reactions that are used for making bread and alcohol. Yeast uses oxygen from the air for aerobic respiration. sugar C 6 H 12 O 6 + oxygen carbon dioxide 6 O 2 6 CO 2 + water 6 H 2 O What effect do you think the carbon dioxide gas has on the bread? Yeast can also carry out respiration without oxygen. This is called anaerobic respiration, or fermentation. This is used to produce the alcohol in beer and wine. 17 of 40 © Boardworks Ltd 2008

Ripening fruit The ripening of fruit is a complex collection of chemical reactions. Take apples as an example: l Starch is broken down into sugars, increasing sweetness. l Acids are neutralized, making the apples less sour. l Chlorophyll (green) changes to anthocyanin (red). l Pectin, a chemical that makes apples hard, is broken down, making the apples softer. Why do food producers and supermarkets need to know about the conditions and reactions involved in ripening fruit? 18 of 40 © Boardworks Ltd 2008

What causes ripening? The ripening of all fruit and vegetables involves similar chemical reactions. The speed of ripening is affected by the temperature and by the presence of a chemical called ethene, C 2 H 4. Food scientists can tell producers and supermarkets the best conditions for slowing down or speeding up the ripening process so that fruit and vegetables arrive in the shops perfectly ripe. 19 of 40 © Boardworks Ltd 2008

Food spoilage The changes that take place as food ‘spoils’ are also caused by chemical reactions. For example, peeled apples turn brown when exposed to the air because they react with oxygen in the air. Knowing what causes this reaction can help to slow it down. Placing sliced apples in water prevents browning because the apples are no longer exposed to air. The chemical ascorbic acid (vitamin C), which is found in lemon juice, is also known to prevent the browning reaction. 20 of 40 © Boardworks Ltd 2008

Chemistry and cleaning Grease and oil on clothing, cutlery, skin and hair are difficult to remove with just water, because oil and water do not mix. Adding soap allows the oil and water to mix. Soap molecules have two ends: one end is attracted to the water molecules; the other end is attracted to the oil molecules. 21 of 40 head of molecule bonds to water molecules tail of molecule bonds to oil molecules © Boardworks Ltd 2008

Chemistry and cosmetics The ingredients in cosmetics like makeup, shampoo and fake tan are made by chemical reactions. Many of the ingredients in these products are made from crude oil. Is it sustainable to use ingredients like these? 22 of 40 © Boardworks Ltd 2008

Chemistry and medicine The medicines that we take are all made by chemists. Many medicines started out as natural products, which came from living things: l aspirin – from the bark of willow trees l penicillin – from mould l morphine – from poppies l digoxin – from foxgloves. Do you know what these medicines are used for? What do you think might be the advantages and disadvantages of using drugs from plants and moulds? 23 of 40 © Boardworks Ltd 2008
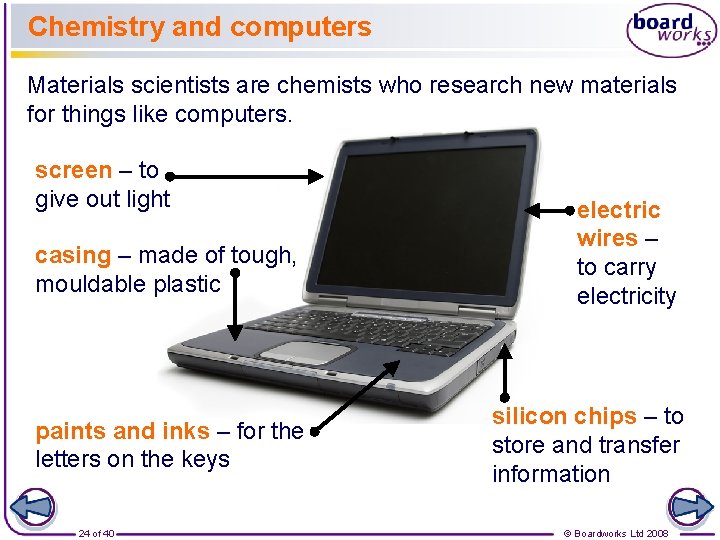
Chemistry and computers Materials scientists are chemists who research new materials for things like computers. screen – to give out light casing – made of tough, mouldable plastic paints and inks – for the letters on the keys 24 of 40 electric wires – to carry electricity silicon chips – to store and transfer information © Boardworks Ltd 2008

Reactants and products In a chemical reaction, one or more new substances are always formed. The starting substances used in a reaction are the reactants. The new substances formed in a reaction are the products. reactants products The arrow means ‘change into’. In a chemical reaction, the reactants change into the products. It is often difficult to reverse a chemical reaction and change the products back into the reactants. 25 of 40 © Boardworks Ltd 2008

Word equations A word equation is a quick, shorthand way of writing a chemical reaction. There always three parts to a word equation: l the names of the reactants l an arrow l the names of the products. What is the word equation for hydrogen reacting with oxygen to form water? hydrogen 26 of 40 + oxygen water © Boardworks Ltd 2008

Reactant or product? In this chemical reaction, which substances are the reactants and which substances are the products? magnesium + copper oxide magnesium oxide + substance reactant or product? magnesium oxide product magnesium reactant copper oxide reactant copper product 27 of 40 copper © Boardworks Ltd 2008

Write the word equations 1. Magnesium burns brightly in oxygen to form magnesium oxide. + magnesium oxygen magnesium oxide 2. Calcium hydroxide reacts with hydrochloric acid to form calcium chloride and water. calcium hydroxide + hydrochloric calcium acid chloride + water 3. Sodium reacts with hydrochloric acid to form sodium chloride and hydrogen. sodium 28 of 40 + hydrochloric sodium acid chloride + hydrogen © Boardworks Ltd 2008
29 of 40 © Boardworks Ltd 2008
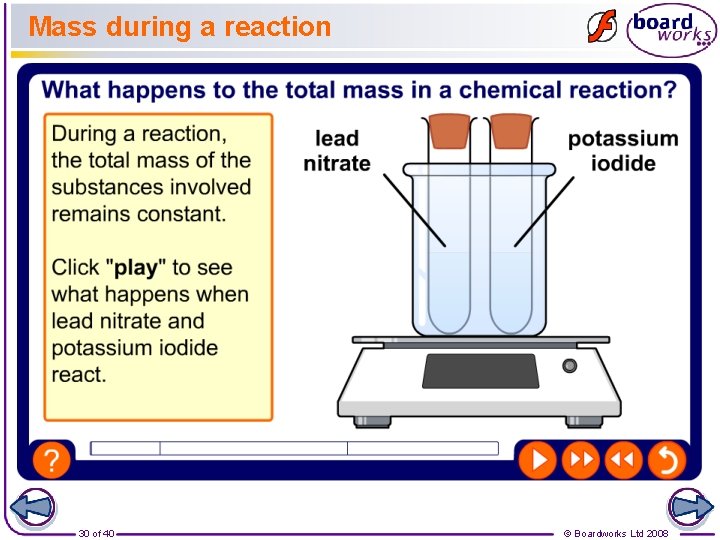
Mass during a reaction 30 of 40 © Boardworks Ltd 2008

Why doesn’t the mass change? In chemical reactions, no atoms can be made or destroyed. Chemical reactions just change how the atoms are bonded together. + + 31 of 40 © Boardworks Ltd 2008

What happens to the mass? 32 of 40 © Boardworks Ltd 2008

Why did the mass increase? In the reaction between magnesium and oxygen, the mass increased. This is because the magnesium atoms joined up with oxygen atoms. + What is the word equation for this reaction? magnesium 33 of 40 + oxygen magnesium oxide © Boardworks Ltd 2008

What happens to the mass? 34 of 40 © Boardworks Ltd 2008

Why did the mass decrease? The mass of the copper carbonate decreased as it was heated because it decomposed to form copper oxide and carbon dioxide. What is the word equation for this reaction? Why did the mass decrease? The mass decreased because the carbon dioxide gas escaped out into the air. Can you calculate the mass of carbon dioxide that was produced in the reaction? 35 of 40 © Boardworks Ltd 2008

Chemical reactions – true or false? 36 of 40 © Boardworks Ltd 2008
37 of 40 © Boardworks Ltd 2008

Glossary 38 of 40 © Boardworks Ltd 2008

Anagrams 39 of 40 © Boardworks Ltd 2008
Multiple-choice quiz 40 of 40 © Boardworks Ltd 2008
- Slides: 40