Types of Chemical Reactions Predicting Products from the


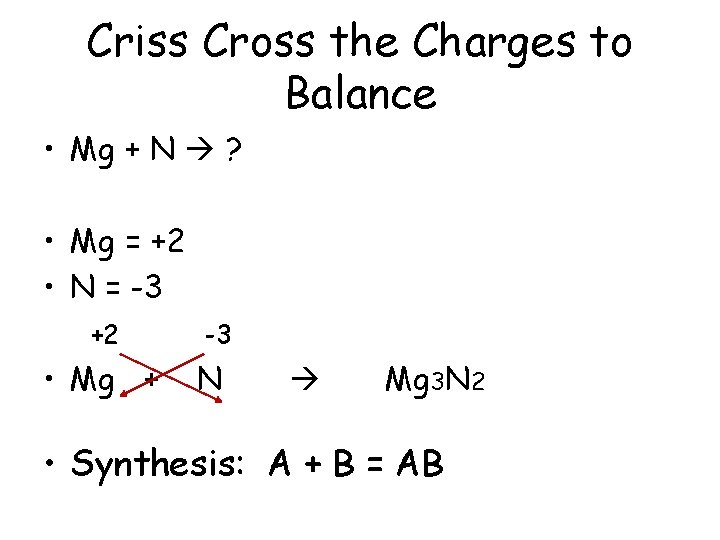

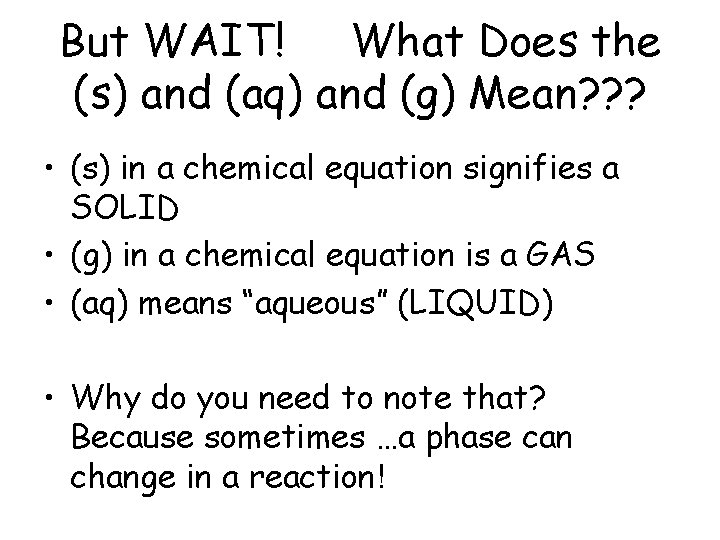






- Slides: 19

Types of Chemical Reactions Predicting Products from the Reactants

Types of Reactions 1. 2. 3. 4. 5. Synthesis reactions Decomposition reactions Single displacement reactions Double displacement reactions Combustion reactions You need to be able to identify each

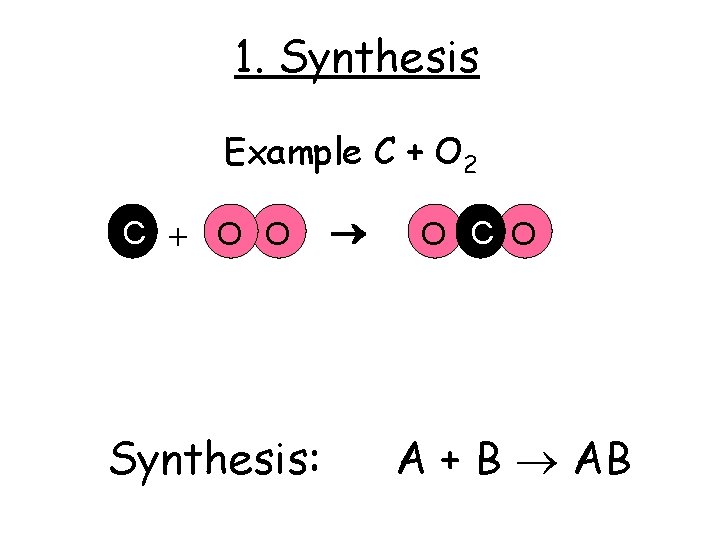
1. Synthesis Example C + O 2 C + O O Synthesis: O C O A + B AB
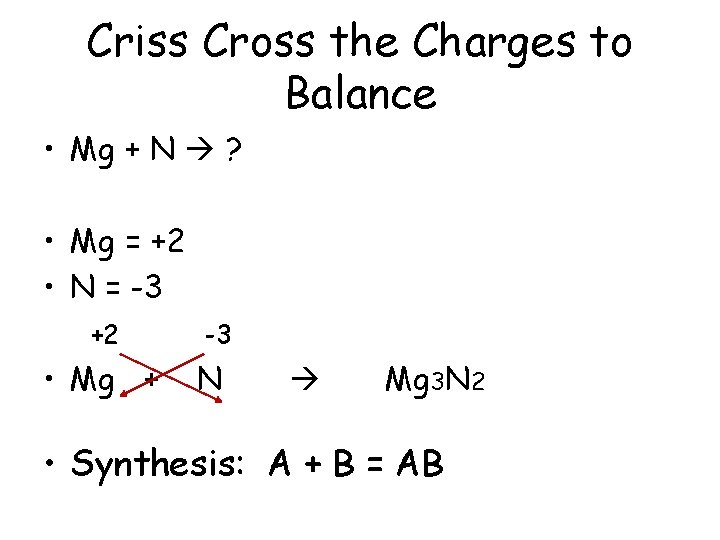
Criss Cross the Charges to Balance • Mg + N ? • Mg = +2 • N = -3 +2 • Mg + -3 N Mg 3 N 2 • Synthesis: A + B = AB

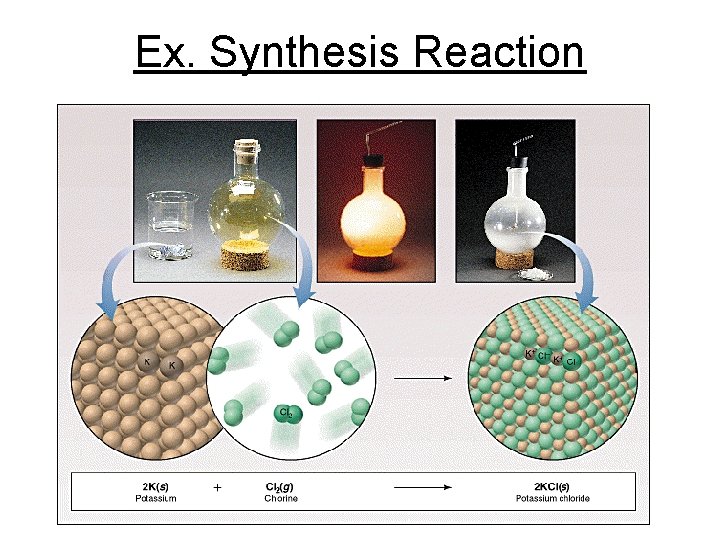
Ex. Synthesis Reaction

Practice • Predict the products: 2 Na(s) + Cl 2(g) 2 Na. Cl(s) Mg(s) + F 2(g) Mg. F 2(s) 2 Al(s) + 3 F 2(g) 2 Al. F 3(s) • Now, balance them. (Criss-Cross!)
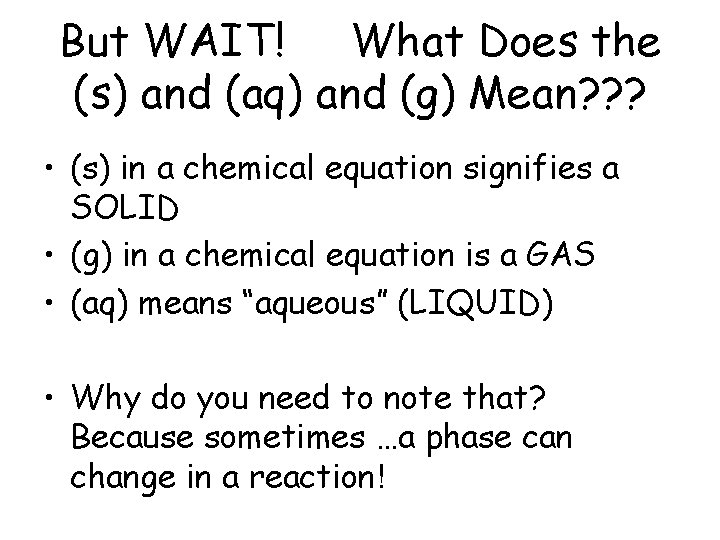
But WAIT! What Does the (s) and (aq) and (g) Mean? ? ? • (s) in a chemical equation signifies a SOLID • (g) in a chemical equation is a GAS • (aq) means “aqueous” (LIQUID) • Why do you need to note that? Because sometimes …a phase can change in a reaction!

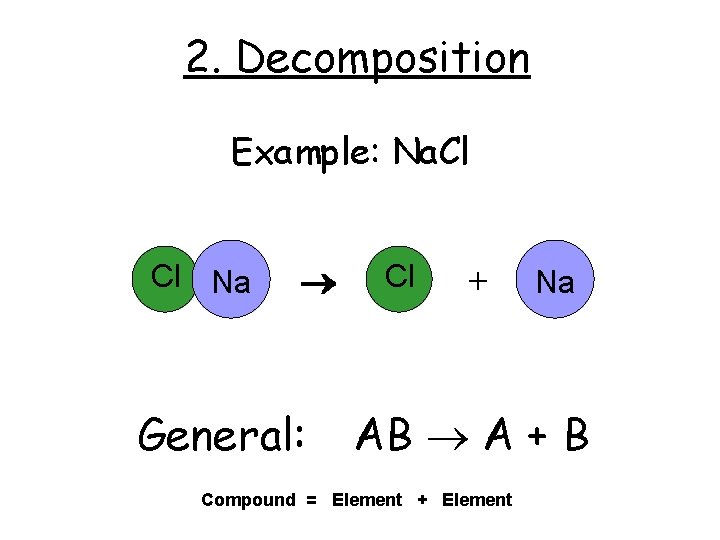
2. Decomposition Example: Na. Cl Cl Na General: Cl + Na AB A + B Compound = Element + Element

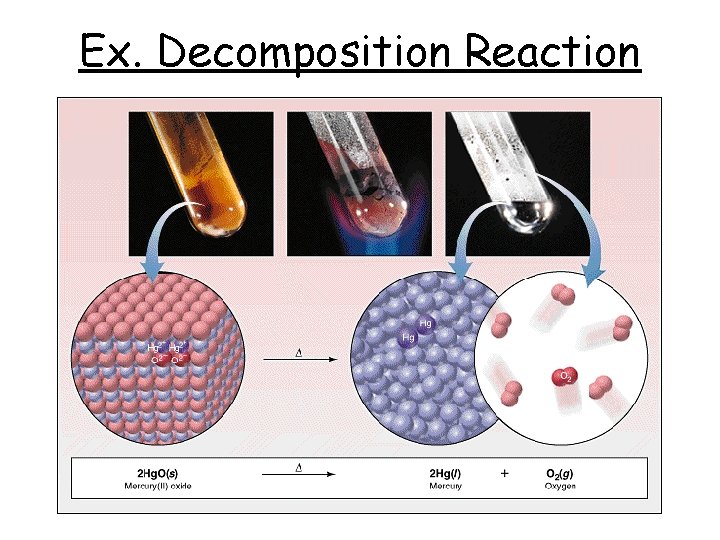
Ex. Decomposition Reaction

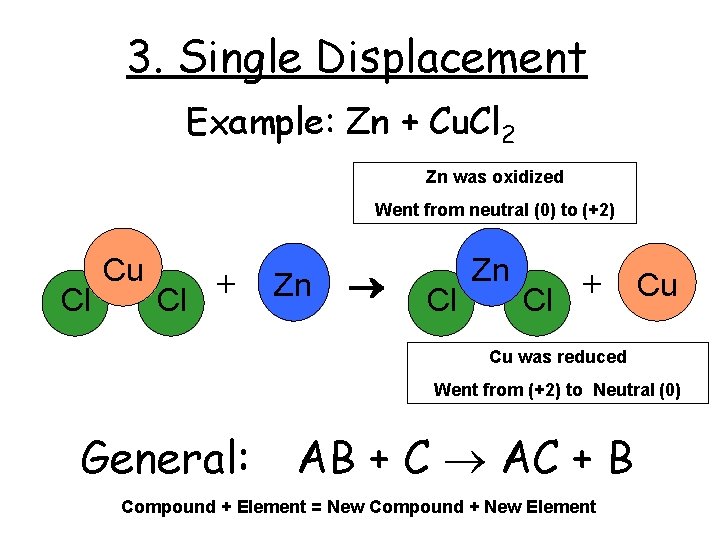
3. Single Displacement Example: Zn + Cu. Cl 2 Zn was oxidized Went from neutral (0) to (+2) Cl Cu + Cl Zn + Cu Cl Cu was reduced Went from (+2) to Neutral (0) General: AB + C AC + B Compound + Element = New Compound + New Element

But WAIT! What do “oxidized” and ”reduced” Mean? • • Remember? LEO the lion says GER Lose electrons = oxidation = LEO Gain electrons = reduction = GER • Metals LOSE electrons • Non-metals GAIN electrons

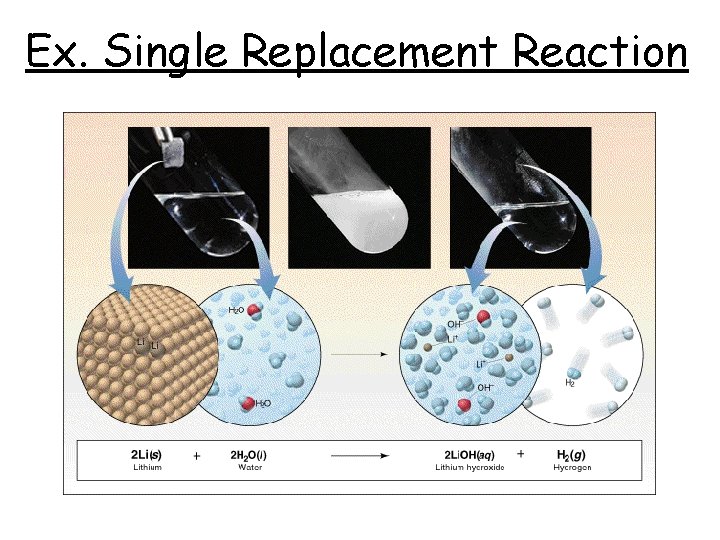
Ex. Single Replacement Reaction

Single Replacement Reactions • Write and balance the following single replacement reaction equations: • Zn(s) + 2 HCl(aq) Zn. Cl 2 + H 2(g) • 2 Na. Cl(s) + F 2(g) 2 Na. F(s) + Cl 2(g) • 2 Al(s)+ 3 Cu(NO 3)2(aq) 3 Cu(s)+ 2 Al(NO 3)3(aq)

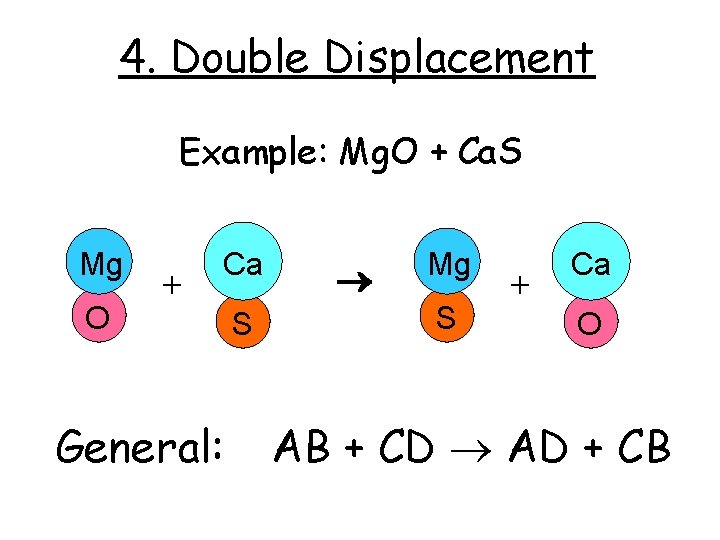
4. Double Displacement Example: Mg. O + Ca. S Mg O + Ca General: S Mg S + Ca O AB + CD AD + CB

Double Replacement Reactions • Think about it like “foil”ing in algebra, first and outer ions go together + inside ions go together • Example: Ag. NO 3(aq) + Na. Cl(s) Ag. Cl(s) + Na. NO 3(aq) • Another example: K 2 SO 4(aq) + Ba(NO 3)2(aq) 2 KNO 3(aq) + Ba. SO 4(s)

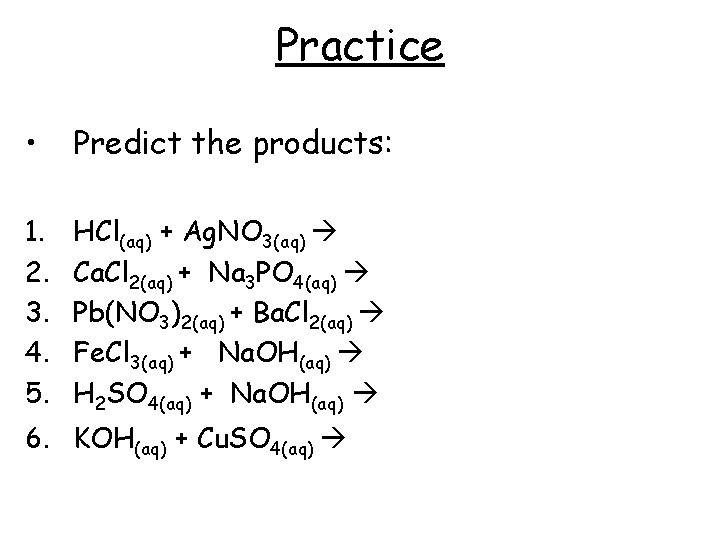
Practice • Predict the products: 1. 2. 3. 4. 5. HCl(aq) + Ag. NO 3(aq) Ca. Cl 2(aq) + Na 3 PO 4(aq) Pb(NO 3)2(aq) + Ba. Cl 2(aq) Fe. Cl 3(aq) + Na. OH(aq) H 2 SO 4(aq) + Na. OH(aq) 6. KOH(aq) + Cu. SO 4(aq)

5. Combustion Reactions • Combustion reactions – when a hydrocarbon reacts with oxygen gas • This is also called BURNING! • In order to burn something you need the 3 things in the “fire triangle”: • 1) Fuel (hydrocarbon) 2) Oxygen 3) Something to ignite the reaction (spark)

Combustion Reactions • In general: Cx. Hy + O 2 CO 2 + H 2 O • Products are ALWAYS CARBON DIOXIDE AND WATER! • Combustion is used to heat homes and run automobiles (octane, as in gasoline, is a hydrocarbon: C 8 H 18 )

Mixed Practice • State the type of reaction & predict the products (try to balance the equation!) 1. 2. 3. 4. 5. Ba. Cl 2 + H 2 SO 4 C 6 H 12 + O 2 Zn + Cu. SO 4 Cs + Br 2 Fe. CO 3