PREDICTING PRODUCTS Chemical Reactions COMBUSTION REACTIONS Always have

PREDICTING PRODUCTS Chemical Reactions

COMBUSTION REACTIONS Always have similar reactants: Cn H n + O 2 Always create the same products: CO 2 + H 2 O Balance! C 3 H 8 + O 2 CO 2 + H 2 O CH 3 CH 2 OH + O 2 CO 2 + H 2 O

SINGLE REPLACEMENT A + BX AX + B Single element replaces one of the elements in compound NEVER carry a subscript, only the charge of the ion! Use your polyatomic ion charges Use the periodic table/ periodic table of ions HNO 3 + Zn ? ? ?

SINGLE REPLACEMENT First - decide if the single element forms a cation or an anion and give it the ionic charge. Zinc is the single element and will form a +2 cation. Second, determine which element in the compound the single element will replace. Cations can only replace cations, anions can only replace anions. (positive replaces positive, negative replaces negative ions) In this reaction, the hydrogen acts as the cation. Zinc should replace hydrogen. Next, check the activity series to determine if the reaction will occur.

ACTIVITY SERIES Activity series - a list of elements based on reactivity. An element in the activity series can replace any element below it. Since zinc is replacing hydrogen, zinc must be above hydrogen in the activity series for the reaction to occur. It is, so the reaction occurs.
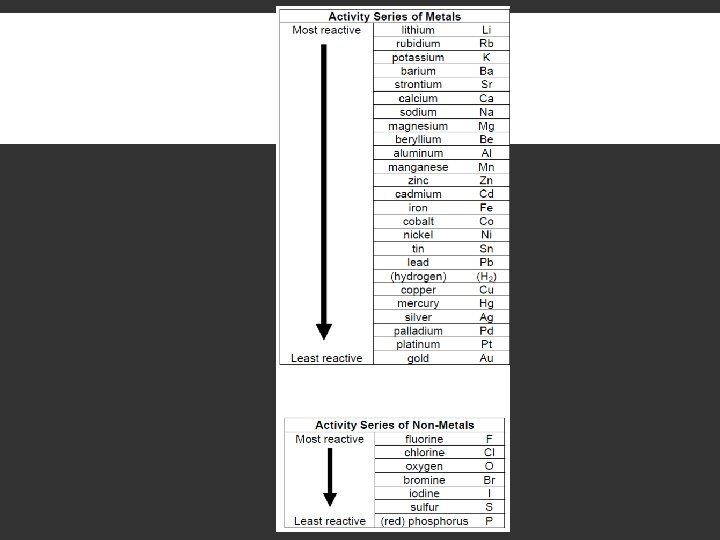

SINGLE REPLACEMENT Form products with correct subscripts Balance equation +1 -1 +2 +1 +2 -1 2 HNO 3 + Zn H 2 + Zn(NO 3)2

DOUBLE REPLACEMENT AX + BY AY + BX Two compounds and the partners will switch! Again…No subscript, only charge! Ag. NO 3 + Na. Cl ? ? ?

DOUBLE REPLACEMENT Assign charges to each of the ions. Positive replaces positive (or negative replaces negative) Compounds MUST have a cation and an anion Form products and give them the correct subscripts Balance the equation. +1 -1 Ag. NO 3 + Na. Cl ? ? ?

DOUBLE REPLACEMENT Ag+1 and Na+1 will switch. Na+1 will compound with NO 3 -1 and Ag+1 will compound with Cl-1 +1 -1 Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3

DOUBLE REPLACEMENT +1 -1 Ag. NO 3 + Na. Cl Ag. Cl + Na. NO 3 +2 -1 +2 -2 +2 -1 Pb(NO 3)2 + Cu. SO 4 Pb. SO 4 + Cu(NO 3)2

DOUBLE REPLACEMENT Na. OH + Fe. Cl 3 ? ? ? Na. Cl + Fe(OH)3 Pb(NO 3)2 + KI ? ? ? Pb. I 2 + KNO 3 Ca. Cl 2 + Na. OH ? ? ? Na. Cl + Ca(OH)2

DECOMPOSITION AX A + X Compound breaks apart into two smaller elements/compounds Acids decompose into water and an oxide. H 2 CO 3 H 2 O + CO 2 Metallic hydroxides (anything with a metal and an OH) decompose to form a metallic oxide and water. Mg(OH)2 Mg. O + H 2 O Metallic carbonates decompose to form a metallic oxide and carbon dioxide. Li 2 CO 3 Li 2 O + CO 2

SYNTHESIS A + X AX Two simple elements combine to form one compound. Again…No subscript, only charge! Na + Cl 2 ? ? ?

SYNTHESIS Assign charges to each of the ions (if you have a metal and a nonmetal) to create an ionic compound. Form the product and give it the correct subscripts. Balance the equation. +1 -1 2 Na + Cl 2 2 Na. Cl

PREDICTING PRODUCTS ACTIVITY Match the Reactants with the Products Take a sheet of paper with you Your name on paper Write each equation out 4 in group – 2 per group member 3 in group – 3 per group member Balance the equation List the reaction Combustion, Decomposition, Single Replacement, Double Replacement, Synthesis Staple group together and turn in when finished
- Slides: 16