Predicting Products of Chemical Reactions Types of Reactions




























- Slides: 31

Predicting Products of Chemical Reactions

Types of Reactions • As part of our course you will need to be able to identify the type of chemical reactions as well as predict the products of a reaction. n n Synthesis Decomposition Single displacement Double displacement reactions n n w/ neutralization as a subset. Combustion reactions

Steps to Writing Reactions Some steps for doing reactions 1. Identify the type of reaction 2. Predict the product(s) using the type of reaction as a model 3. Balance it. Don’t forget about the diatomic elements! (Br. INCl. HOF) For example, Oxygen is O 2 as an element.

1. Synthesis reactions -> occur when two substances (generally elements) combine and form a compound. (Sometimes these are called combination or addition reactions. ) reactant + reactant 1 product Basically: A + B AB Example: 2 H 2 + O 2 2 H 2 O Example: C + O 2 CO 2

Synthesis Reactions Here is another example of a synthesis reaction

Practice Predict the products. Write and balance the following synthesis reaction equations. Sodium metal reacts with chlorine gas Na(s) + Cl 2(g) Solid Magnesium reacts with fluorine gas Mg(s) + F 2(g) Aluminum metal reacts with fluorine gas Al(s) + F 2(g)

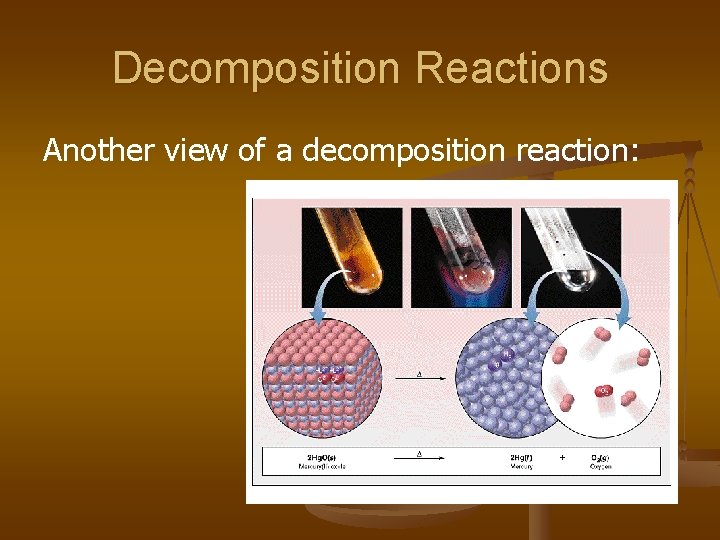
2. Decomposition Reactions Decomposition reactions-> occur when a compound breaks up into the elements or in a few to simpler compounds 1 Reactant Product + Product In general: AB A + B Example: 2 H 2 O 2 H 2 + O 2 Example: 2 Hg. O 2 Hg + O 2

Decomposition Reactions Another view of a decomposition reaction:

Decomposition Exceptions • Carbonates and chlorates are special case decomposition reactions that do not go to the elements. • Carbonates (CO 32 -) decompose to carbon dioxide and a metal oxide • • Example: Ca. CO 3 CO 2 + Ca. O Chlorates (Cl. O 3 -) decompose to oxygen gas and a metal chloride • Example: 2 Al(Cl. O 3)3 2 Al. Cl 3 + 9 O 2

Practice Predict the products. Then, write and balance the following decomposition reaction equations: Solid Lead (IV) oxide decomposes Pb. O 2(s) Aluminum nitride decomposes Al. N(s)

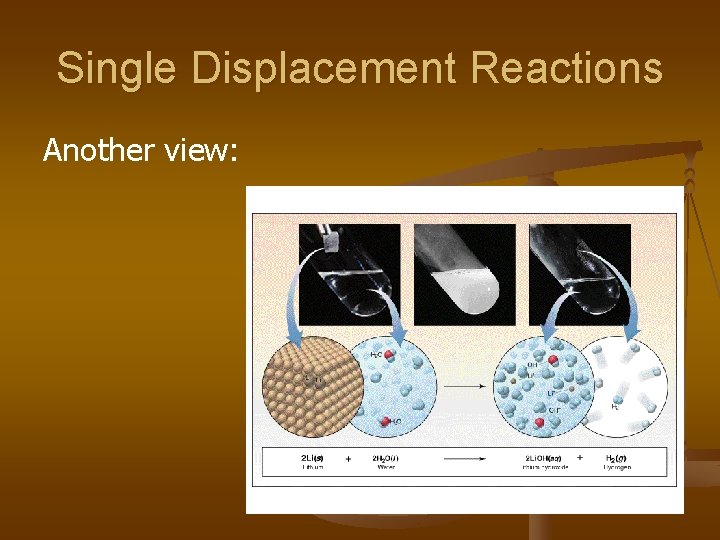
3. Single Displacement Reactions -> occur when one element replaces another in a compound. A metal can replace a metal (+) OR a nonmetal can replace a nonmetal (-). element + compound A + BC AC + B (if A is a metal) OR A + BC BA + C (if A is a nonmetal) (remember the cation always goes first!) When H 2 O splits into ions, it splits into H + and OH - (not H+ and O -2 !!)

Single Displacement Reactions Another view:

Single Displacement Reactions Write and balance the following single replacement reaction equation: Zinc metal reacts with aqueous hydrochloric acid. Zn(s) + 2 HCl(aq) Zn. Cl 2 + H 2(g) Note: Zinc replaces the hydrogen ion in the reaction.

Single Displacement Reactions Predict the products. Then, write and balance the following Single Displacement reaction equations: Al(s)+ Cu(NO 3)2(aq) Fe(s)+ Cu. Cl 2(aq) Cl 2(g)+ Ca. Br 2(aq)

4. Double Displacement Reactions -> occur when a metal replaces a metal in a compound a nonmetal replaces a nonmetal in a compound. Compound + compound+ compound AB + CD AD + CB

Double Displacement Reactions Think about it like “foil”ing in algebra, first and last ions go together + inside ions go together Example: Ag. NO 3(aq) + Na. Cl(s) Ag. Cl(s) + Na. NO 3(aq) Another example: K 2 SO 4(aq) + Ba(NO 3)2(aq) 2 KNO 3(aq) +Ba. SO 4(s)

Practice Predict the products. Balance the equation. HCl(aq) + Ag. NO 3(aq) Ca. Cl 2(aq) + Na 3 PO 4(aq) Pb(NO 3)2(aq) + Ba. Cl 2(aq) Fe. Cl 3(aq) + Na. OH(aq) KOH(aq) + Cu. SO 4(aq)

Neutralization Reactions Remember that neutralization reactions are a special type of double displacement. In a neutralization reaction - an acid is reacted with a base to produce an ionic compound and water. HA + BOH BA + H 2 O HCl + Na. OH Na. Cl + H 2 O H 2 SO 4 + 2 Na. OH Na 2 SO 4 +2 H 2 O

Practice Predict the products. Balance the equation. HCl + Ba(OH)2 H 3 PO 4 + Na. OH H 2 SO 4 + Al(OH)3

5. Combustion Reactions Combustion reactions -> occur when a hydrocarbon reacts with oxygen gas. In order to burn something you need the 3 things in the “fire triangle”: 1) A Fuel (hydrocarbon) 2) Oxygen to burn it with 3) Something to ignite the reaction (spark)

Combustion Reactions In general: Cx. Hy + O 2 CO 2 + H 2 O Products in combustion are ALWAYS carbon dioxide and water. (although incomplete burning does cause some byproducts like carbon monoxide)

Combustion Write the products and balance the following combustion reaction: C 10 H 22 + O 2 C 5 H 12 + O 2 C 2 H 6 + O 2

Mixed Review State the type, predict the products, and balance the following reactions: Ba. Cl 2 + H 2 SO 4 C 6 H 12 + O 2 Zn + Cu. SO 4 Cs + Br 2 Fe 2 O 3 H 3 PO 4 + Na. OH

What about NO reaction? There are times when reactions do NOT occur. For our cases they will usually be single displacement reactions. If the element that is trying to replace the other in a compound does NOT have a HIGHER ACTIVITY then NO REACTION will occur.


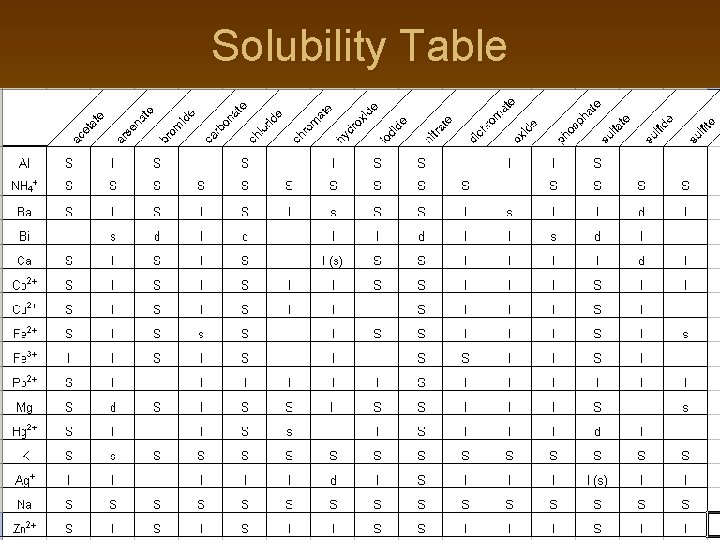
Total Ionic Equations (HONORS ONLY) n n n Once you write the molecular equation (synthesis, decomposition, etc. ), you should check for reactants and products that are soluble or insoluble. We usually assume the reaction is in water We can use a solubility table to tell us what compounds dissolve in water. If the compound is soluble (does dissolve in water), then splits the compound into its component ions If the compound is insoluble (does NOT dissolve in water), then it remains as a compound

Solubility Table

Solubilities Not on the Table! n n Gases only slightly dissolve in water Strong acids and bases dissolve in water n n n Hydrochloric, Hydrobromic, Hydroiodic, Nitric, Sulfuric, Perchloric Acids Group I hydroxides (should be on your chart anyway) Water slightly dissolves in water! (H+ and OH-) For the homework… Sr. SO 4 is insoluble; Be. I 2 and the products are soluble There are other tables and rules that cover more compounds than your table!

Total Ionic Equations Molecular Equation: K 2 Cr. O 4 + Pb(NO 3)2 Pb. Cr. O 4 + 2 KNO 3 Soluble Insoluble Soluble Total Ionic Equation: 2 K+ + Cr. O 4 -2 + Pb+2 + 2 NO 3 - Pb. Cr. O 4 (s) + 2 K+ + 2 NO 3 -

Net Ionic Equations These are the same as total ionic equations, but you should cancel out ions that appear on BOTH sides of the equation Total Ionic Equation: 2 K+ + Cr. O 4 -2 + Pb+2 + 2 NO 3 - Pb. Cr. O 4 (s) + 2 K+ + 2 NO 3 Net Ionic Equation: Cr. O 4 -2 + Pb+2 Pb. Cr. O 4 (s) n

Net Ionic Equations n Try this one! Write the molecular, total ionic, and net ionic equations for this reaction: Silver nitrate reacts with Lead (II) Chloride in hot water. Molecular: Total Ionic: Net Ionic: