Stereochemistry refers to the 3 dimensional properties and



![Specific Rotation, [α] = α / cl a = observed rotation c = concentration Specific Rotation, [α] = α / cl a = observed rotation c = concentration](https://slidetodoc.com/presentation_image_h2/6a0354032aeeb5e4fd96e2f97bdf0060/image-9.jpg)





















- Slides: 53

Stereochemistry refers to the 3 -dimensional properties and reactions of molecules. It has its own language and terms that need to be learned in order to fully communicate and understand the concepts.


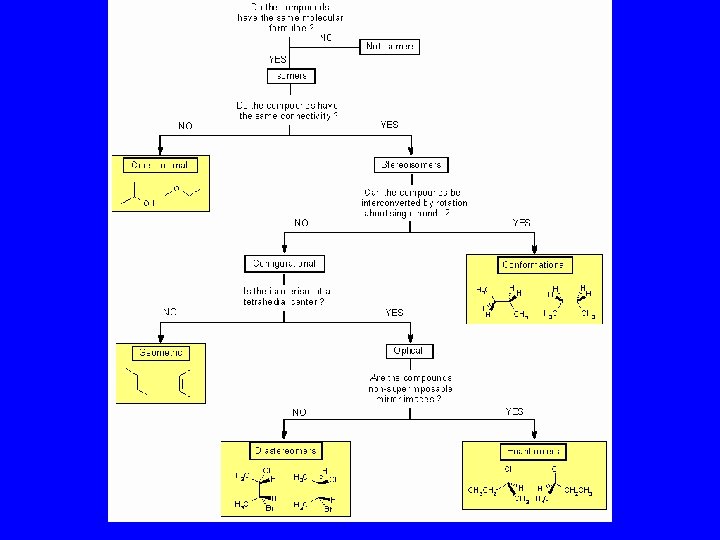
Definitions • Stereoisomers – compounds with the same connectivity, different arrangement in space • Enantiomers – stereoisomers that are nonsuperimposible mirror images; only properties that differ are direction (+ or -) of optical rotation • Diastereomers – stereoisomers that are not mirror images; different compounds with different physical properties

More Definitions • Asymmetric center – sp 3 carbon with 4 different groups attached • Optical activity – the ability to rotate the plane of plane –polarized light • Chiral compound – a compound that is optically active (achiral compound will not rotate light) • Polarimeter – device that measures the optical rotation of the chiral compound

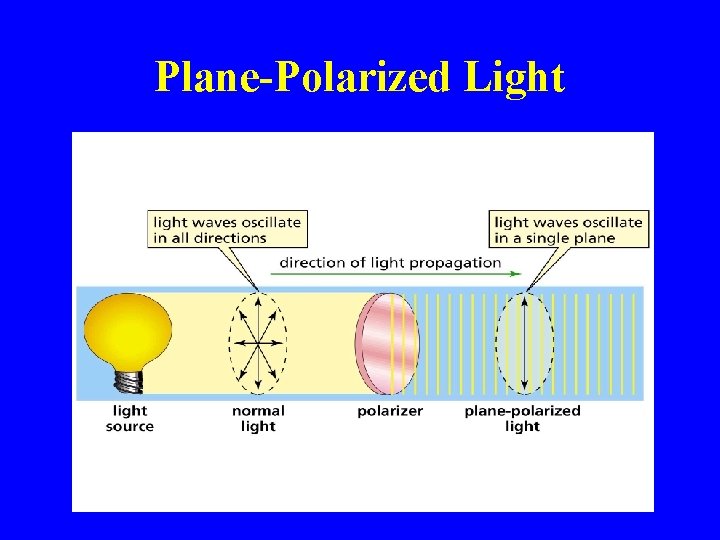
Plane-Polarized Light

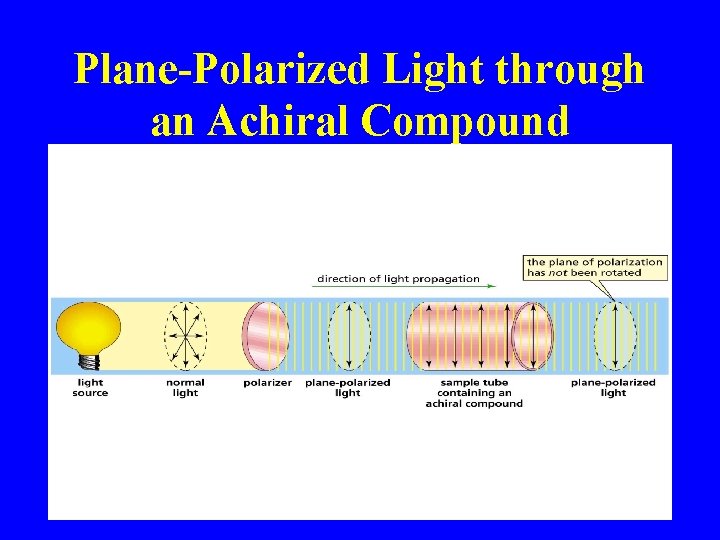
Plane-Polarized Light through an Achiral Compound

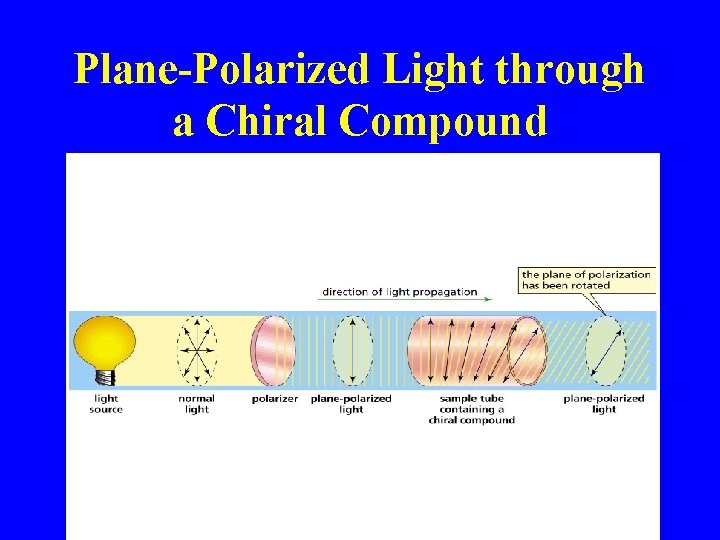
Plane-Polarized Light through a Chiral Compound

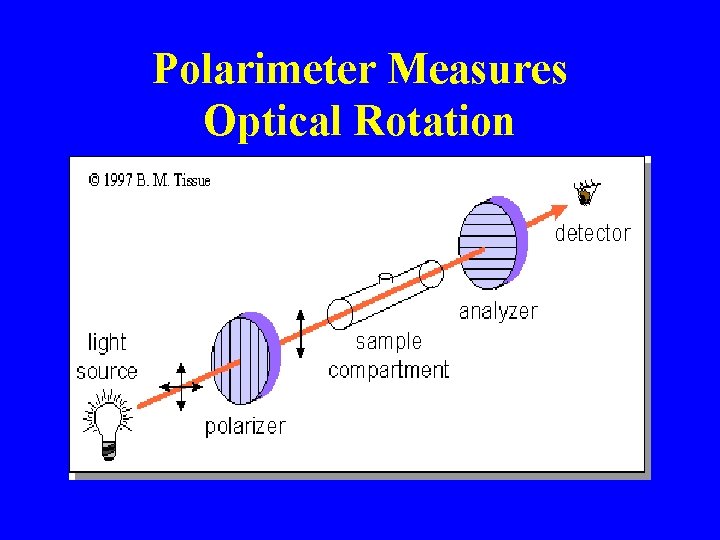
Polarimeter Measures Optical Rotation
![Specific Rotation α α cl a observed rotation c concentration Specific Rotation, [α] = α / cl a = observed rotation c = concentration](https://slidetodoc.com/presentation_image_h2/6a0354032aeeb5e4fd96e2f97bdf0060/image-9.jpg)
Specific Rotation, [α] = α / cl a = observed rotation c = concentration in g/m. L l = length of tube in dm Dextrorotary designated as d or (+), clockwise rotation Levorotary designated as l or (-), counterclockwise rotation

Specific Rotations of some Common Organic Compounds Compound Penicillin V Sucrose Camphor MSG Cholesterol Morphine [a] +233. 0 +66. 5 +44. 3 +25. 5 -31. 3 -132. 0 # * centers 3 10 2 1 8 5

Chirality Center Carbon has four different groups attached

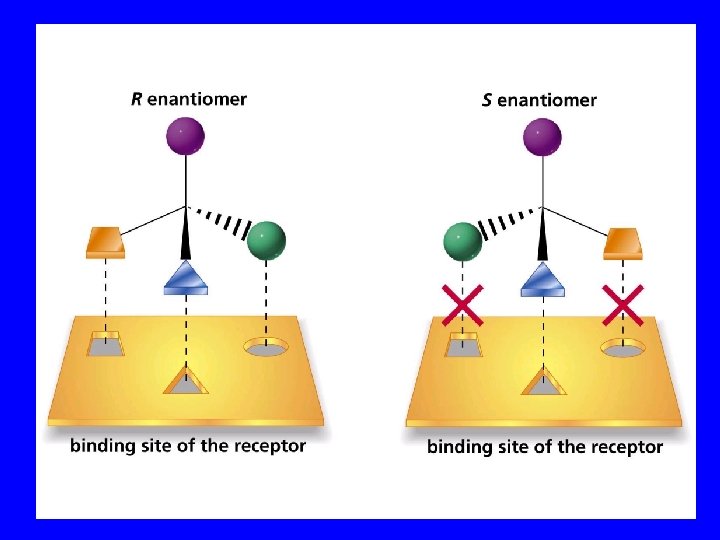
Enantiomers nonsuperimposible mirror images

Enantiomeric Excess (Optical Purity)


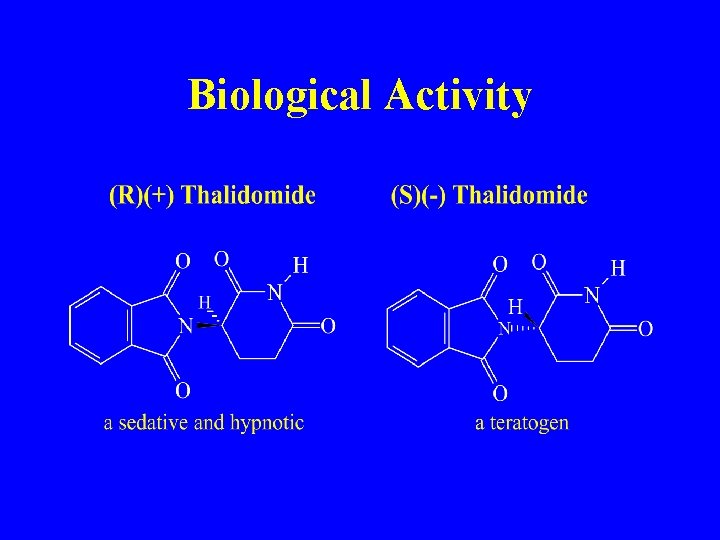
Biological Activity

SSRI Efficacy depends on Stereochemistry

Absolute Configuration

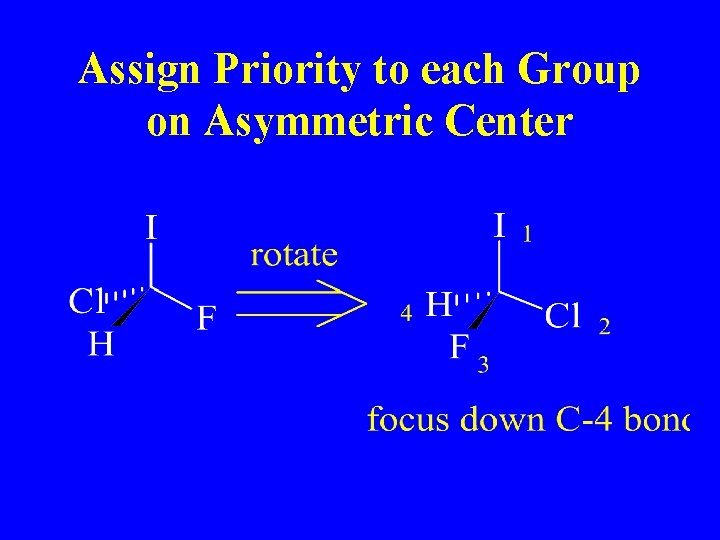
Assign Priority to each Group on Asymmetric Center

Lactic Acid

C. I. P. Priorities

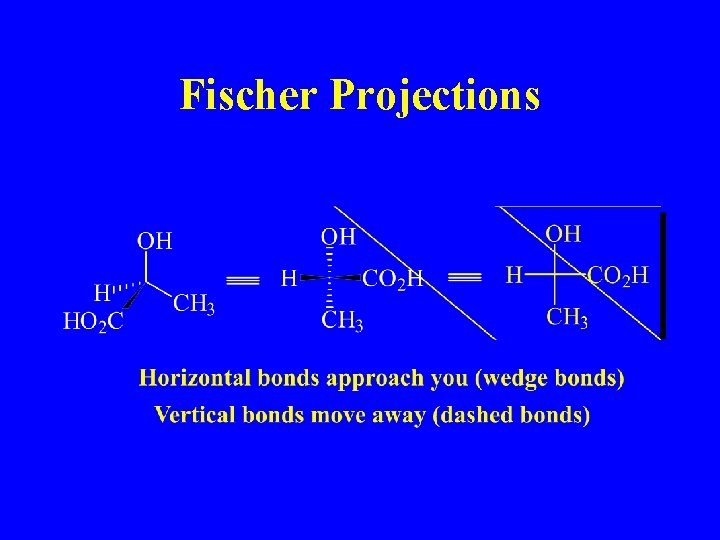
Fischer Projections

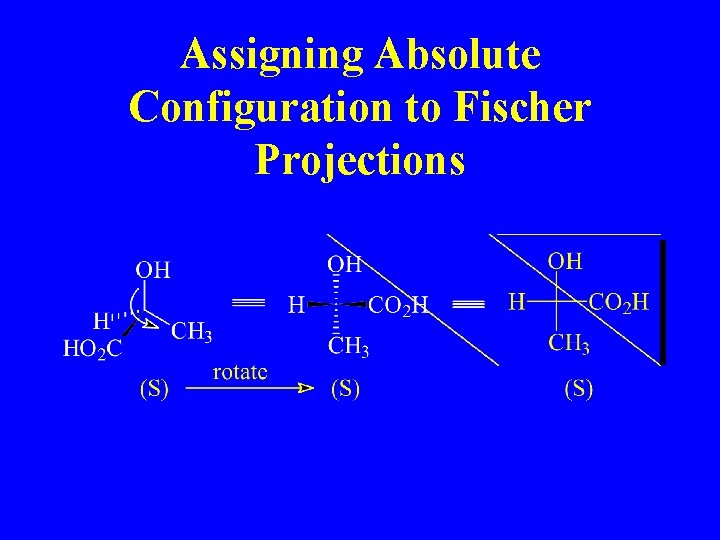
Assigning Absolute Configuration to Fischer Projections

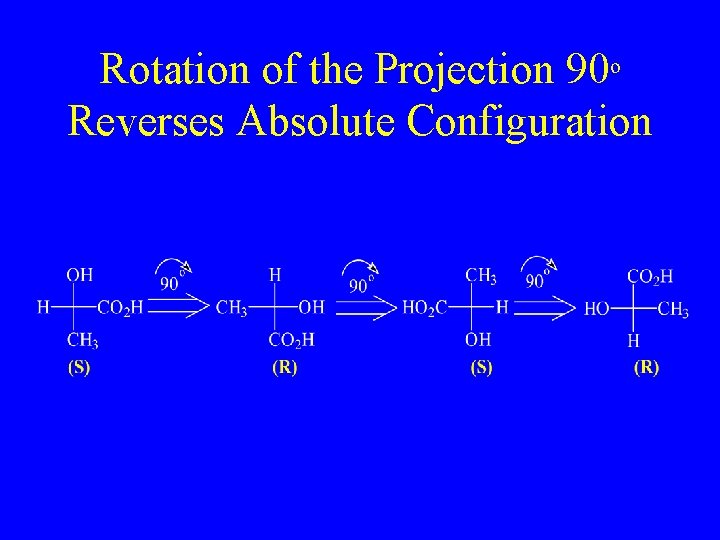
Rotation of the Projection 90 o Reverses Absolute Configuration

Diastereomers Stereoisomers That Are Not Mirror Images

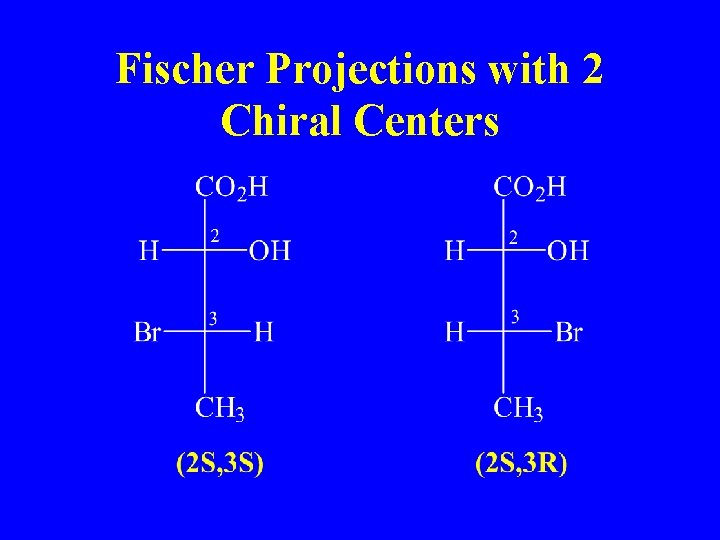
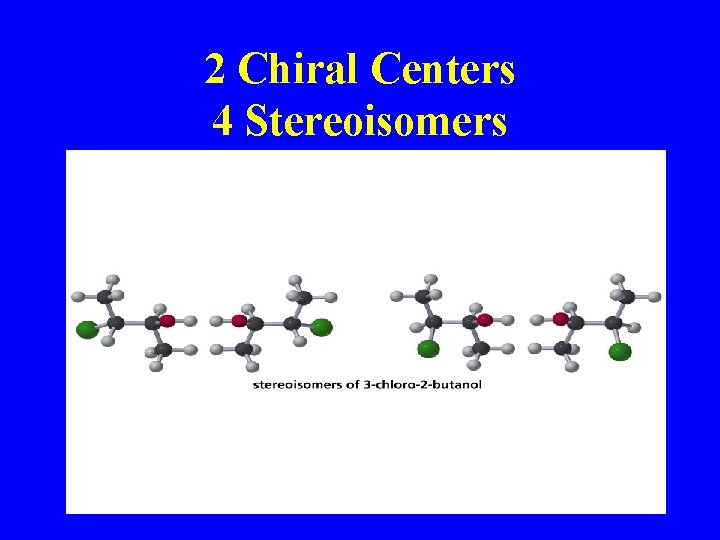
Fischer Projections with 2 Chiral Centers

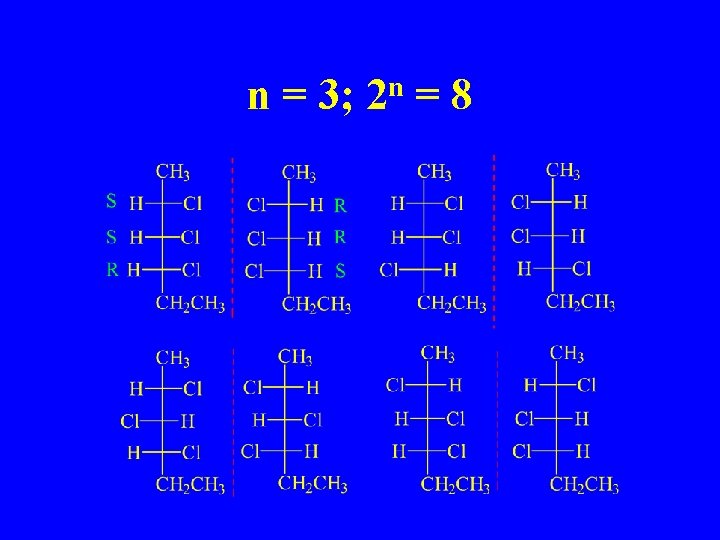
2 Chiral Centers 4 Stereoisomers

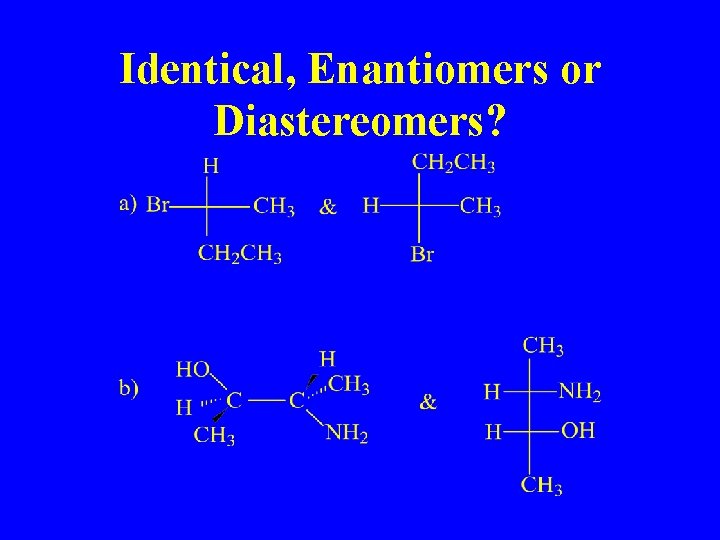
Identical, Enantiomers or Diastereomers?

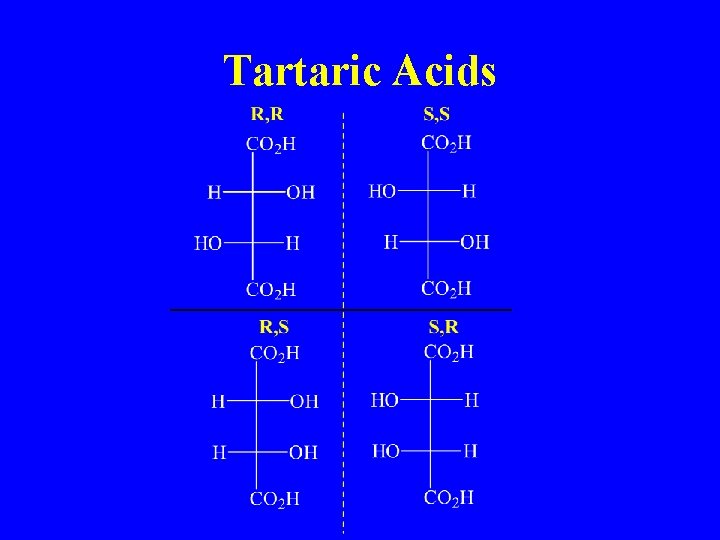
Tartaric Acids

Racemic Mixture

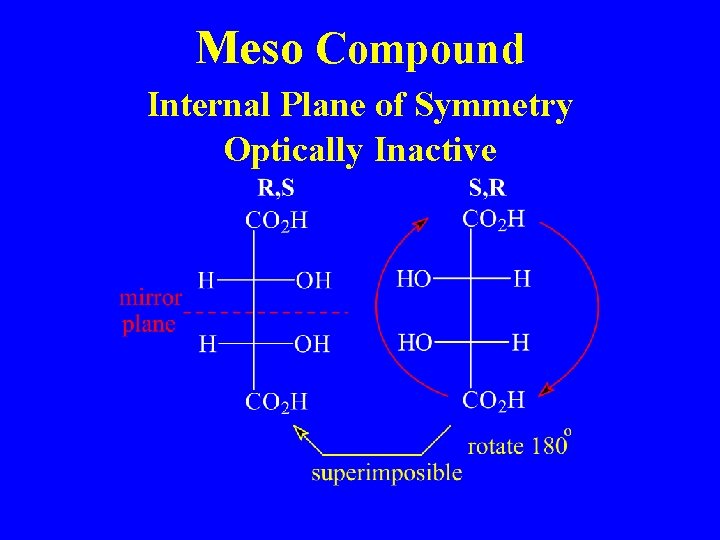
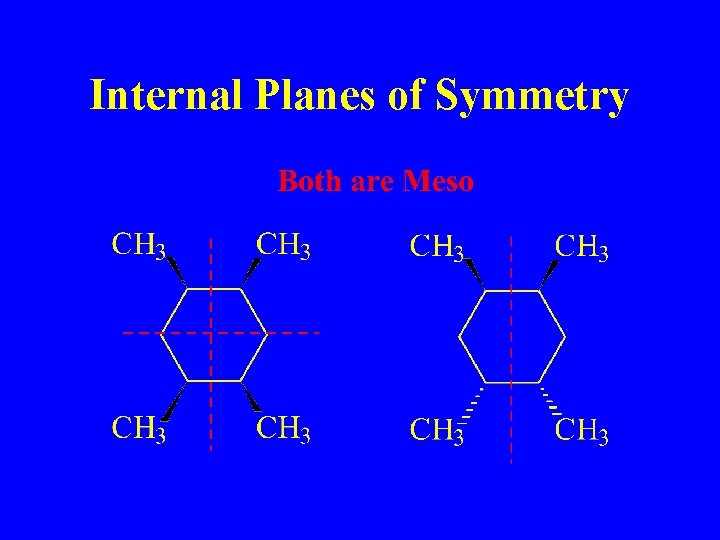
Meso Compound Internal Plane of Symmetry Optically Inactive

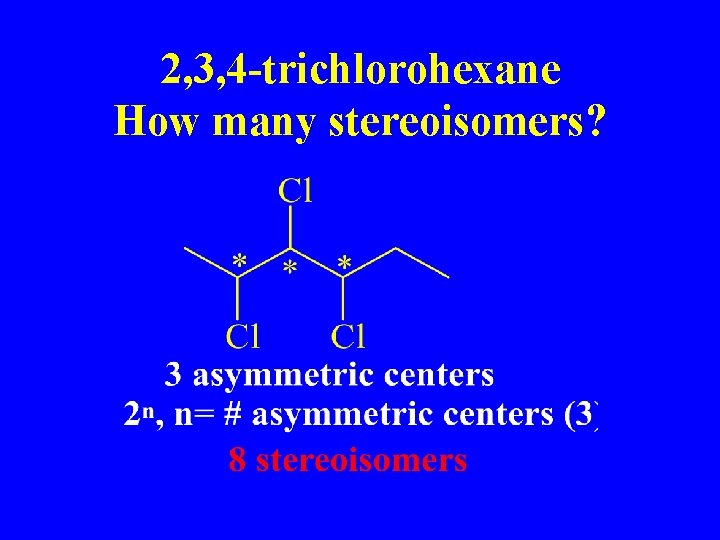
2, 3, 4 -trichlorohexane How many stereoisomers?

n = 3; n 2 =8

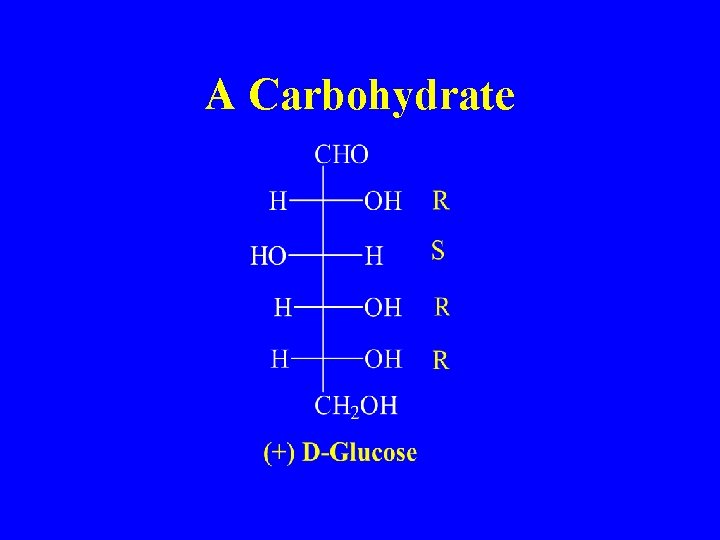
A Carbohydrate

Internal Planes of Symmetry

Asymmetric Centers on Rings

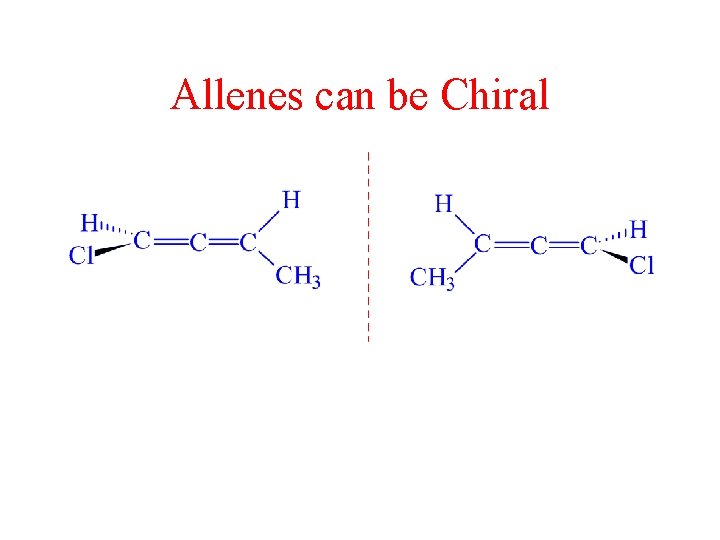
Allenes can be Chiral

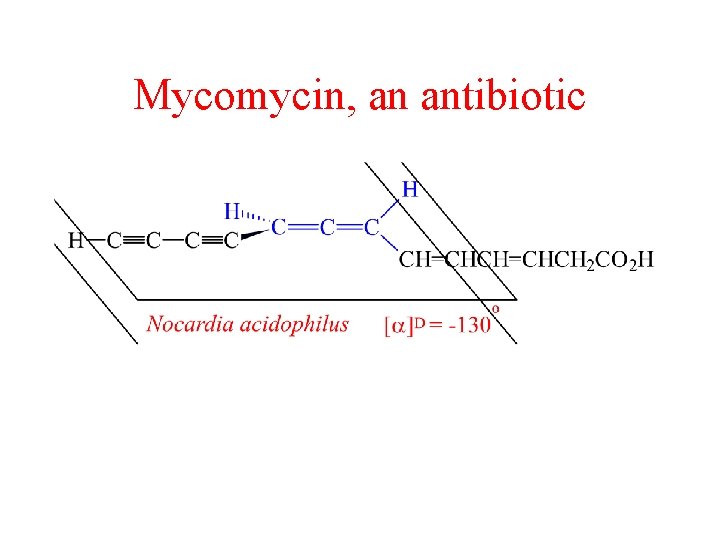
Mycomycin, an antibiotic

Reactions that Generate Chirality Centers Hydrogenation, syn

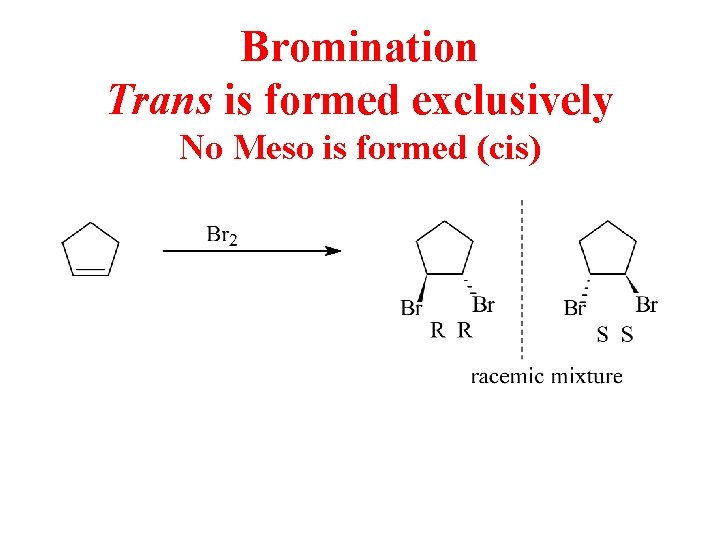
Bromination Trans is formed exclusively No Meso is formed (cis)

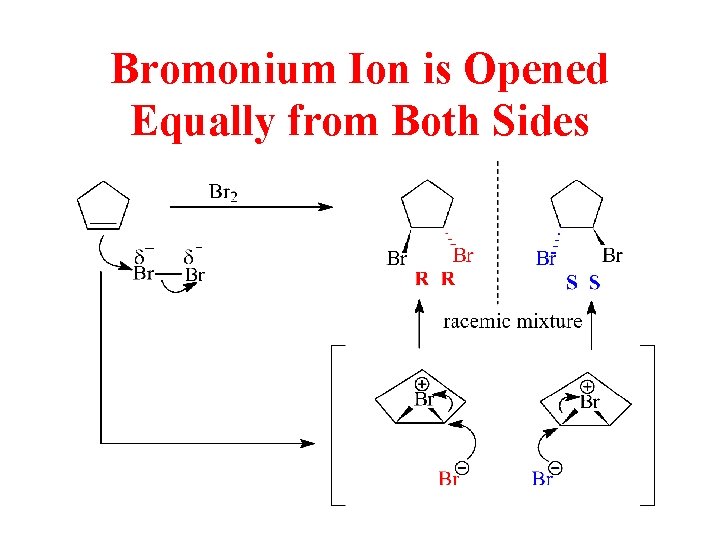
Bromonium Ion is Opened Equally from Both Sides

trans alkene + anti addition = MESO

cis Alkene + anti addition = racemic mixture

Brominations Often Generate Asymmetric Centers

Asymmetric Center is Generated Racemic Mixture Formed

Asymmetric Induction

Preparation of (L)-Dopa for Treatment of Parkinson’s

Relevance of Stereochemistry

One-step synthesis

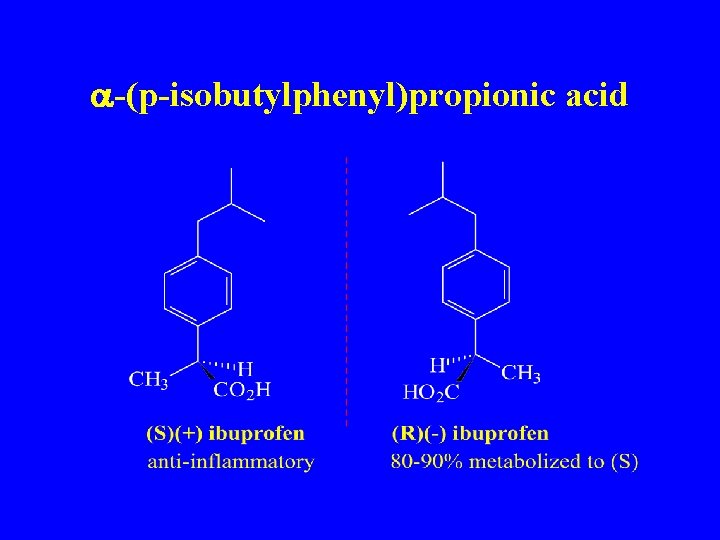
a-(p-isobutylphenyl)propionic acid

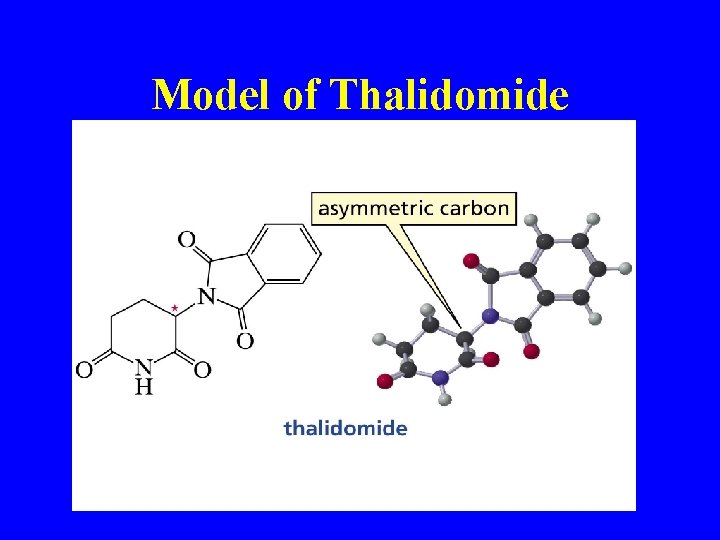
Model of Thalidomide

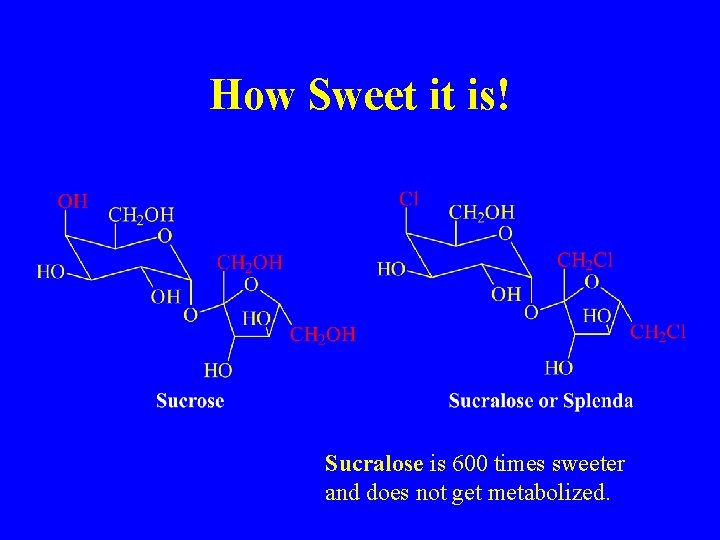
How Sweet it is! Sucralose is 600 times sweeter and does not get metabolized.

Sildenafil (Viagra) and Caffeine

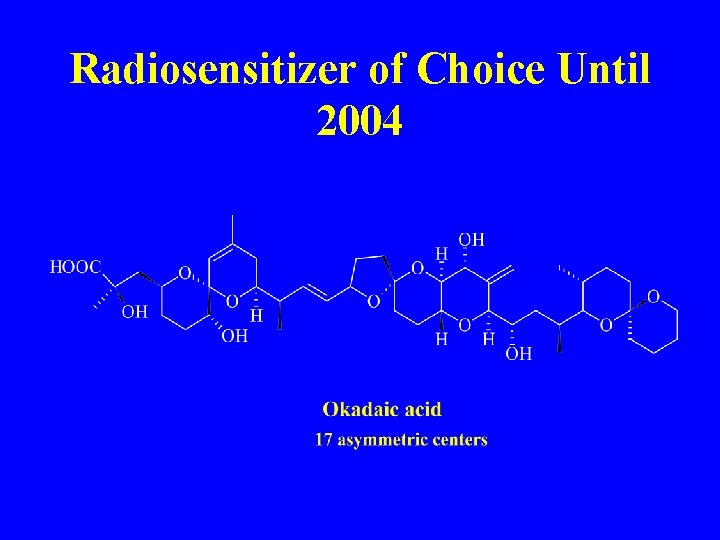
Radiosensitizer of Choice Until 2004