Carbohydrates Lecture 2 Stereochemistry Stereochemistry study of arrangement

Carbohydrates Lecture 2

Stereochemistry • Stereochemistry – study of arrangement of atoms in 3 D space • Carbohydrates with identical functional groups linked in the same order but with different spatial orientation

Are these compounds the same?

Non-superimposable mirror images

Different Compounds • • • Non-superimposable so different compounds One carbon atom is chiral (not symmetric) Non-superimposable mirror images are called enantiomers

Stereochemistry • There are two facts about the molecule that contains one (or more) chiral center: (1) it is optically active and (2) it occurs in more than one isomeric form. • All monosaccharides except dihydroxyacetone are optically active isomeric forms because they contain one or more chiral carbon atoms in their structures. • They are able to rotate the plane of polarized light either clockwise (right) or counter clockwise (left).
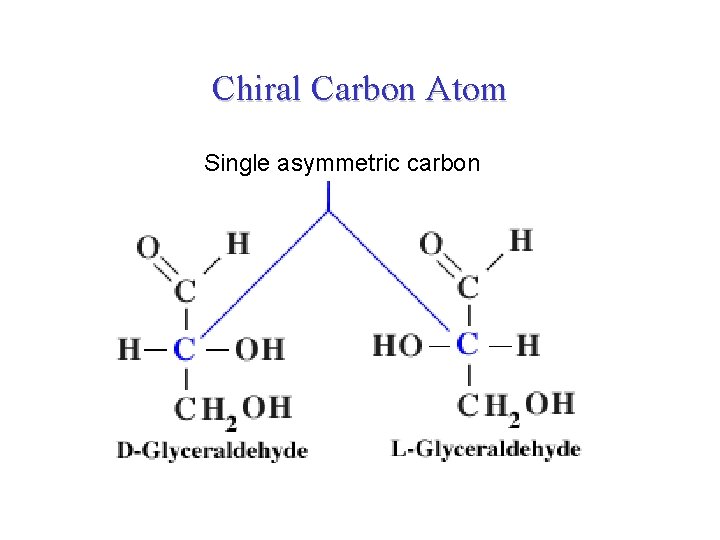
Chiral Carbon Atom Single asymmetric carbon

Configuration about the chiral center • Configuration: the spatial relative arrangement of the substituents around the chiral center. • There are two configurations about each chiral center, the molecule can exist in two isomeric forms. • Accordingly, glyceraldehyde (the simplest aldose) has two different stereoisomers because it has one chiral center at the middle carbon atom. • The two forms are mirror images of each other so they are called enantiomers. By convention, one of these two forms is designated the D isomer, the other the L isomer. • In general, a molecule with n chiral centers can have 2 n Glyceraldehyde has 21 = 2 stereoisomers.
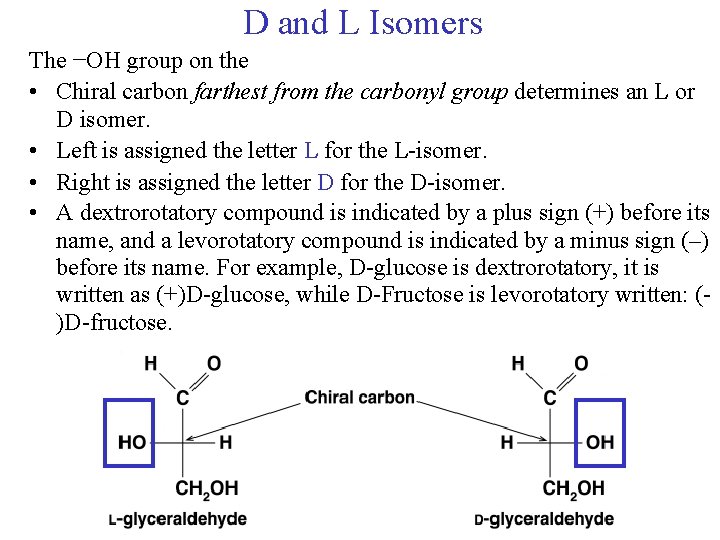
D and L Isomers The −OH group on the • Chiral carbon farthest from the carbonyl group determines an L or D isomer. • Left is assigned the letter L for the L-isomer. • Right is assigned the letter D for the D-isomer. • A dextrorotatory compound is indicated by a plus sign (+) before its name, and a levorotatory compound is indicated by a minus sign (–) before its name. For example, D-glucose is dextrorotatory, it is written as (+)D-glucose, while D-Fructose is levorotatory written: ()D-fructose.
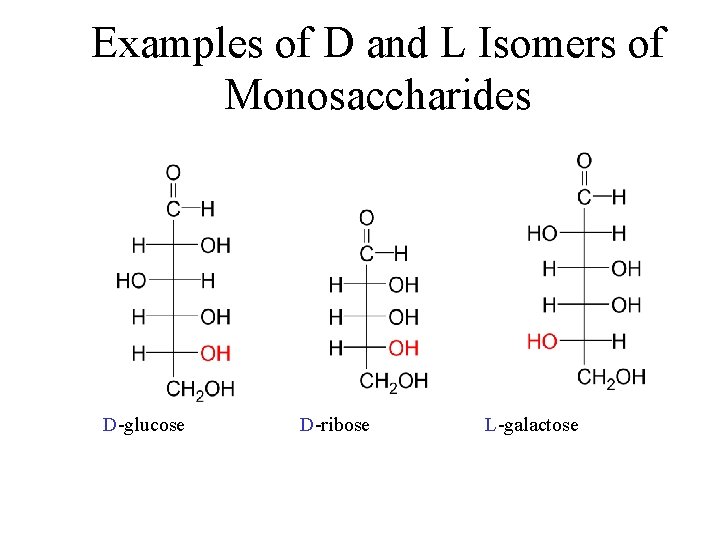
Examples of D and L Isomers of Monosaccharides D-glucose D-ribose L-galactose
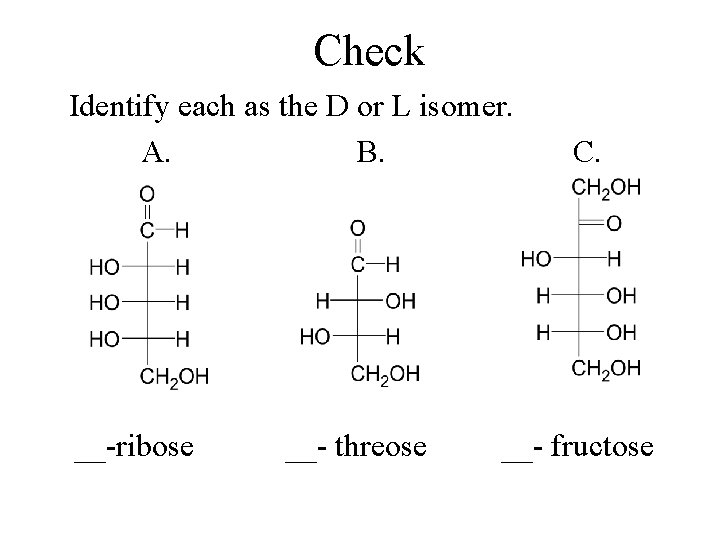
Check Identify each as the D or L isomer. A. B. __-ribose __- threose C. __- fructose
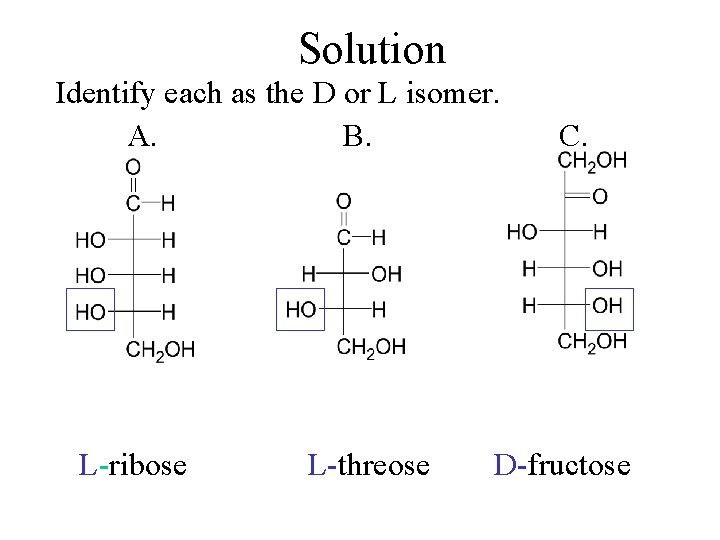
Solution Identify each as the D or L isomer. A. B. L-ribose L-threose C. D-fructose

• • • It is important to know that the carbons of a sugar are numbered beginning from the carbonyl group (the functional group). Stereoisomers that are mirror images of each other are called enantiomers. Pairs of stereoisomers that are not mirror images of each other are called diastereomers. Sugars that differ only by the configuration about one C atom are known as epimers of one another. D-glucose and D-mannose are epimers with respect to C 2, whereas D-glucose and D-galactose are epimers with respect to C 4. However, D-mannose and D-galactose are not epimers of each other because they differ in configuration about two of their carbon atoms.

- Slides: 14