Organic Chemistry 5 th Edition L G Wade


















- Slides: 62

Organic Chemistry, 5 th Edition L. G. Wade, Jr. Chapter 21 Carboxylic Acid Derivatives Jo Blackburn Richland College, Dallas, TX Dallas County Community College District Chapter 21 ã 2003, Prentice Hall

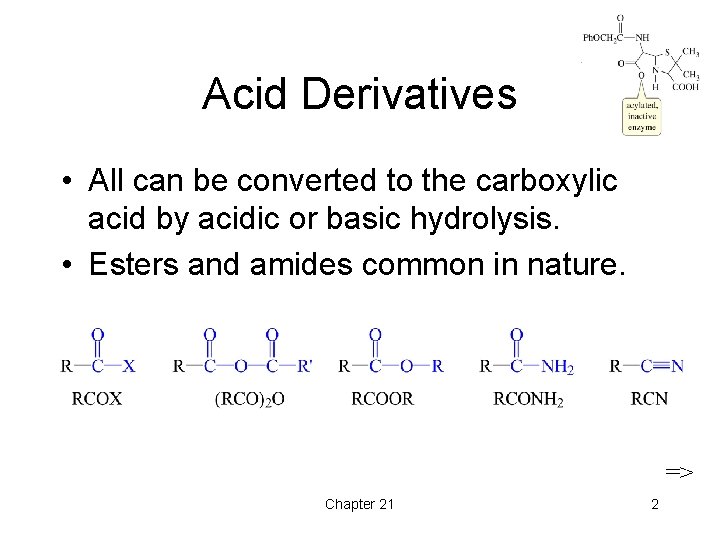
Acid Derivatives • All can be converted to the carboxylic acid by acidic or basic hydrolysis. • Esters and amides common in nature. => Chapter 21 2

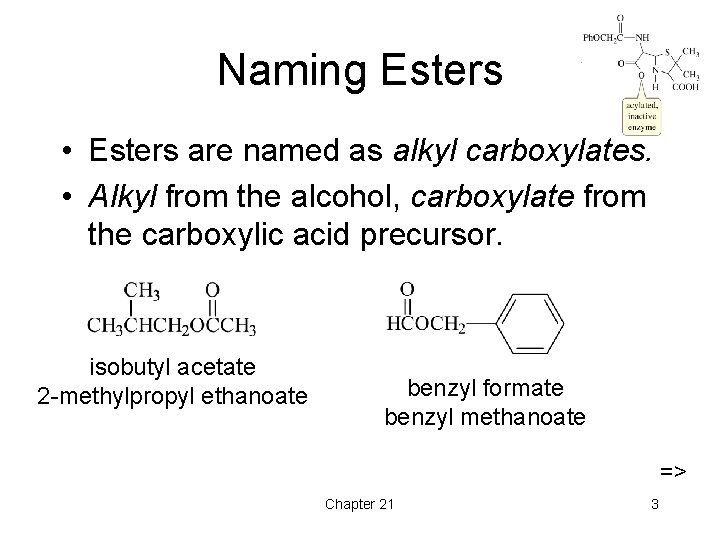
Naming Esters • Esters are named as alkyl carboxylates. • Alkyl from the alcohol, carboxylate from the carboxylic acid precursor. isobutyl acetate 2 -methylpropyl ethanoate benzyl formate benzyl methanoate => Chapter 21 3

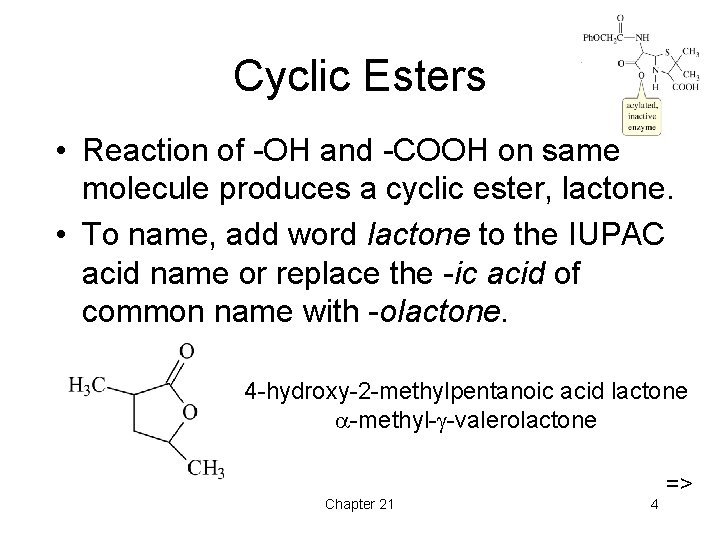
Cyclic Esters • Reaction of -OH and -COOH on same molecule produces a cyclic ester, lactone. • To name, add word lactone to the IUPAC acid name or replace the -ic acid of common name with -olactone. 4 -hydroxy-2 -methylpentanoic acid lactone -methyl- -valerolactone Chapter 21 4 =>

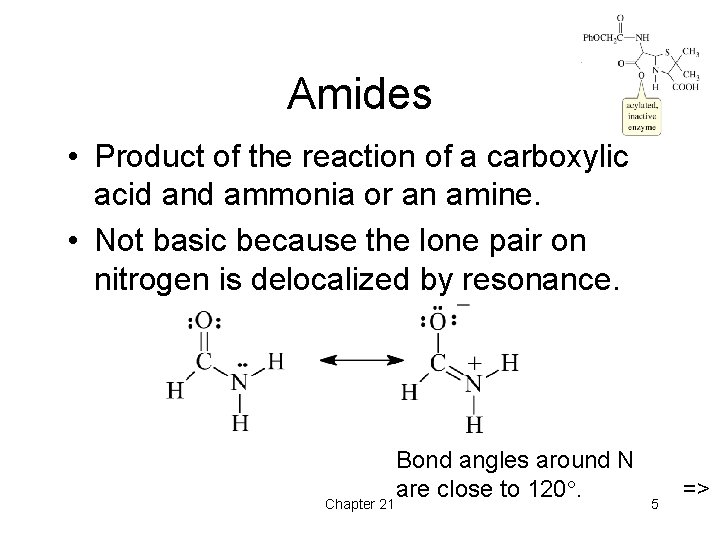
Amides • Product of the reaction of a carboxylic acid and ammonia or an amine. • Not basic because the lone pair on nitrogen is delocalized by resonance. Chapter 21 Bond angles around N are close to 120. 5 =>

Classes of Amides • 1 amide has one C-N bond (two N-H). • 2 amide or N-substituted amide has two C-N bonds (one N-H). • 3 amide or N, N-disubstituted amide has three C-N bonds (no N-H). => Chapter 21 6

Naming Amides • For 1 amide, drop -ic or -oic acid from the carboxylic acid name, add -amide. • For 2 and 3 amides, the alkyl groups bonded to nitrogen are named with N- to indicate their position. N-ethyl-N, 2 -dimethylpropanamide N-ethyl-N-methylisobutyramide Chapter 21 7 =>

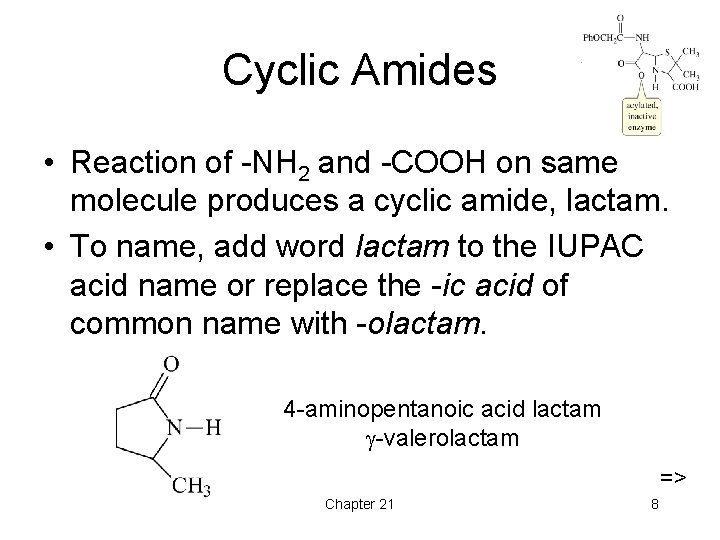
Cyclic Amides • Reaction of -NH 2 and -COOH on same molecule produces a cyclic amide, lactam. • To name, add word lactam to the IUPAC acid name or replace the -ic acid of common name with -olactam. 4 -aminopentanoic acid lactam -valerolactam => Chapter 21 8

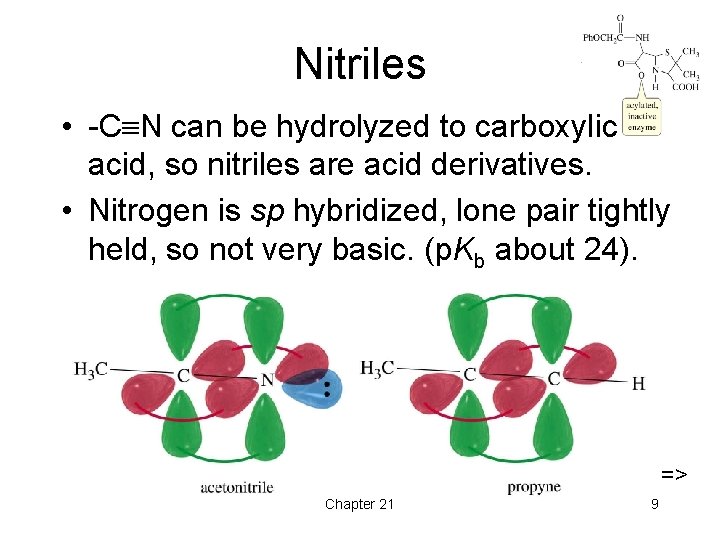
Nitriles • -C N can be hydrolyzed to carboxylic acid, so nitriles are acid derivatives. • Nitrogen is sp hybridized, lone pair tightly held, so not very basic. (p. Kb about 24). => Chapter 21 9

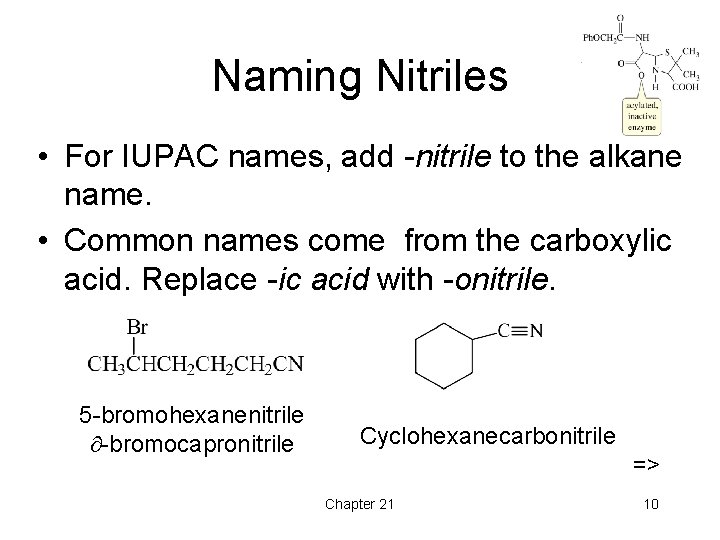
Naming Nitriles • For IUPAC names, add -nitrile to the alkane name. • Common names come from the carboxylic acid. Replace -ic acid with -onitrile. 5 -bromohexanenitrile -bromocapronitrile Cyclohexanecarbonitrile => Chapter 21 10

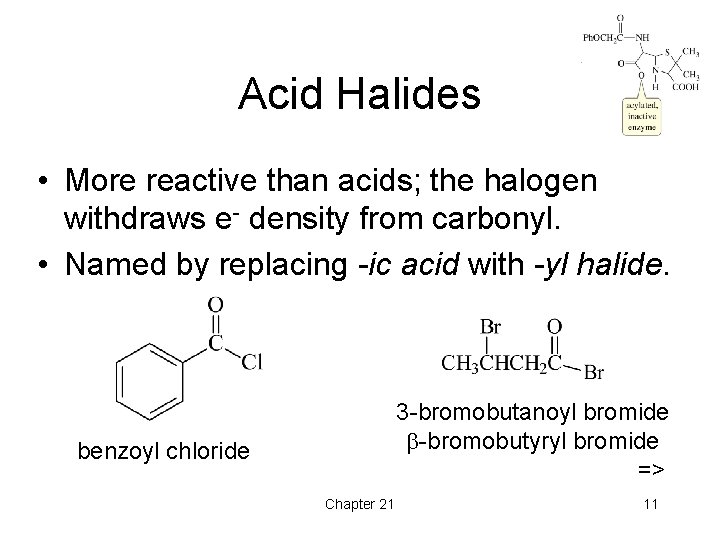
Acid Halides • More reactive than acids; the halogen withdraws e- density from carbonyl. • Named by replacing -ic acid with -yl halide. 3 -bromobutanoyl bromide -bromobutyryl bromide => benzoyl chloride Chapter 21 11

Acid Anhydrides • Two molecules of acid combine with the loss of water to form the anhydride. • Anhydrides are more reactive than acids, but less reactive than acid chlorides. • A carboxylate ion is the leaving group in nucleophilic acyl substitution reactions. => Chapter 21 12

Naming Anhydrides • The word acid is replaced with anhydride. • For a mixed anhydride, name both acids. • Diacids may form anhydrides if a 5 - or 6 membered ring is the product. ethanoic anhydride acetic anhydride 1, 2 -benzenedicarboxylic anhydride phthalic anhydride => Chapter 21 13

Multifunctional Compounds • The functional group with the highest priority determines the parent name. • Acid > ester > amide > nitrile > aldehyde > ketone > alcohol > amine > alkene > alkyne. ethyl o-cyanobenzoate => Chapter 21 14

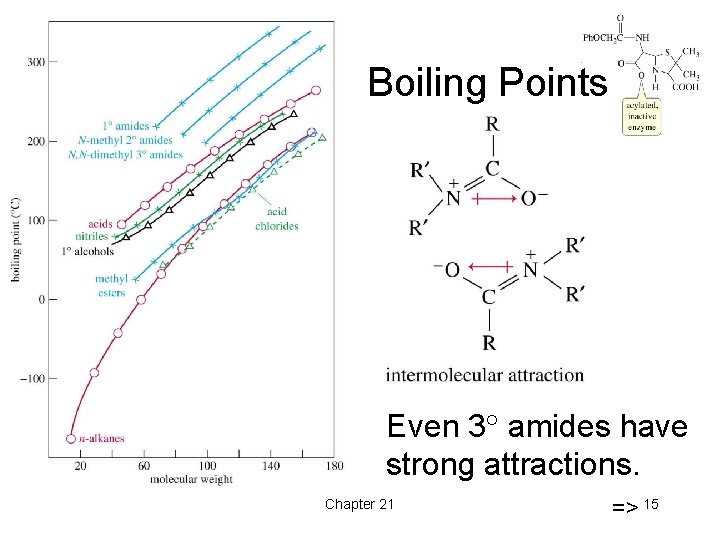
Boiling Points Even 3 amides have strong attractions. Chapter 21 => 15

Melting Points • Amides have very high melting points. • Melting points increase with increasing number of N-H bonds. m. p. -61 C m. p. 28 C m. p. 79 C => Chapter 21 16

Solubility • Acid chlorides and anhydrides are too reactive to be used with water or alcohol. • Esters, 3 amides, and nitriles are good polar aprotic solvents. • Solvents commonly used in organic reactions: ØEthyl acetate ØDimethylformamide (DMF) ØAcetonitrile Chapter 21 =>17

IR Spectroscopy => => Chapter 21 18

1 H NMR Spectroscopy => Chapter 21 19

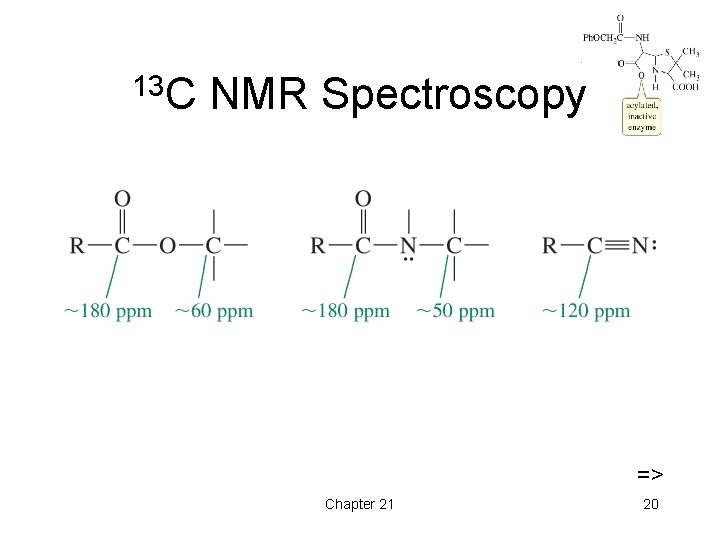
13 C NMR Spectroscopy => Chapter 21 20

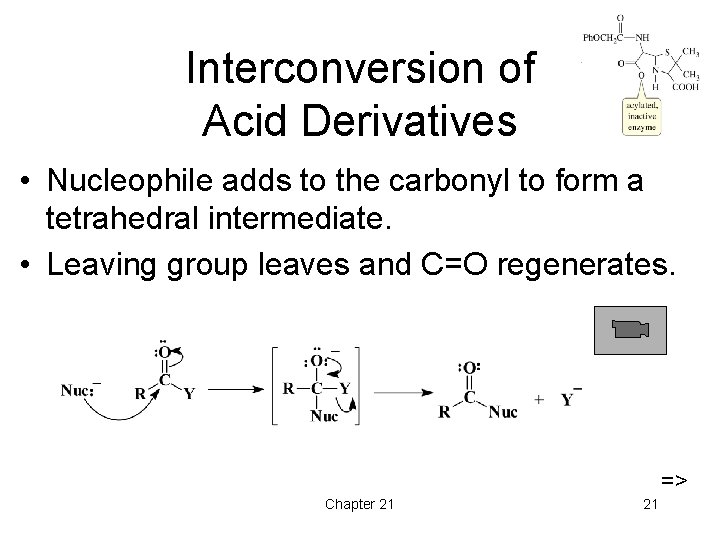
Interconversion of Acid Derivatives • Nucleophile adds to the carbonyl to form a tetrahedral intermediate. • Leaving group leaves and C=O regenerates. => Chapter 21 21

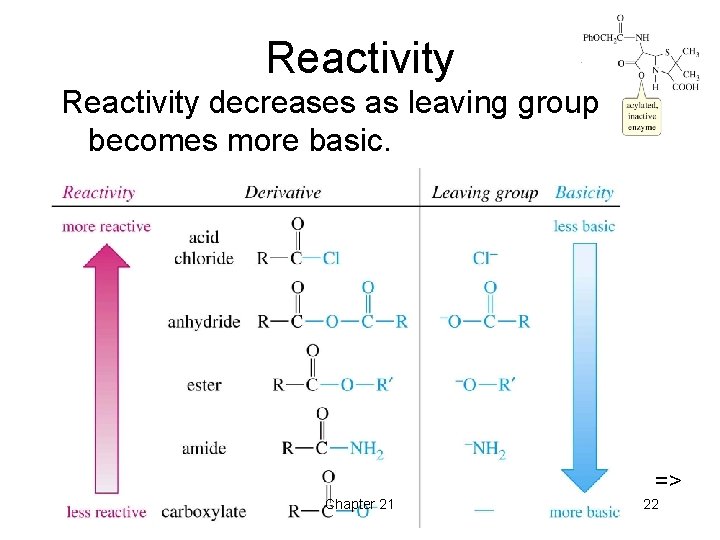
Reactivity decreases as leaving group becomes more basic. => Chapter 21 22

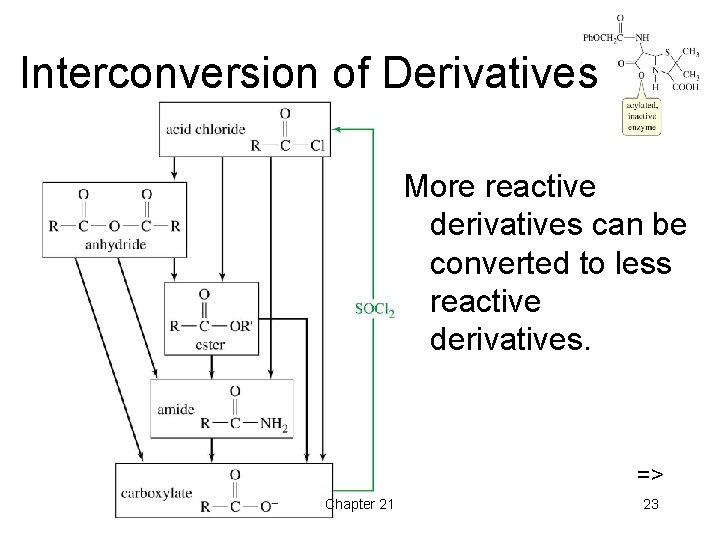
Interconversion of Derivatives More reactive derivatives can be converted to less reactive derivatives. => Chapter 21 23

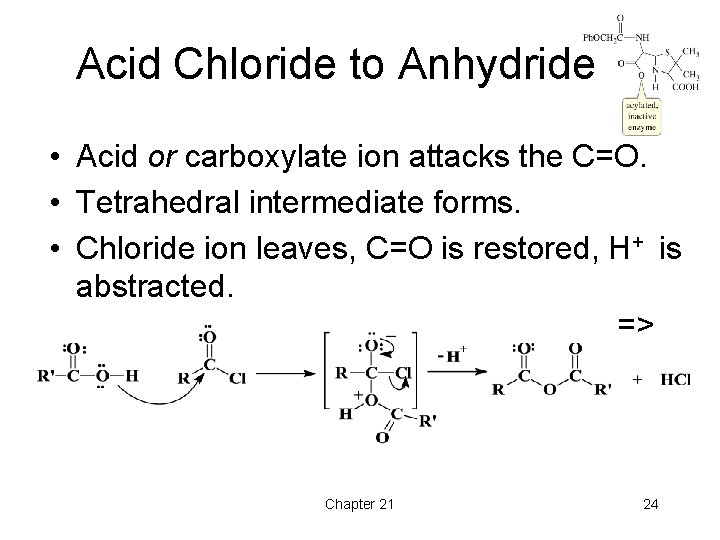
Acid Chloride to Anhydride • Acid or carboxylate ion attacks the C=O. • Tetrahedral intermediate forms. • Chloride ion leaves, C=O is restored, H+ is abstracted. => Chapter 21 24

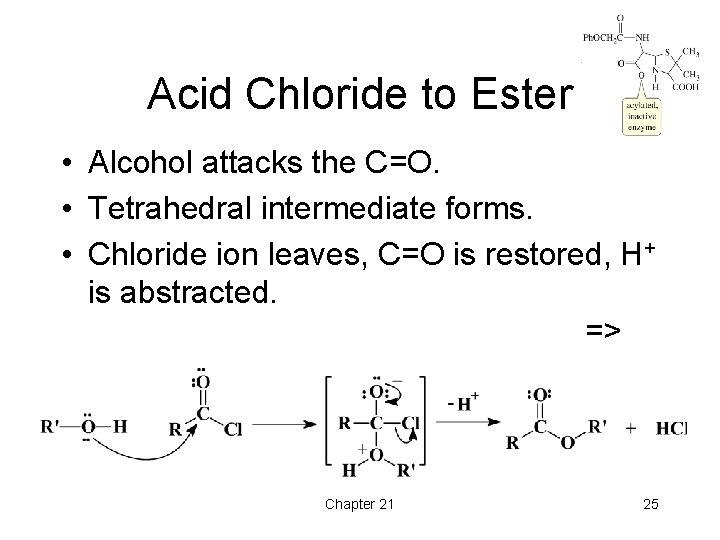
Acid Chloride to Ester • Alcohol attacks the C=O. • Tetrahedral intermediate forms. • Chloride ion leaves, C=O is restored, H+ is abstracted. => Chapter 21 25

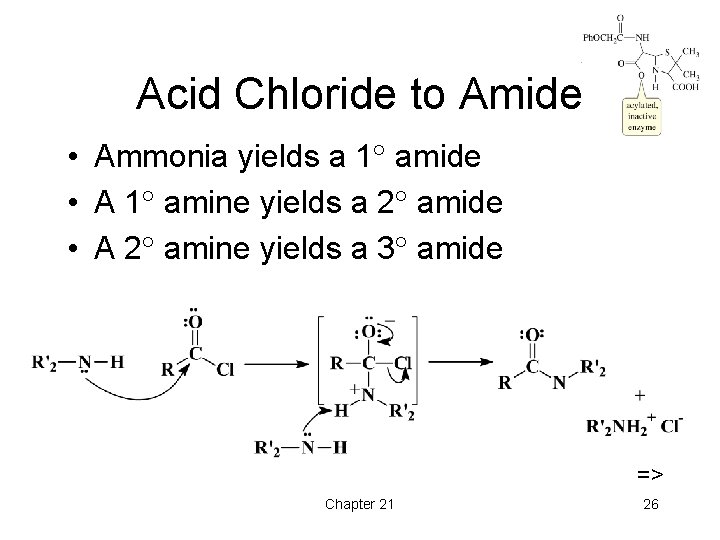
Acid Chloride to Amide • Ammonia yields a 1 amide • A 1 amine yields a 2 amide • A 2 amine yields a 3 amide => Chapter 21 26

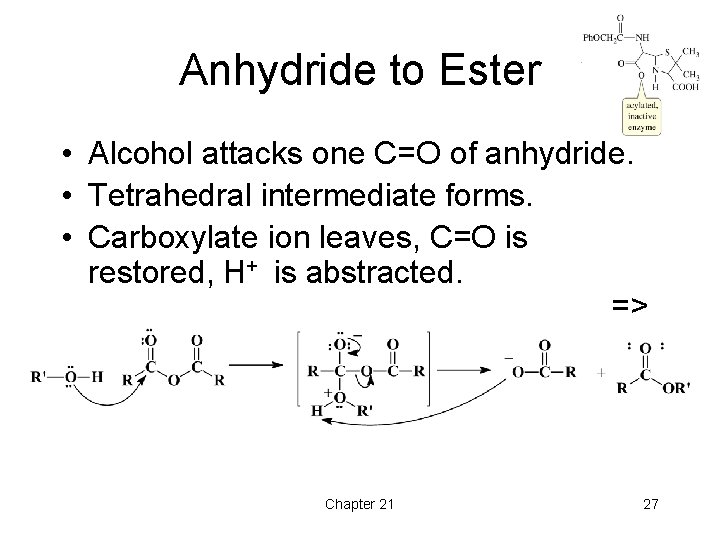
Anhydride to Ester • Alcohol attacks one C=O of anhydride. • Tetrahedral intermediate forms. • Carboxylate ion leaves, C=O is restored, H+ is abstracted. => Chapter 21 27

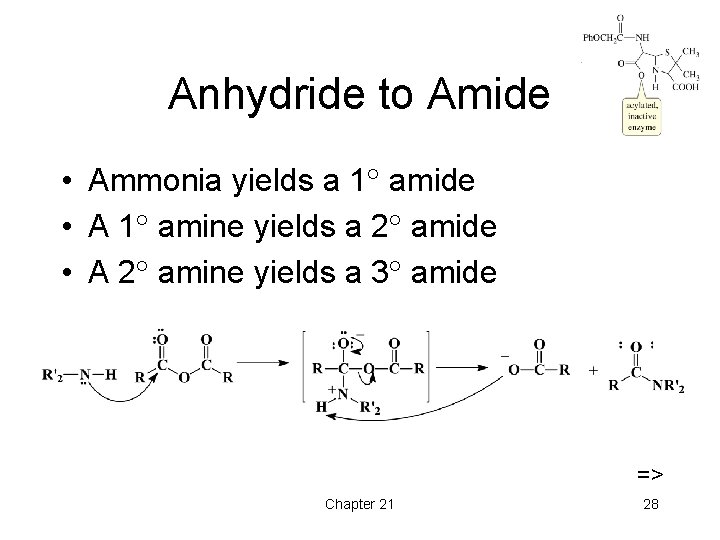
Anhydride to Amide • Ammonia yields a 1 amide • A 1 amine yields a 2 amide • A 2 amine yields a 3 amide => Chapter 21 28

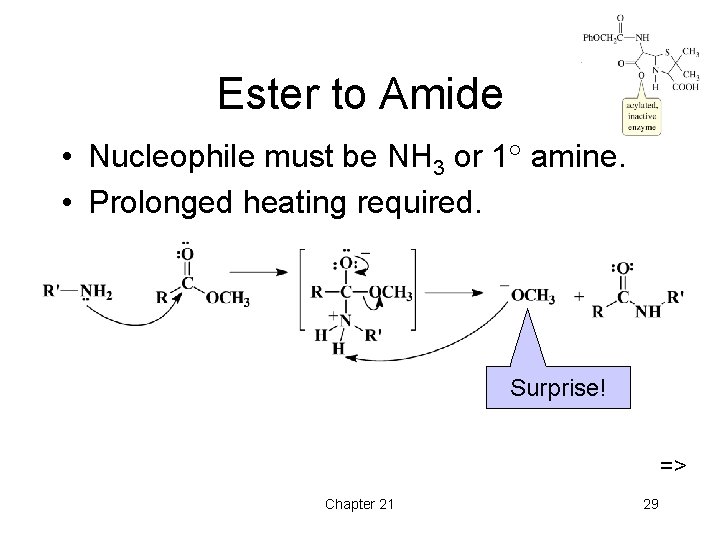
Ester to Amide • Nucleophile must be NH 3 or 1 amine. • Prolonged heating required. Surprise! => Chapter 21 29

Leaving Groups A strong base is not usually a leaving group unless it’s in an exothermic step. => Chapter 21 30

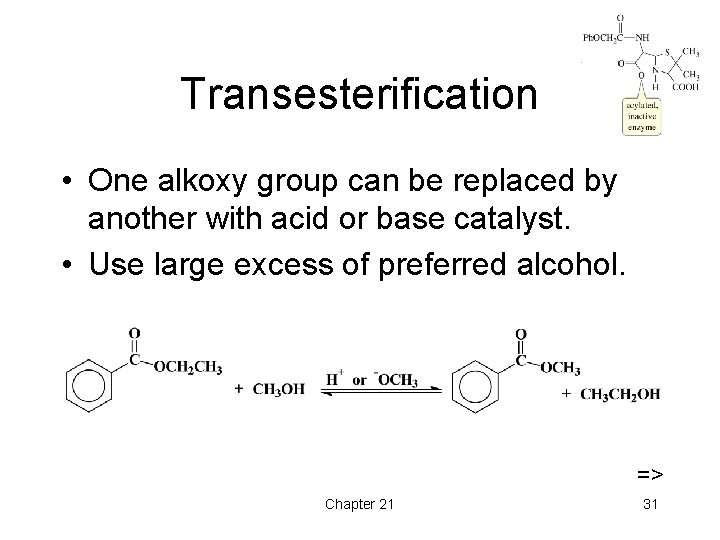
Transesterification • One alkoxy group can be replaced by another with acid or base catalyst. • Use large excess of preferred alcohol. => Chapter 21 31

Hydrolysis of Acid Chlorides and Anhydrides • Hydrolysis occurs quickly, even in moist air with no acid or base catalyst. • Reagents must be protected from moisture. => Chapter 21 32

Acid Hydrolysis of Esters • Reverse of Fischer esterification. • Reaches equilibrium. • Use a large excess of water. => Chapter 21 33

Saponification • Base-catalyzed hydrolysis of ester. • “Saponification” means “soap-making. ” • Soaps are made by heating Na. OH with a fat (triester of glycerol) to produce the sodium salt of a fatty acid - a soap. • One example of a soap is sodium stearate, Na+ -OOC(CH 2)16 CH 3. => Chapter 21 34

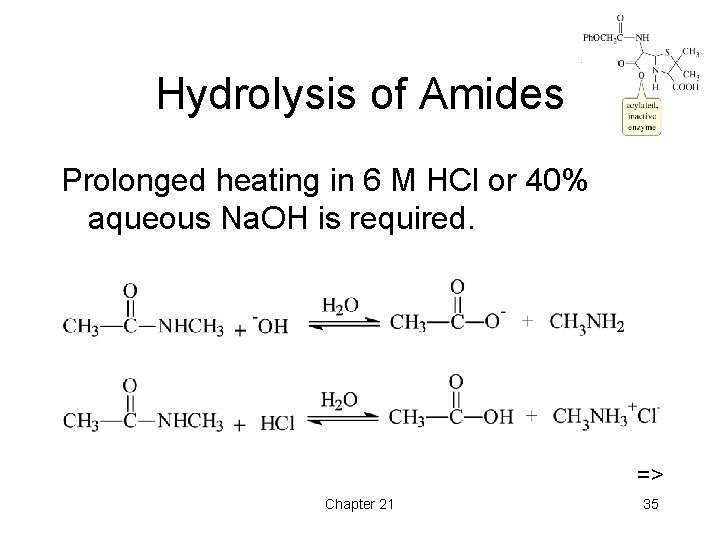
Hydrolysis of Amides Prolonged heating in 6 M HCl or 40% aqueous Na. OH is required. => Chapter 21 35

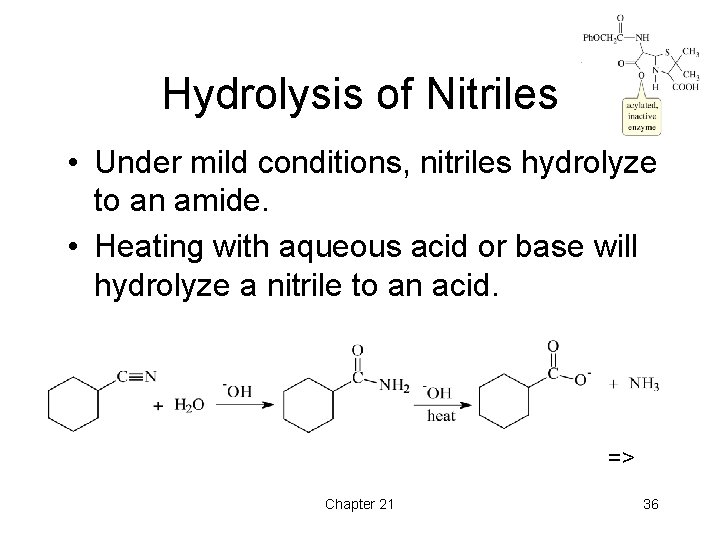
Hydrolysis of Nitriles • Under mild conditions, nitriles hydrolyze to an amide. • Heating with aqueous acid or base will hydrolyze a nitrile to an acid. => Chapter 21 36

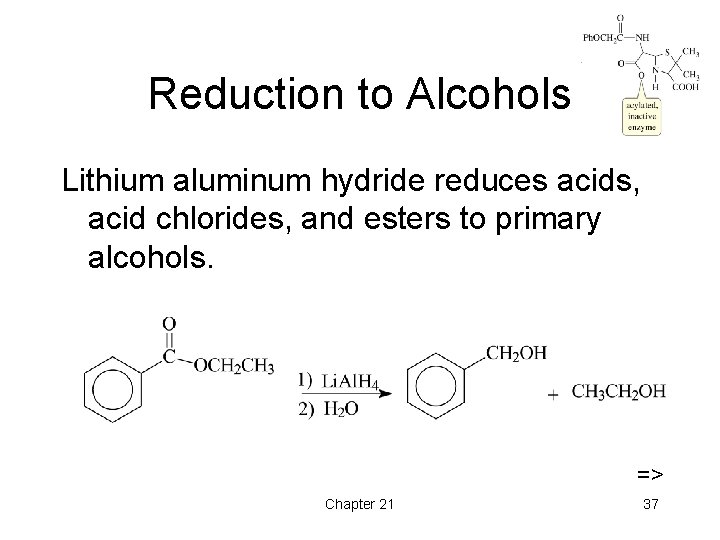
Reduction to Alcohols Lithium aluminum hydride reduces acids, acid chlorides, and esters to primary alcohols. => Chapter 21 37

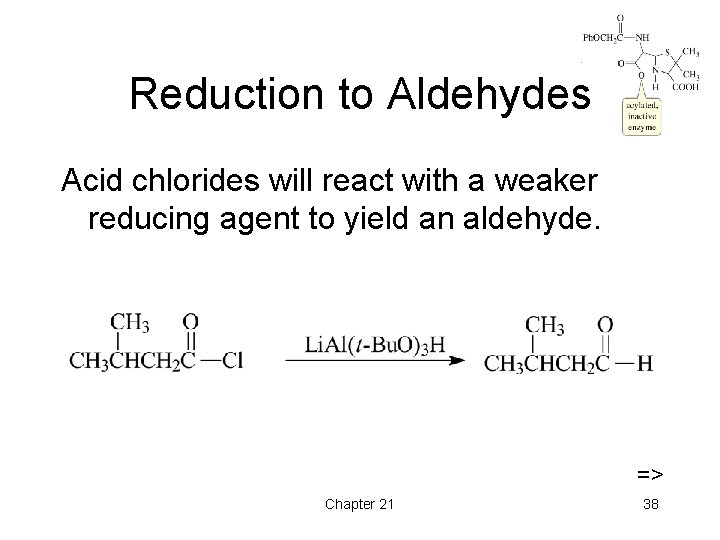
Reduction to Aldehydes Acid chlorides will react with a weaker reducing agent to yield an aldehyde. => Chapter 21 38

Reduction to Amines • Lithium aluminum hydride reduces amides and nitriles to amines. • Nitriles and 1 amides reduce to 1 amines. • A 2 amide reduces to a 2 amine. • A 3 amide reduces to a 3 amine. => Chapter 21 39

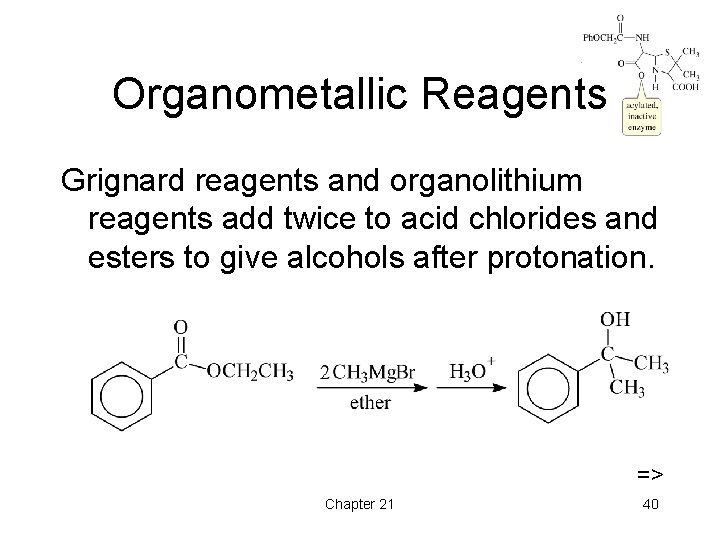
Organometallic Reagents Grignard reagents and organolithium reagents add twice to acid chlorides and esters to give alcohols after protonation. => Chapter 21 40

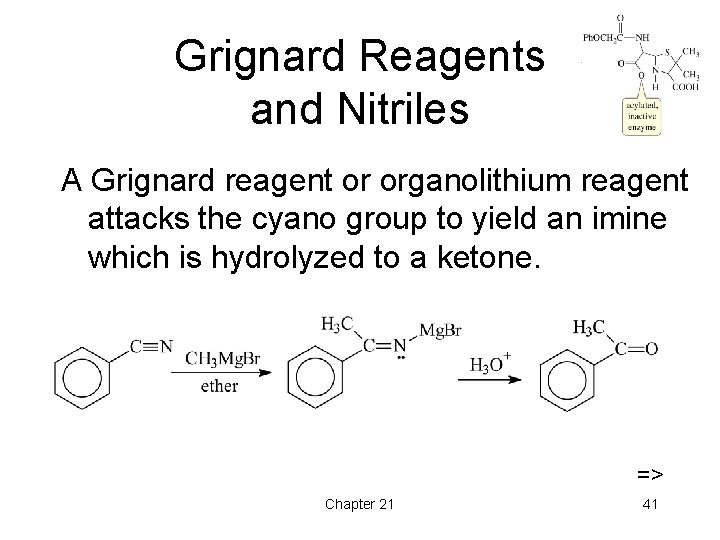
Grignard Reagents and Nitriles A Grignard reagent or organolithium reagent attacks the cyano group to yield an imine which is hydrolyzed to a ketone. => Chapter 21 41

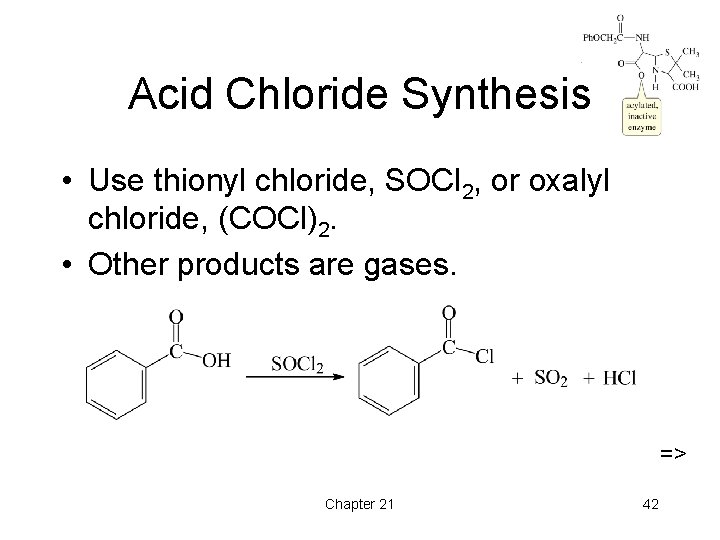
Acid Chloride Synthesis • Use thionyl chloride, SOCl 2, or oxalyl chloride, (COCl)2. • Other products are gases. => Chapter 21 42

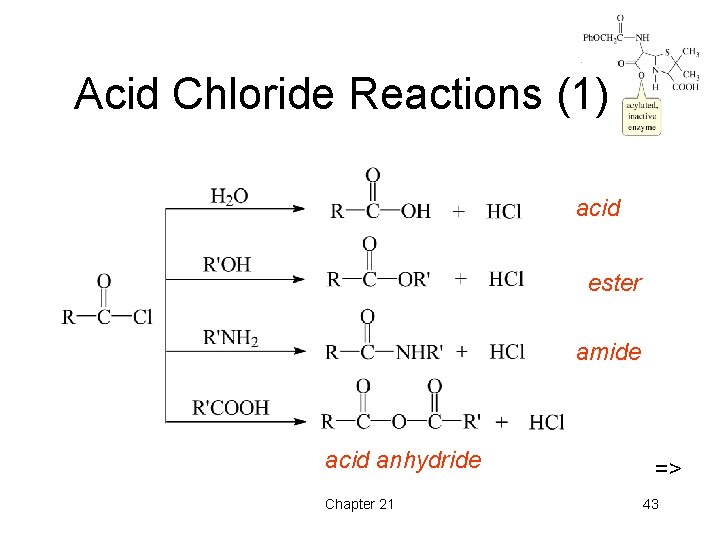
Acid Chloride Reactions (1) acid ester amide acid anhydride Chapter 21 => 43

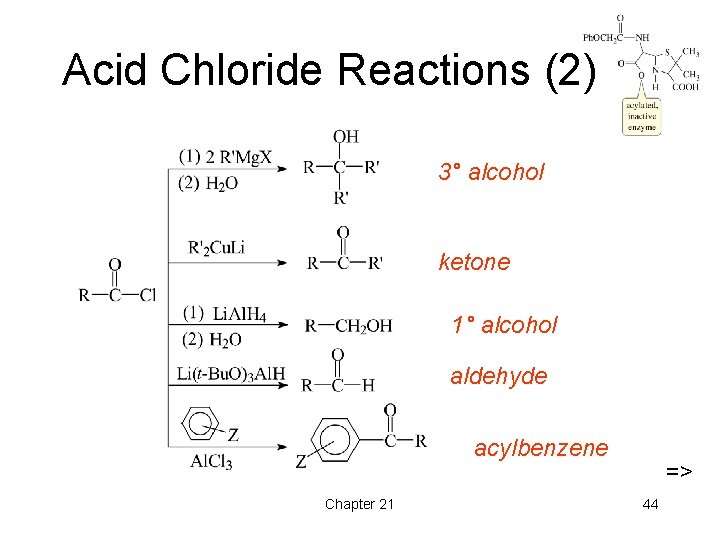
Acid Chloride Reactions (2) 3° alcohol ketone 1° alcohol aldehyde acylbenzene Chapter 21 => 44

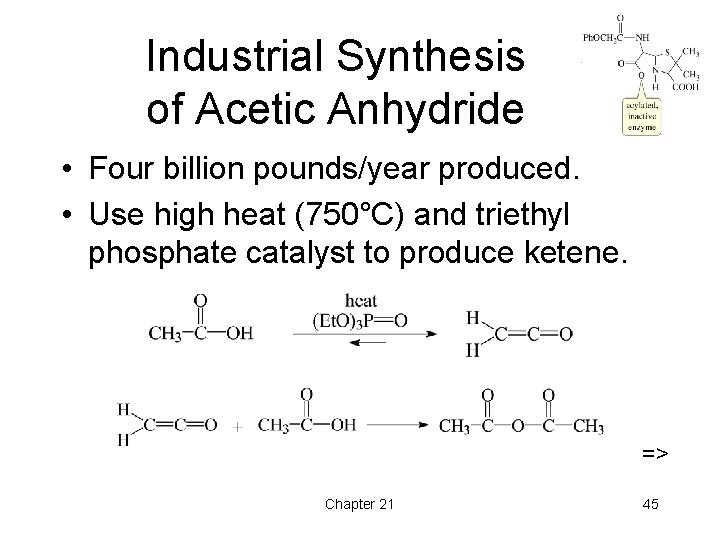
Industrial Synthesis of Acetic Anhydride • Four billion pounds/year produced. • Use high heat (750°C) and triethyl phosphate catalyst to produce ketene. => Chapter 21 45

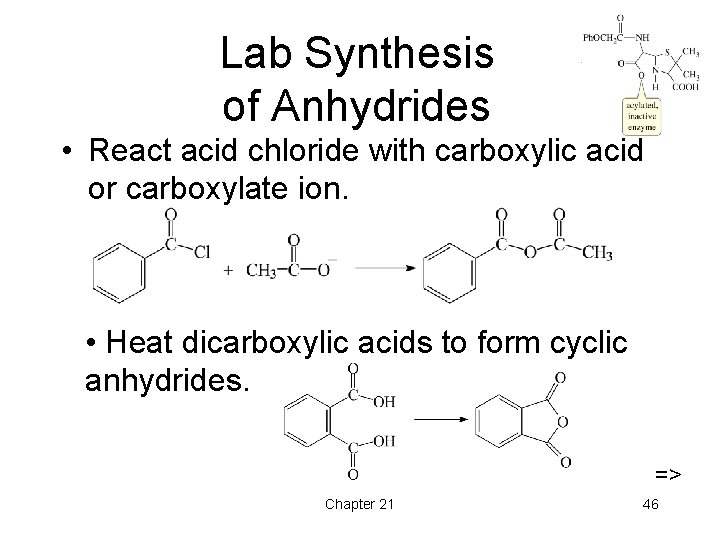
Lab Synthesis of Anhydrides • React acid chloride with carboxylic acid or carboxylate ion. • Heat dicarboxylic acids to form cyclic anhydrides. => Chapter 21 46

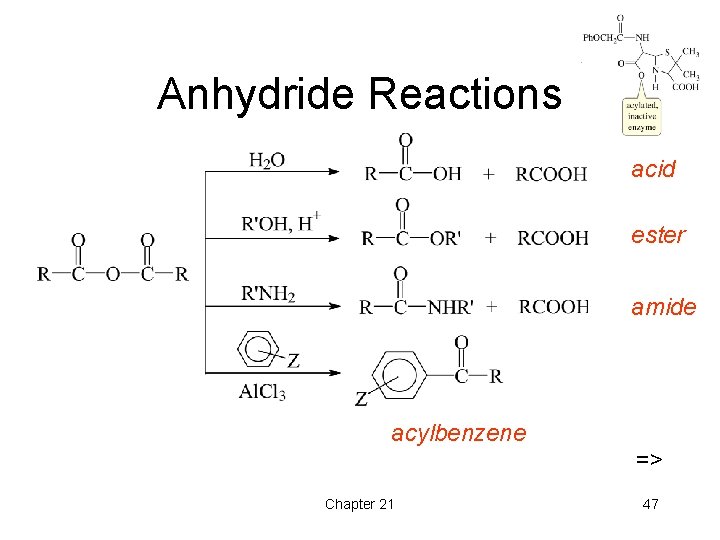
Anhydride Reactions acid ester amide acylbenzene => Chapter 21 47

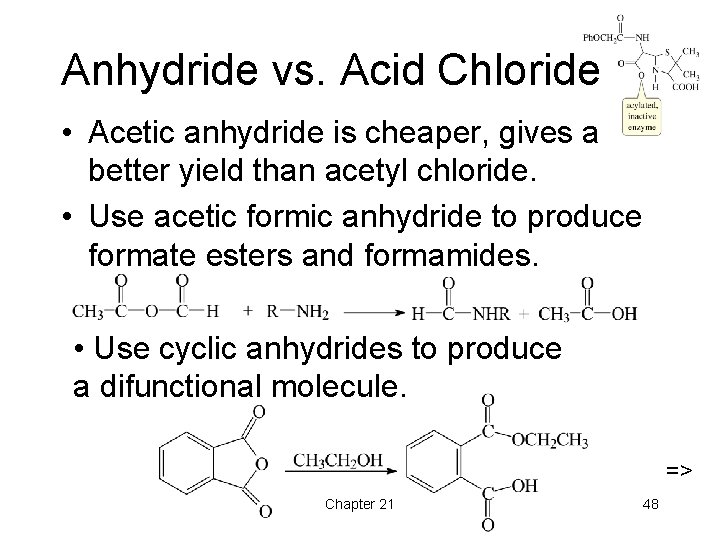
Anhydride vs. Acid Chloride • Acetic anhydride is cheaper, gives a better yield than acetyl chloride. • Use acetic formic anhydride to produce formate esters and formamides. • Use cyclic anhydrides to produce a difunctional molecule. => Chapter 21 48

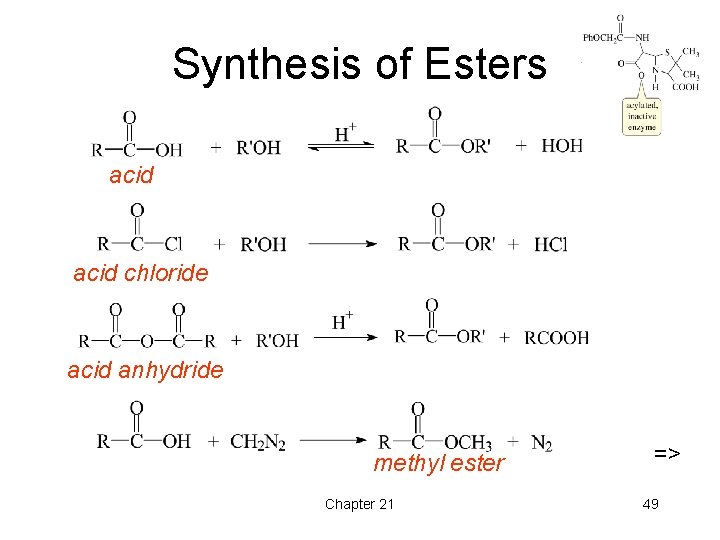
Synthesis of Esters acid chloride acid anhydride methyl ester Chapter 21 => 49

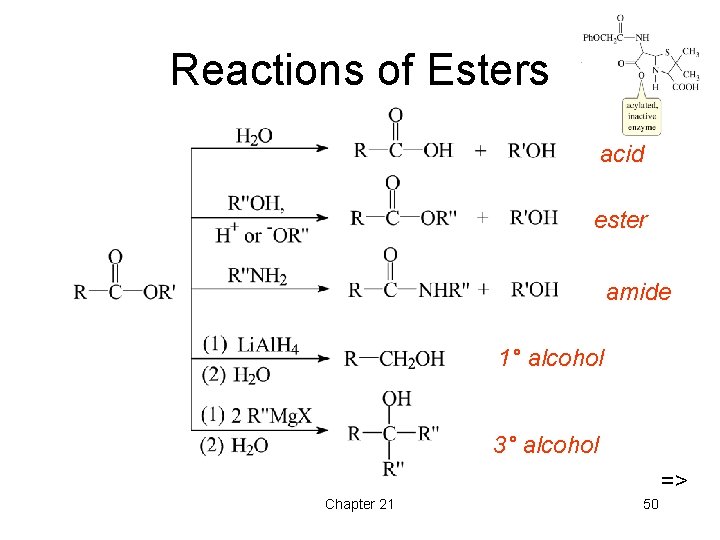
Reactions of Esters acid ester amide 1° alcohol 3° alcohol => Chapter 21 50

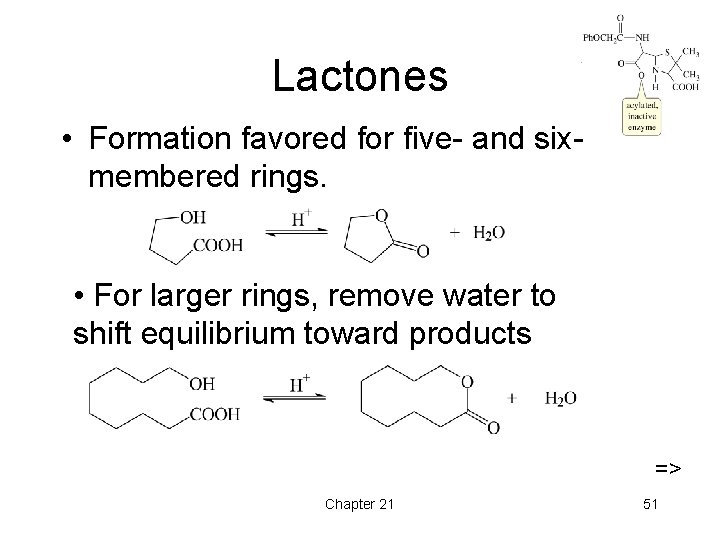
Lactones • Formation favored for five- and sixmembered rings. • For larger rings, remove water to shift equilibrium toward products => Chapter 21 51

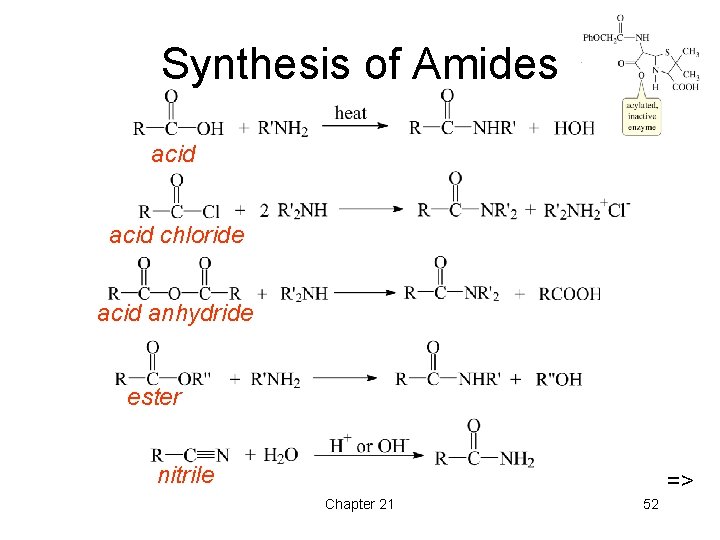
Synthesis of Amides acid chloride acid anhydride ester nitrile => Chapter 21 52

Reactions of Amides acid and amine 1° amine nitrile => Chapter 21 53

Lactam Formation • Five- and six-membered rings can be formed by heating - and -amino acids. • Smaller or larger rings do not form readily. => Chapter 21 54

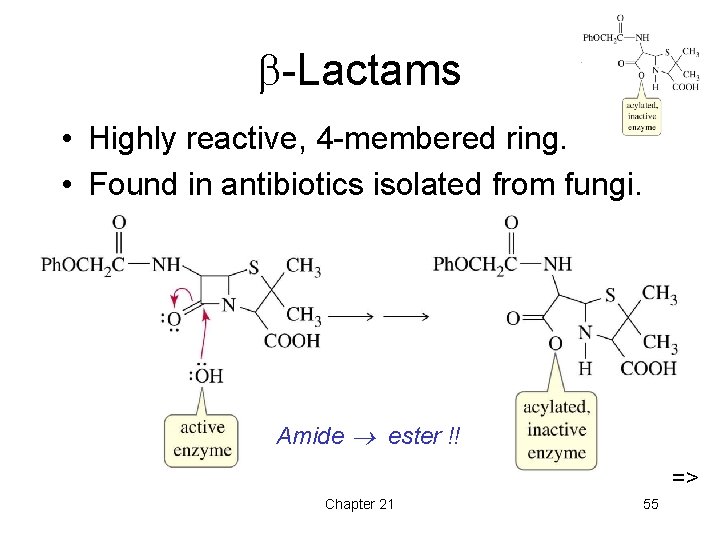
-Lactams • Highly reactive, 4 -membered ring. • Found in antibiotics isolated from fungi. Amide ester !! => Chapter 21 55

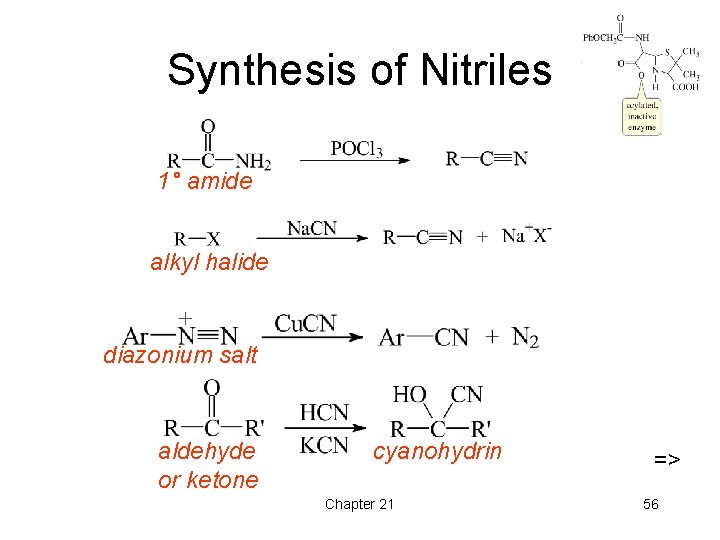
Synthesis of Nitriles 1° amide alkyl halide diazonium salt aldehyde or ketone cyanohydrin Chapter 21 => 56

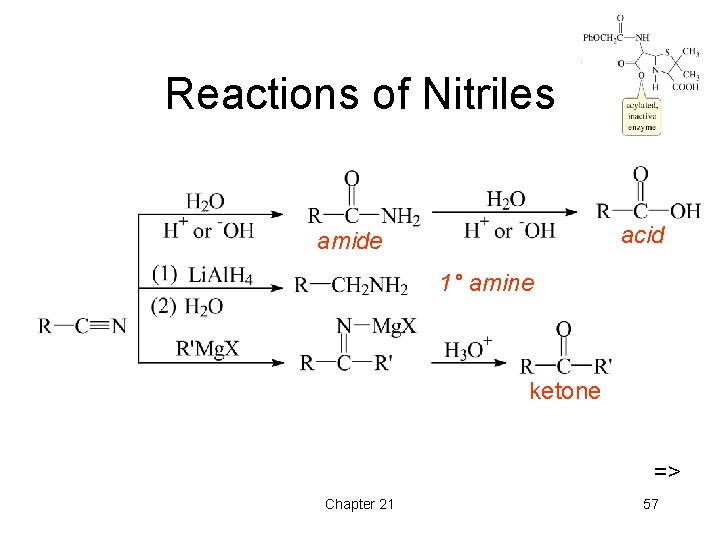
Reactions of Nitriles acid amide 1° amine ketone => Chapter 21 57

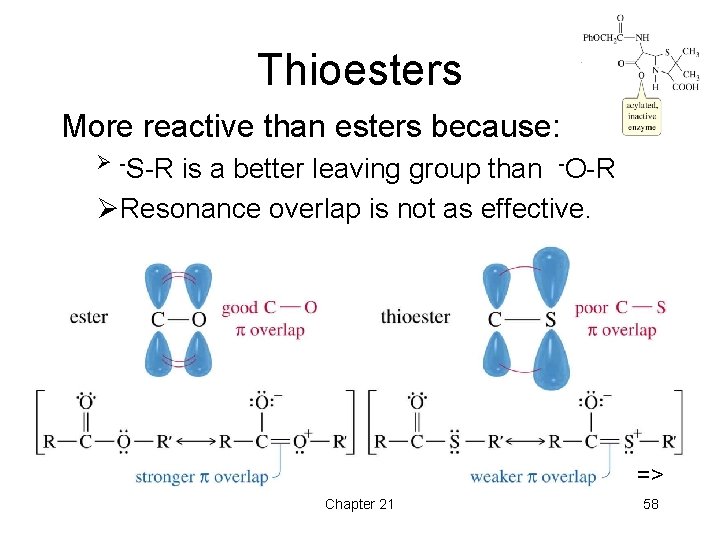
Thioesters More reactive than esters because: Ø -S-R is a better leaving group than -O-R ØResonance overlap is not as effective. => Chapter 21 58

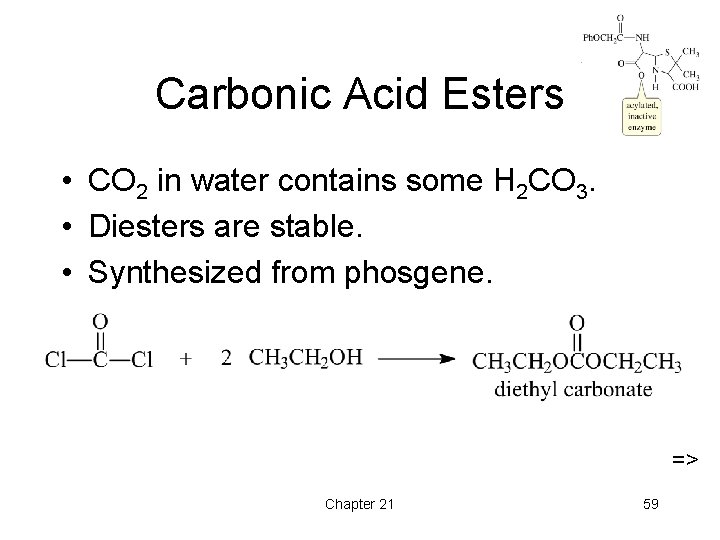
Carbonic Acid Esters • CO 2 in water contains some H 2 CO 3. • Diesters are stable. • Synthesized from phosgene. => Chapter 21 59

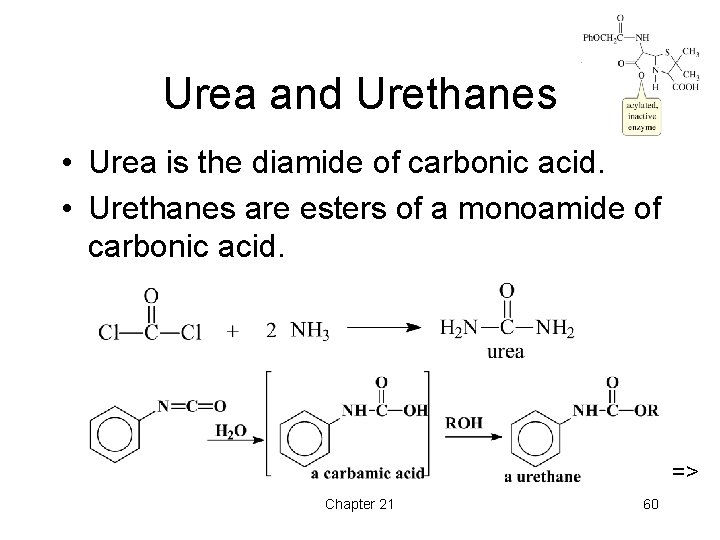
Urea and Urethanes • Urea is the diamide of carbonic acid. • Urethanes are esters of a monoamide of carbonic acid. => Chapter 21 60

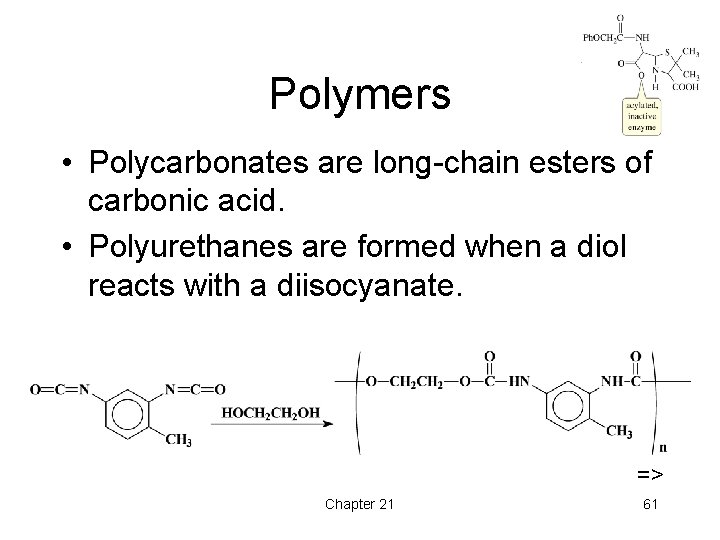
Polymers • Polycarbonates are long-chain esters of carbonic acid. • Polyurethanes are formed when a diol reacts with a diisocyanate. => Chapter 21 61

End of Chapter 21 62