Organic Chemistry What is organic chemistry organic carbon

Organic Chemistry

What is “organic” chemistry? � organic – carbon compound ◦ Includes the substances of “life” found in your body � Hydrocarbons – compounds made of H + C � Saturated – Carbons have all single bonds ◦ Fats that are solids at room temp. � Unsaturated bonds – Carbons have double and/or triple ◦ Fats that are liquids at room temp.

Info about Carbon � always forms 4 bonds ◦ because it has 4 valence electrons � very stable compounds � Examples: ◦ Pharmaceuticals (aspirin, vitamins, insulin) ◦ Petrochemicals (propane, butane, & octane) �AKA: hydrocarbons

SOL Released Question

Carbon Chains Prefix 1) Meth 2) Eth 3) Prop 4) But 5) Pent 6) Hex 7) Hept 8) Oct Suffix -ane single bonds -ene double bond -yne triple bond

Saturated vs. Unsaturated Carbon Chains
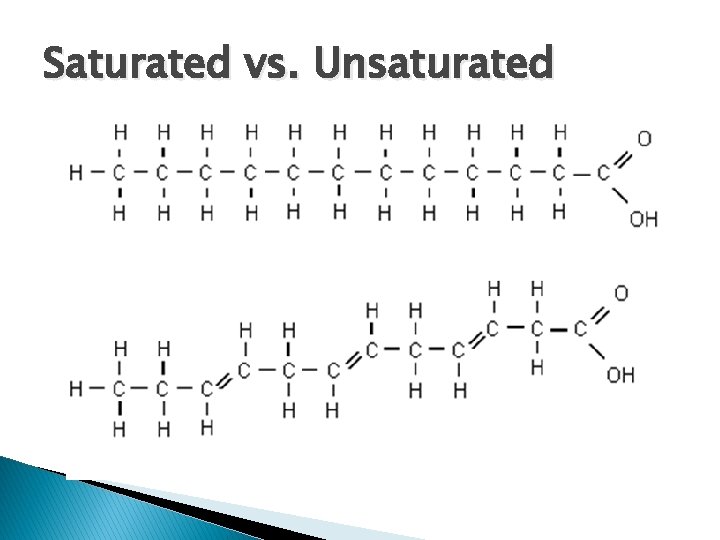
Saturated vs. Unsaturated

� Saturated ◦ Single bonds to carbon � Unsaturated ◦ Double or triple bond to carbon
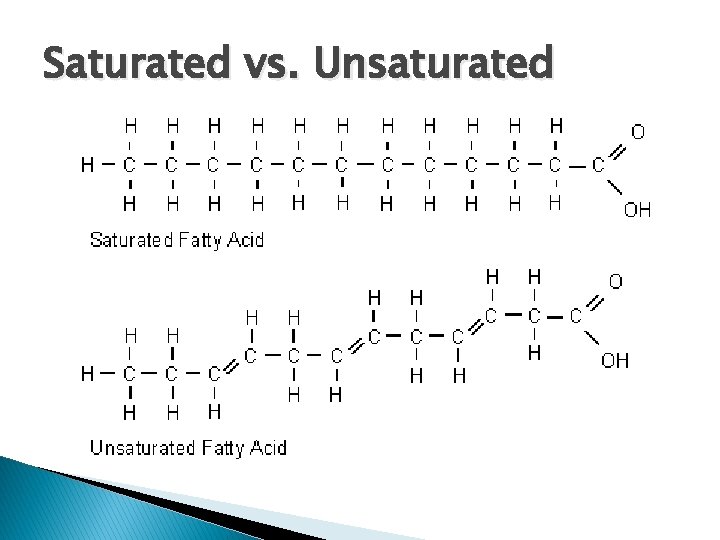
Saturated vs. Unsaturated
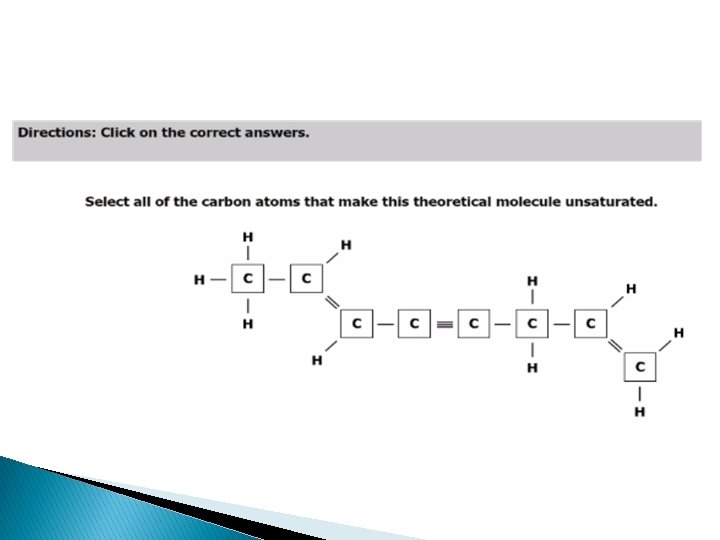
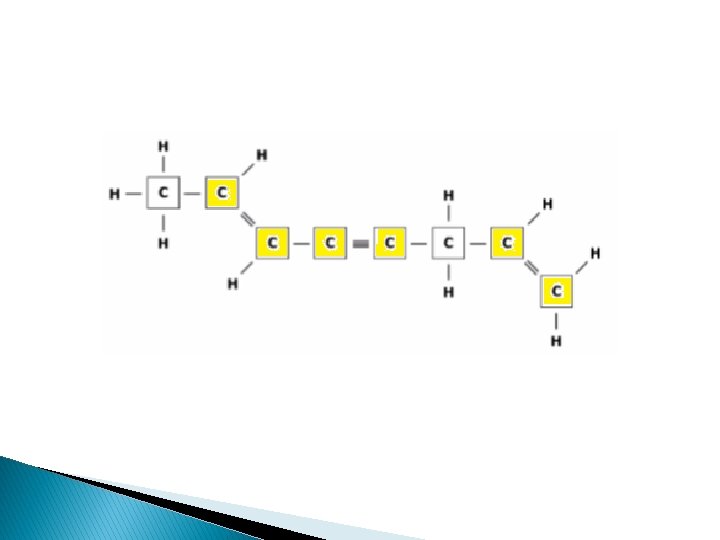

SOL Released Question

Polymers An example of carbon chains.


Natural Polymers � Carbohydrates � Nucleic ◦ DNA ◦ RNA Acids � Proteins
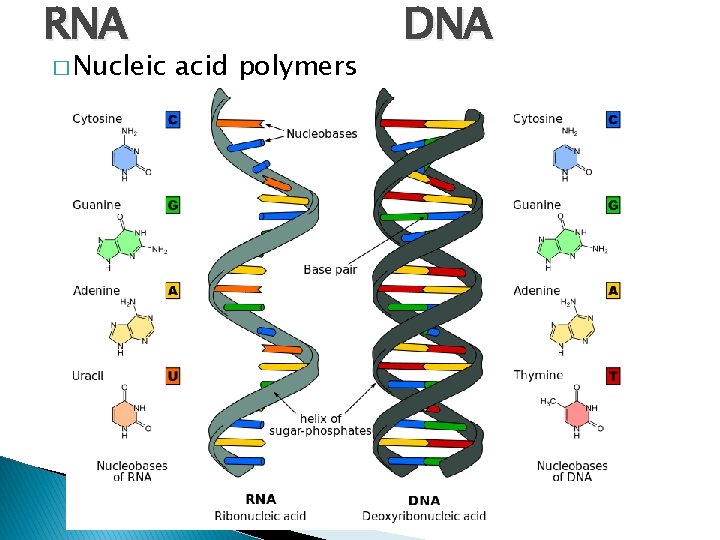
RNA � Nucleic acid polymers DNA
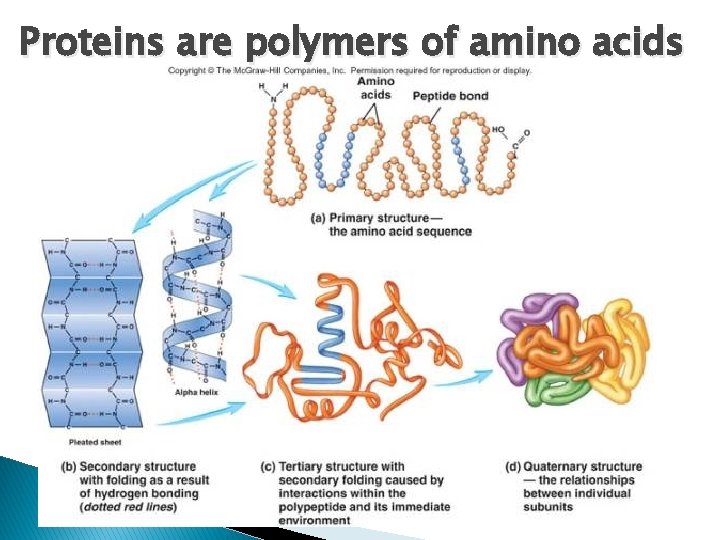
Proteins are polymers of amino acids

Synthetic Polymers � All plastics ◦ Made from petroleum ◦ Long chains of carbon monomers � monomer + monomer = polymer ◦ “polymerization reaction”

Examples of Synthetic Polymers � Nylon � Polyester � Teflon � Kevlar � Plastics!!

SOL Released Question

SOL Released Question
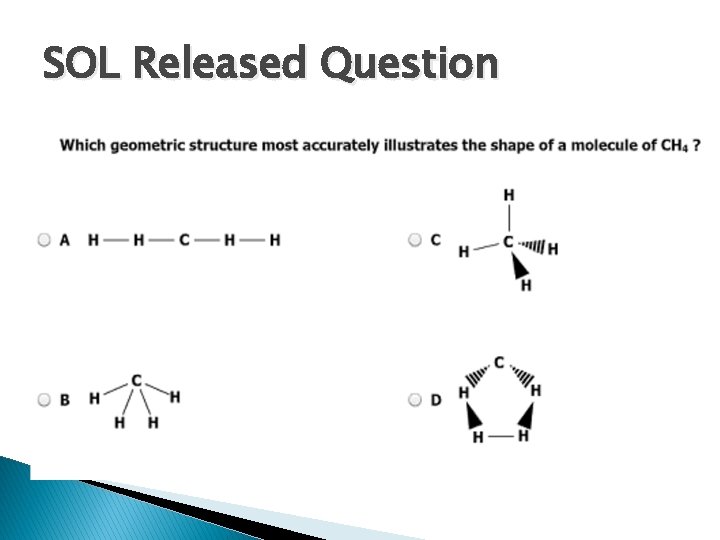
SOL Released Question
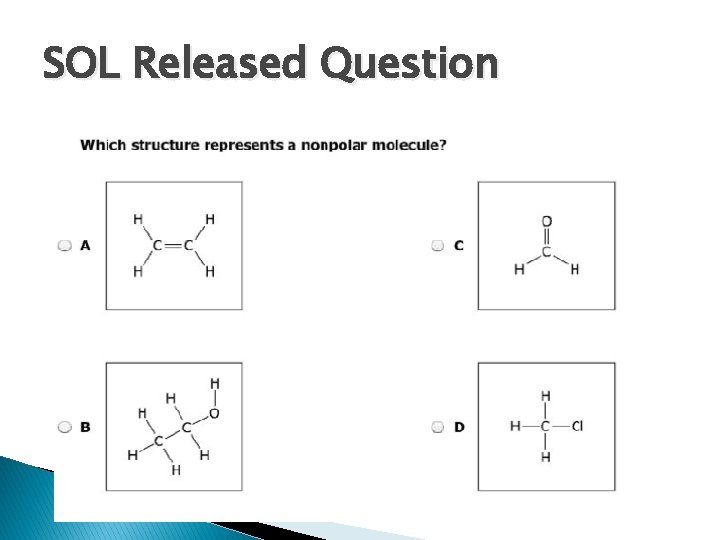
SOL Released Question

Lab Molecular Model Kits

Build the following: 1) 2) 3) 4) 5) 6) 1, 3 -hexene and 3, 5 -hexene � Compare the two structures. Ethanoic acid and Ethanal � Compare the two structures. 2, 3 -pentanol Methane 2 -butanone � Is the following an accurate representation? 4 -pentyne Red = oxygen white = hydrogen Black = carbon

Build the following: 1) 2) 3) 4) Ethanoic acid and Ethanal � Compare the two structures. 2, 3 -pentanol � What orientation will the molecule spend most of its time in? 2 -butanone � Is the following an accurate representation? 4 -pentyne � How does the triple bond affect the structure? Red = oxygen white = hydrogen Black = carbon

Build the following: 1) 2) 3) 4) Butane Methane 1, 3 -pentene Ethyne white = hydrogen Black = carbon
- Slides: 27