The Art of Writing Reasonable Organic Reaction Mechanisms

The Art of Writing Reasonable Organic Reaction Mechanisms Robert B. Grossman University of Kentucky Professor of Chemistry Synthetic Organic Chemistry 1987 B. A. , Princeton University 1992 Ph. D. , Massachusetts Institute of Technology 1992 -1994 Post-doctoral Fellow, Cambridge University

課程須知 參考書目 Robert B. Grossman University of Kentucky Organic Synthesis Michael B. Smith University of Connecticut Organic Reaction Mechanisms A. C. Knipe University of Ulster


課程須知 Mechanisms are the means by which organic reactions are discovered, rationalized, optimized, and incorporated into the canon. They represent the framework that allows us to understand organic chemistry. The purpose of this book is to help you learn how to draw reasonable mechanisms for organic reactions. The general approach is to familiarize you with the classes and types of reaction mechanisms that are known and to give you the tools to learn how to draw mechanisms for reactions that you have never seen before. *Common error alerts are scattered throughout the text to warn you about common pitfalls and misconceptions that bedevil students. Pay attention to these alerts, as failure to observe their strictures has caused many, many exam points to be lost over the years.

Chapter One The Basics of Organic Chemistry
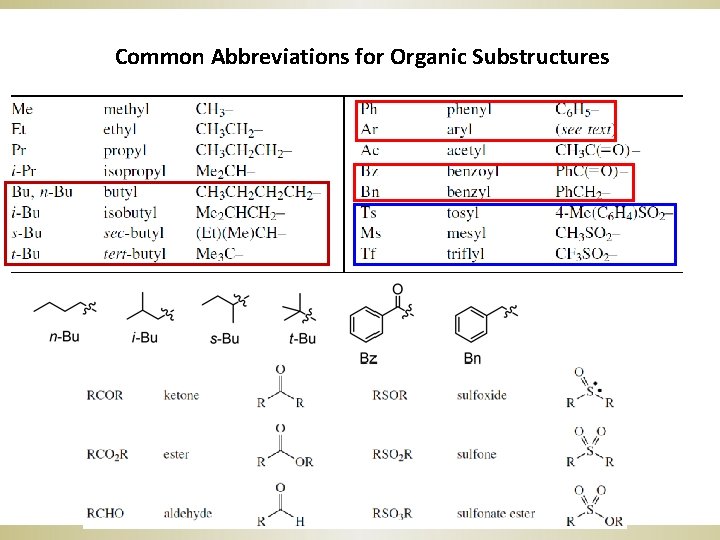
Common Abbreviations for Organic Substructures

Formal Charge Formal charges are a useful tool for ensuring that electrons are not gained or lost in the course of a reaction. EX: Calculate the formal charge of Al, N, Br, C *Common error alert: Formal charges are not a reliabe guide to chemical reactivity. CH 3+ 6 e- NH 4+ which is more stable? 8 e-
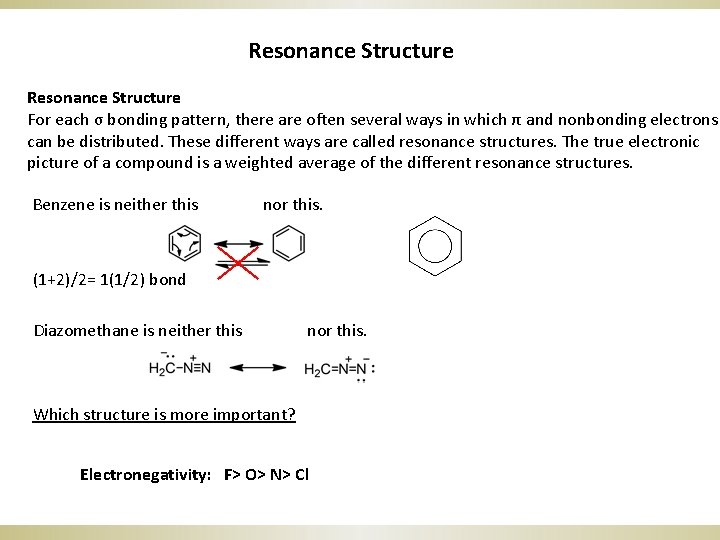
Resonance Structure For each σ bonding pattern, there are often several ways in which π and nonbonding electrons can be distributed. These different ways are called resonance structures. The true electronic picture of a compound is a weighted average of the different resonance structures. Benzene is neither this nor this. (1+2)/2= 1(1/2) bond Diazomethane is neither this nor this. Which structure is more important? Electronegativity: F> O> N> Cl
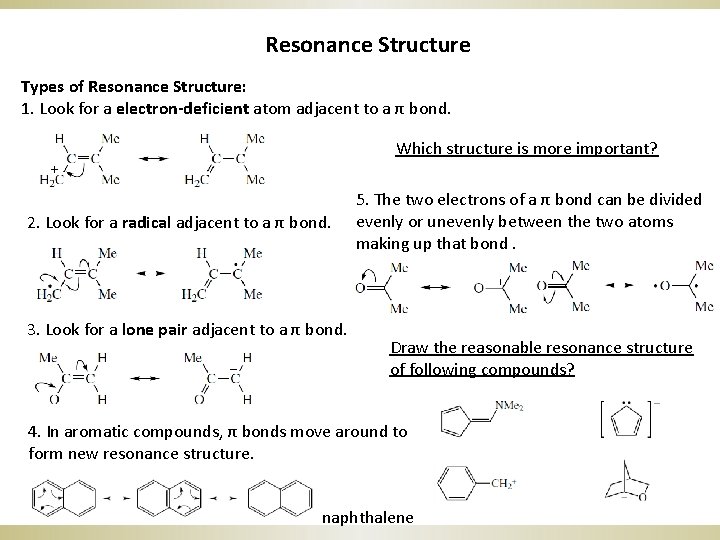
Resonance Structure Types of Resonance Structure: 1. Look for a electron-deficient atom adjacent to a π bond. Which structure is more important? 2. Look for a radical adjacent to a π bond. 3. Look for a lone pair adjacent to a π bond. 5. The two electrons of a π bond can be divided evenly or unevenly between the two atoms making up that bond. Draw the reasonable resonance structure of following compounds? 4. In aromatic compounds, π bonds move around to form new resonance structure. naphthalene
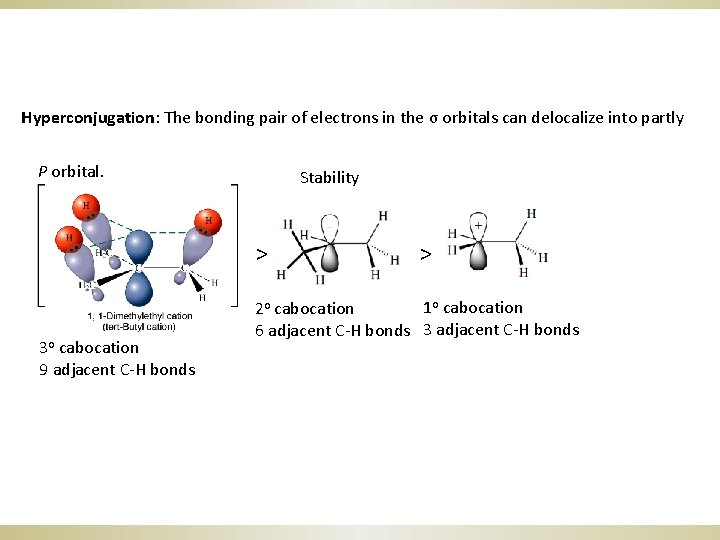
Hyperconjugation: The bonding pair of electrons in the σ orbitals can delocalize into partly P orbital. Stability > 3 o cabocation 9 adjacent C-H bonds > 1 o cabocation 2 o cabocation 6 adjacent C-H bonds 3 adjacent C-H bonds

Molecular Shape: Lewis Structure and Molecular Orbitals Lewis Structure 1. 劃出分子結構,並使所有原子符合八隅體(H例外),統計其電子數A 2. 計算所有原子的價電子數總和B 3. (A- B)/2為共用電子對,一般形成共用電子對的方法為環、雙鍵或三鍵 Linear Trigonal Tetrahedron SP SP 2 SP 3 eg. COCl 2 A= 26 B= 4+6+(7*2)= 24 (26 -24)/2= 1 SP 2 trigonal Draw the Lewis structure for following molecules and determinate the molecular shape? HI, C 22 -, CH 3 OH, Si. O 2, CS 2, CN-
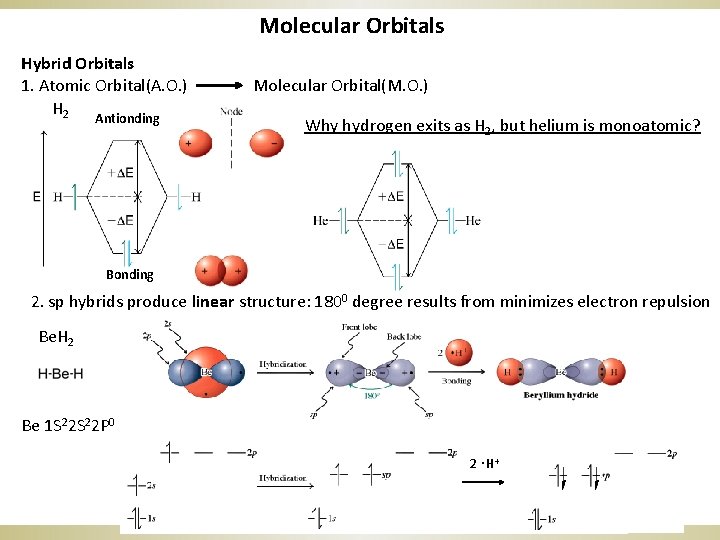
Molecular Orbitals Hybrid Orbitals 1. Atomic Orbital(A. O. ) H 2 Antionding Molecular Orbital(M. O. ) Why hydrogen exits as H 2, but helium is monoatomic? Bonding 2. sp hybrids produce linear structure: 1800 degree results from minimizes electron repulsion Be. H 2 Be 1 S 22 P 0 . 2 H+
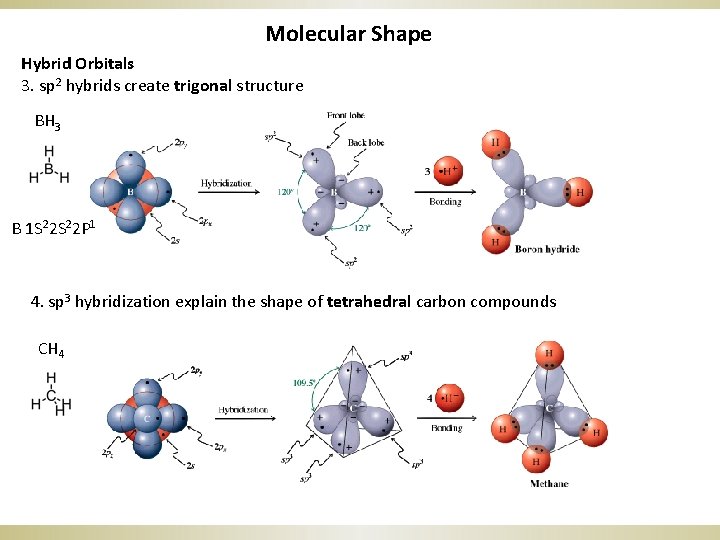
Molecular Shape Hybrid Orbitals 3. sp 2 hybrids create trigonal structure BH 3 B 1 S 22 P 1 4. sp 3 hybridization explain the shape of tetrahedral carbon compounds CH 4

Structure and Stability of Organic Compounds Bond energy and bond length C-C C=C bond length(pm) 154 134 120 bond energy(k. J/mol) 348 614 839 Determine the hybridization of C, N, O, B and F atoms in each of following compounds? NH 3, H 2 O, BF 3,
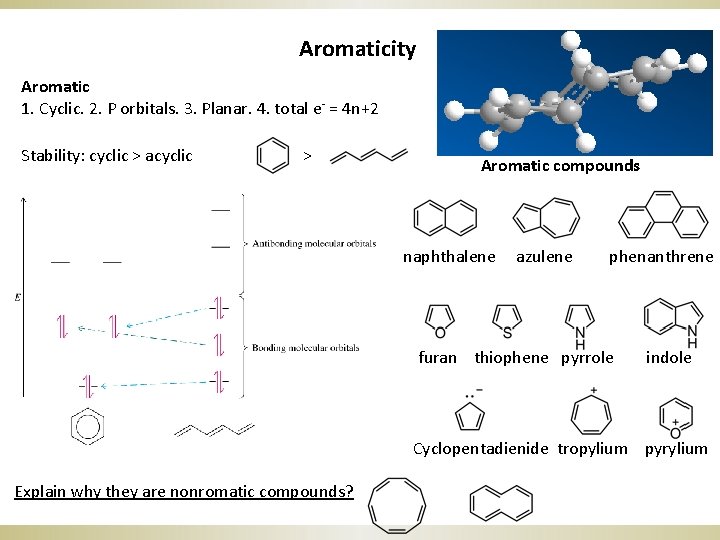
Aromaticity Aromatic 1. Cyclic. 2. P orbitals. 3. Planar. 4. total e- = 4 n+2 Stability: cyclic > acyclic > Aromatic compounds naphthalene azulene phenanthrene furan thiophene pyrrole indole Cyclopentadienide tropylium pyrylium Explain why they are nonromatic compounds?
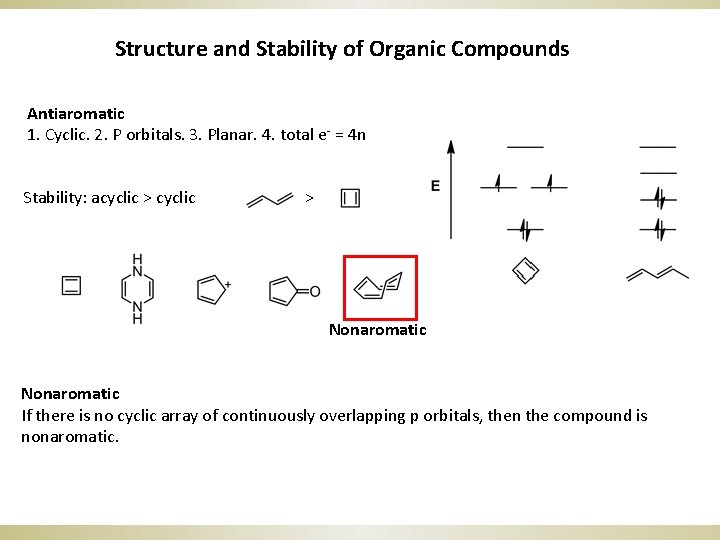
Structure and Stability of Organic Compounds Antiaromatic 1. Cyclic. 2. P orbitals. 3. Planar. 4. total e- = 4 n Stability: acyclic > Nonaromatic If there is no cyclic array of continuously overlapping p orbitals, then the compound is nonaromatic.
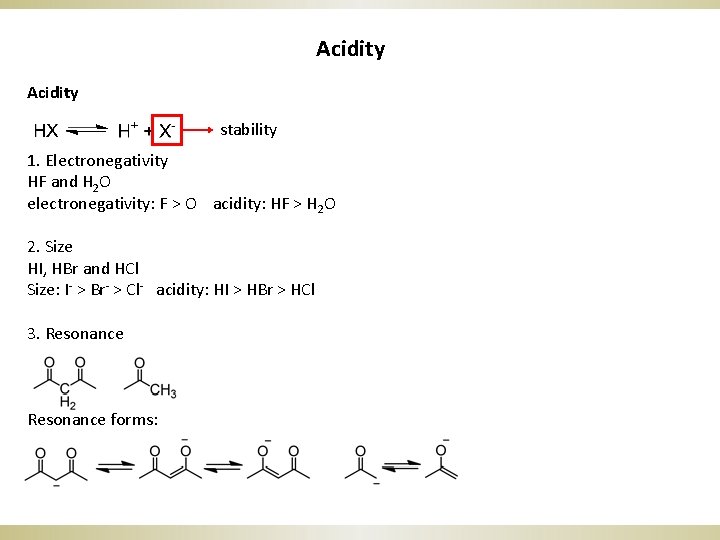
Acidity stability 1. Electronegativity HF and H 2 O electronegativity: F > O acidity: HF > H 2 O 2. Size HI, HBr and HCl Size: I- > Br- > Cl- acidity: HI > HBr > HCl 3. Resonance forms:

p. Ka Values Acidity
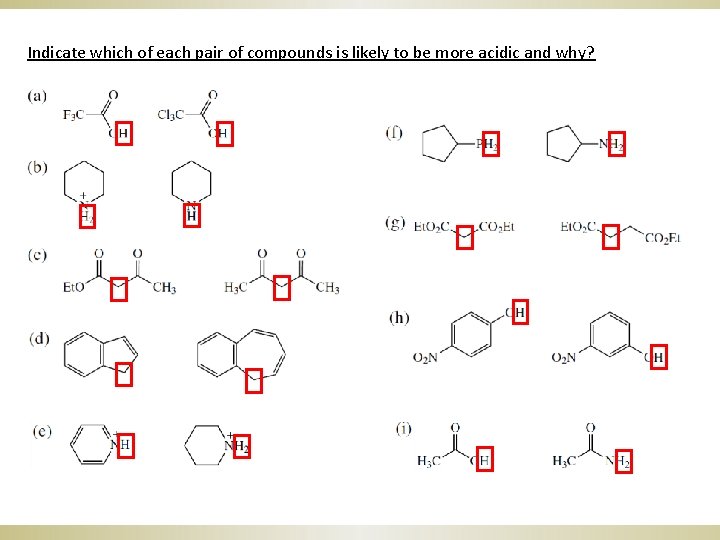
Indicate which of each pair of compounds is likely to be more acidic and why?
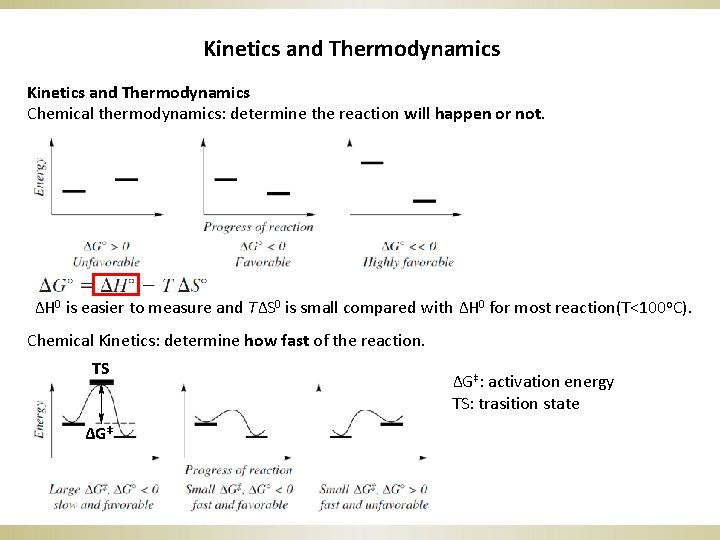
Kinetics and Thermodynamics Chemical thermodynamics: determine the reaction will happen or not. ΔH 0 is easier to measure and TΔS 0 is small compared with ΔH 0 for most reaction(T<100 o. C). Chemical Kinetics: determine how fast of the reaction. TS ΔG‡: activation energy TS: trasition state
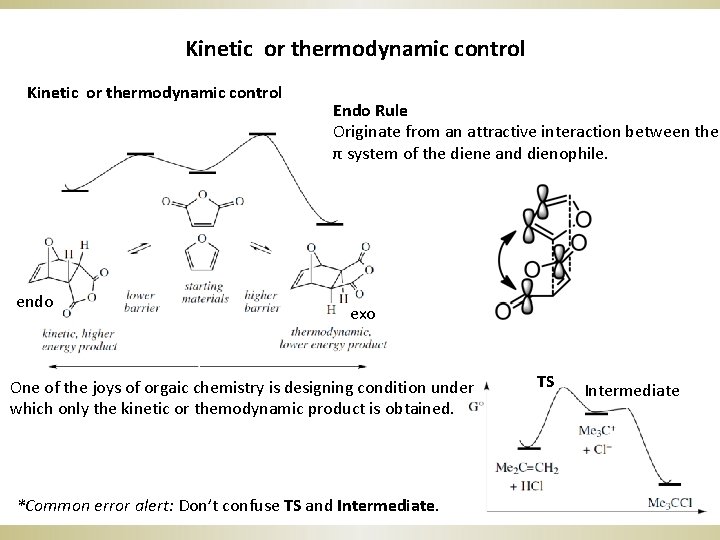
Kinetic or thermodynamic control endo Endo Rule Originate from an attractive interaction between the π system of the diene and dienophile. exo One of the joys of orgaic chemistry is designing condition under which only the kinetic or themodynamic product is obtained. *Common error alert: Don’t confuse TS and Intermediate. TS Intermediate
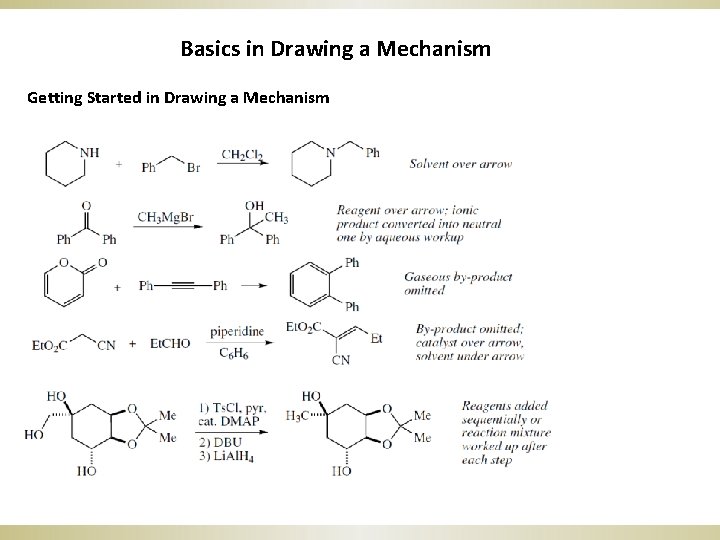
Basics in Drawing a Mechanism Getting Started in Drawing a Mechanism
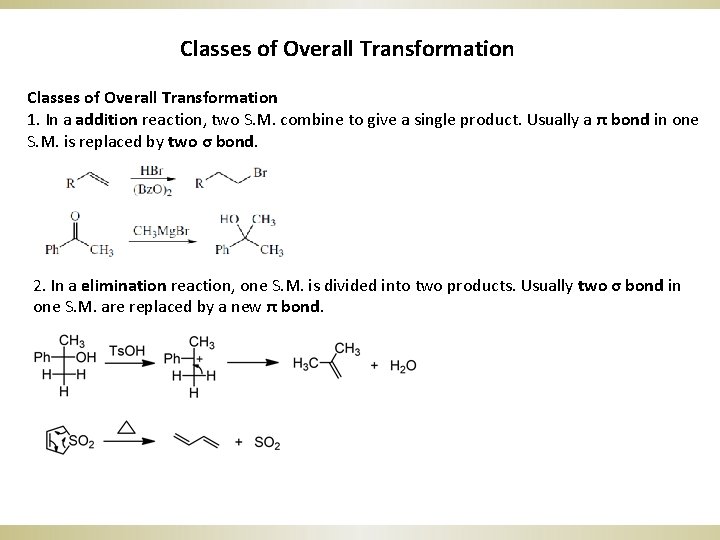
Classes of Overall Transformation 1. In a addition reaction, two S. M. combine to give a single product. Usually a π bond in one S. M. is replaced by two σ bond. 2. In a elimination reaction, one S. M. is divided into two products. Usually two σ bond in one S. M. are replaced by a new π bond.
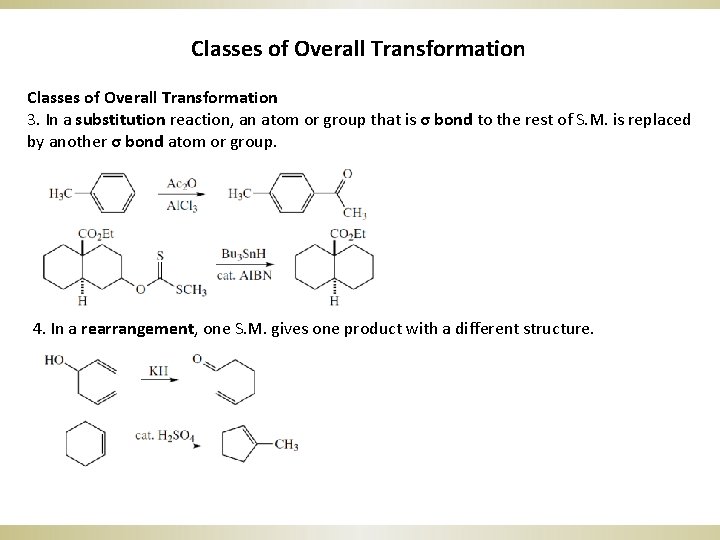
Classes of Overall Transformation 3. In a substitution reaction, an atom or group that is σ bond to the rest of S. M. is replaced by another σ bond atom or group. 4. In a rearrangement, one S. M. gives one product with a different structure.
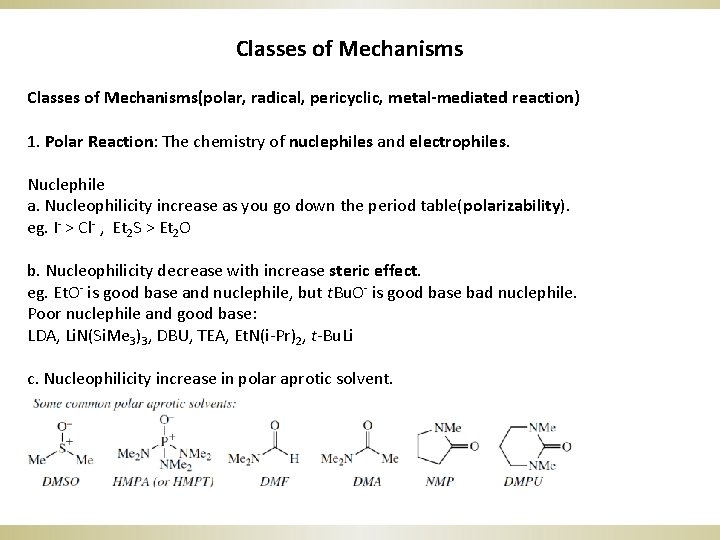
Classes of Mechanisms(polar, radical, pericyclic, metal-mediated reaction) 1. Polar Reaction: The chemistry of nuclephiles and electrophiles. Nuclephile a. Nucleophilicity increase as you go down the period table(polarizability). eg. I- > Cl- , Et 2 S > Et 2 O b. Nucleophilicity decrease with increase steric effect. eg. Et. O- is good base and nuclephile, but t. Bu. O- is good base bad nuclephile. Poor nuclephile and good base: LDA, Li. N(Si. Me 3)3, DBU, TEA, Et. N(i-Pr)2, t-Bu. Li c. Nucleophilicity increase in polar aprotic solvent.
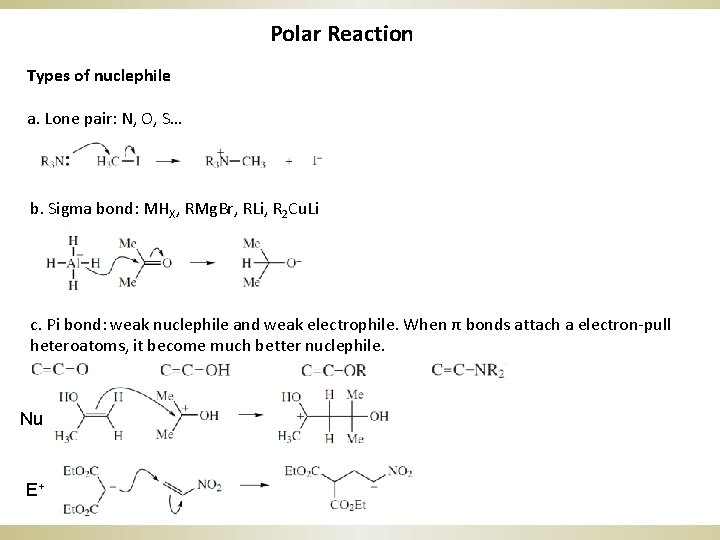
Polar Reaction Types of nuclephile a. Lone pair: N, O, S… b. Sigma bond: MHX, RMg. Br, RLi, R 2 Cu. Li c. Pi bond: weak nuclephile and weak electrophile. When π bonds attach a electron-pull heteroatoms, it become much better nuclephile. Nu E+
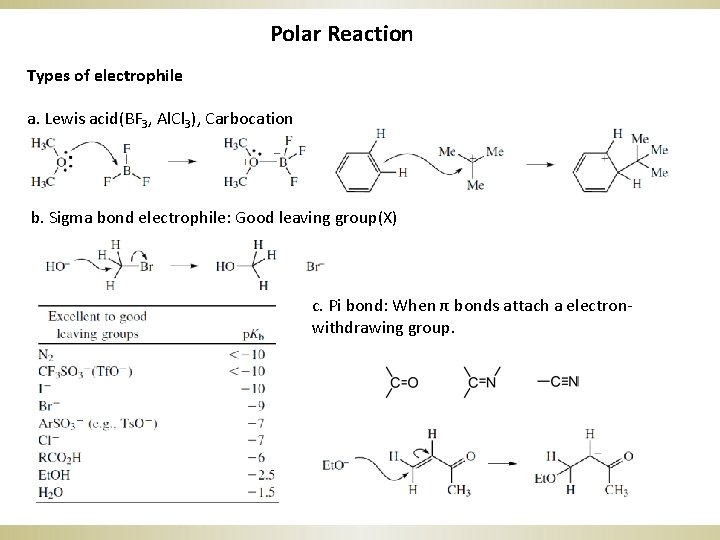
Polar Reaction Types of electrophile a. Lewis acid(BF 3, Al. Cl 3), Carbocation b. Sigma bond electrophile: Good leaving group(X) c. Pi bond: When π bonds attach a electronwithdrawing group.
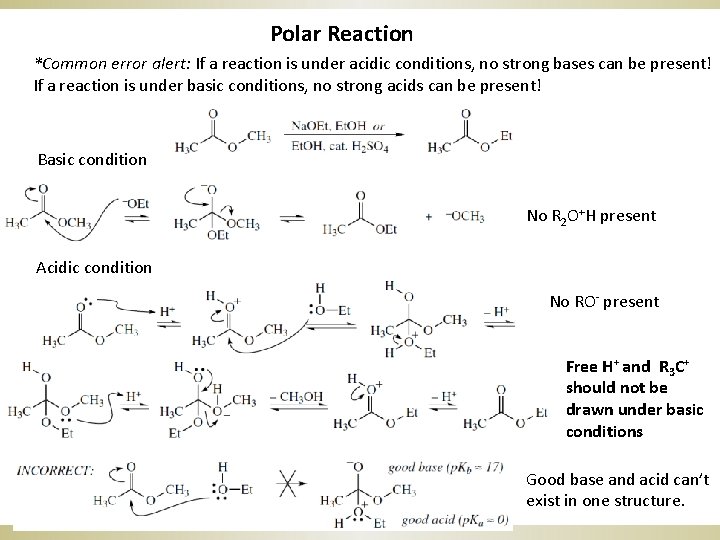
Polar Reaction *Common error alert: If a reaction is under acidic conditions, no strong bases can be present! If a reaction is under basic conditions, no strong acids can be present! Basic condition No R 2 O+H present Acidic condition No RO- present Free H+ and R 3 C+ should not be drawn under basic conditions Good base and acid can’t exist in one structure.
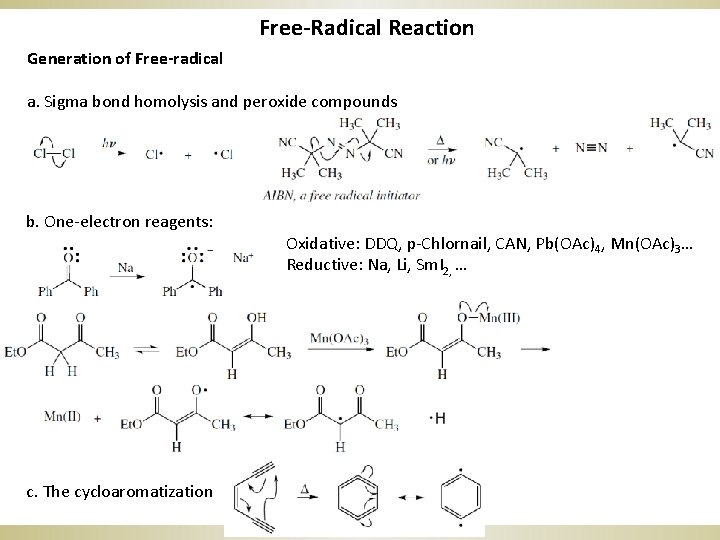
Free-Radical Reaction Generation of Free-radical a. Sigma bond homolysis and peroxide compounds b. One-electron reagents: c. The cycloaromatization Oxidative: DDQ, p-Chlornail, CAN, Pb(OAc)4, Mn(OAc)3… Reductive: Na, Li, Sm. I 2, …
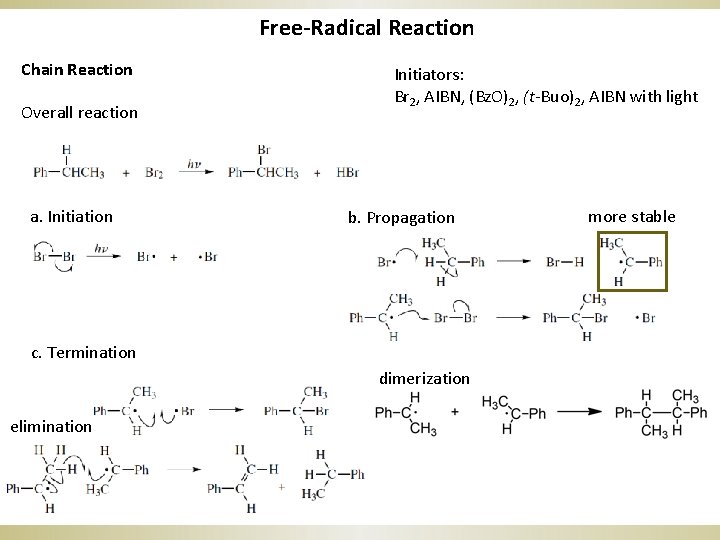
Free-Radical Reaction Chain Reaction Overall reaction a. Initiation Initiators: Br 2, AIBN, (Bz. O)2, (t-Buo)2, AIBN with light b. Propagation c. Termination dimerization elimination more stable
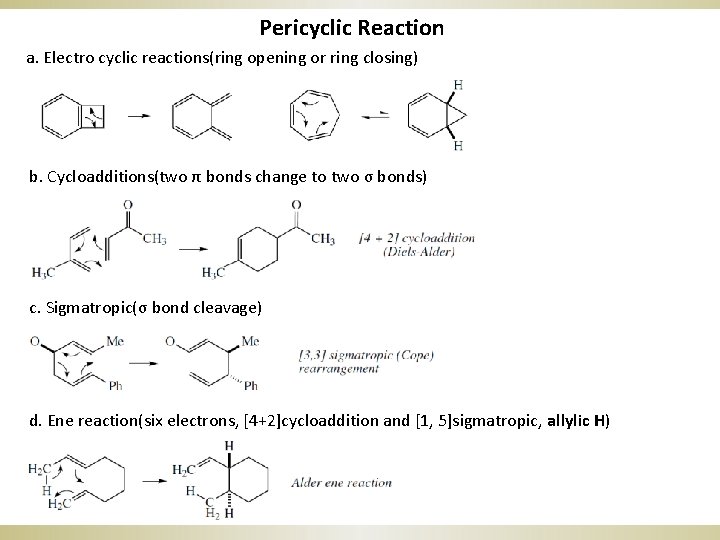
Pericyclic Reaction a. Electro cyclic reactions(ring opening or ring closing) b. Cycloadditions(two π bonds change to two σ bonds) c. Sigmatropic(σ bond cleavage) d. Ene reaction(six electrons, [4+2]cycloaddition and [1, 5]sigmatropic, allylic H)
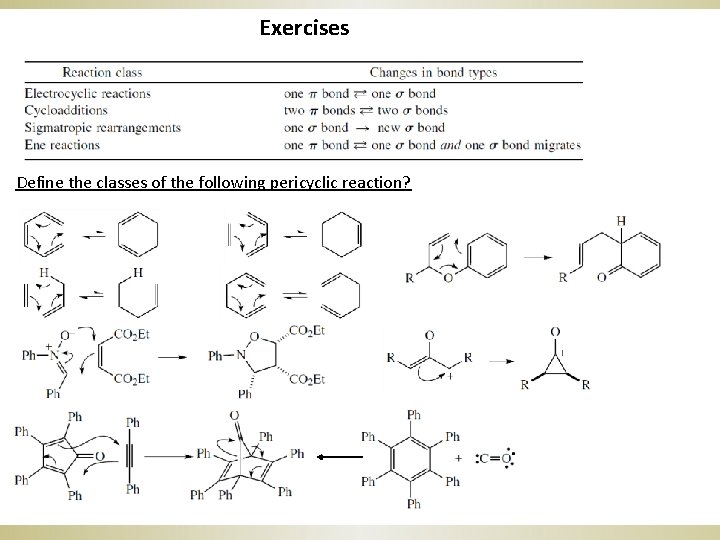
Exercises Define the classes of the following pericyclic reaction?
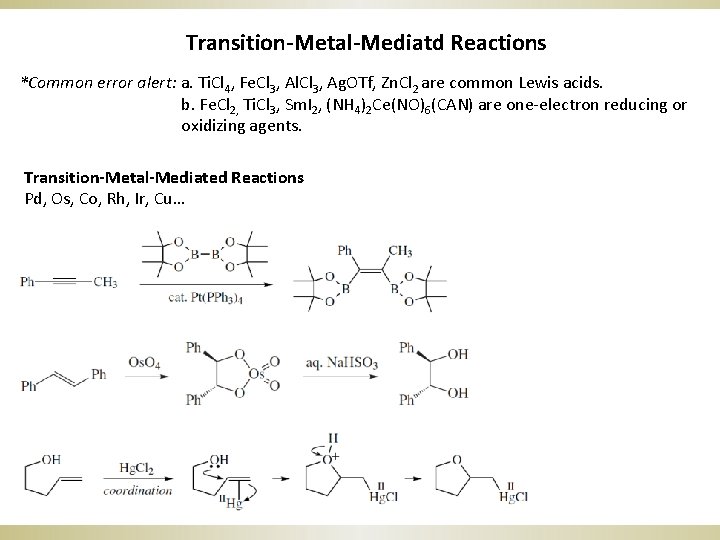
Transition-Metal-Mediatd Reactions *Common error alert: a. Ti. Cl 4, Fe. Cl 3, Al. Cl 3, Ag. OTf, Zn. Cl 2 are common Lewis acids. b. Fe. Cl 2, Ti. Cl 3, Sm. I 2, (NH 4)2 Ce(NO)6(CAN) are one-electron reducing or oxidizing agents. Transition-Metal-Mediated Reactions Pd, Os, Co, Rh, Ir, Cu…
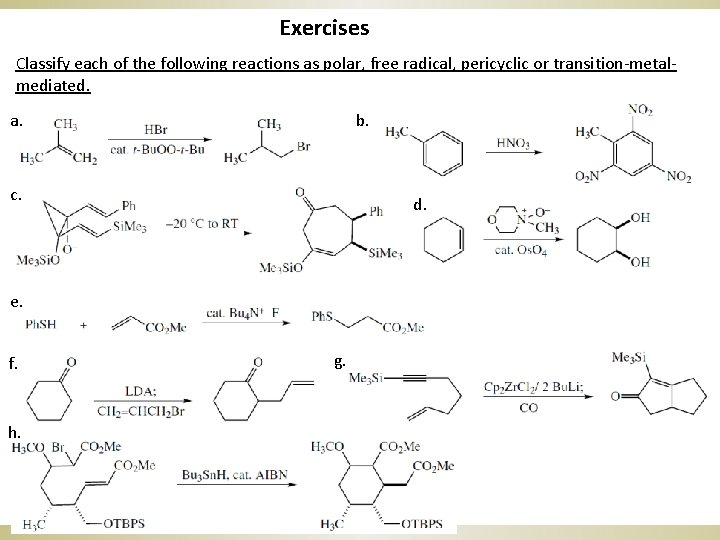
Exercises Classify each of the following reactions as polar, free radical, pericyclic or transition-metalmediated. a. b. c. d. e. f. h. g.

- Slides: 35