Organic Chemistry Unit What is Organic Chemistry The





- Slides: 17

Organic Chemistry Unit

What is Organic Chemistry? The study of carbon-containing compounds made up of non-metal elements (covalent bonds)

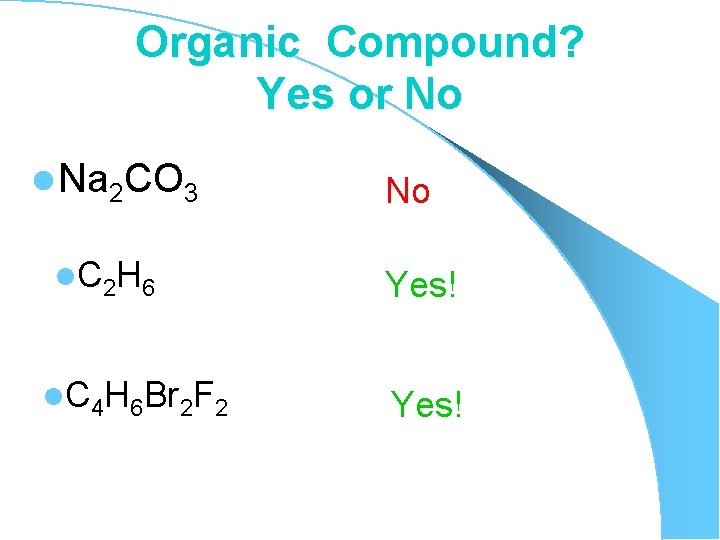
Organic Compound? Yes or No l Na 2 CO 3 l C 2 H 6 l. C 4 H 6 Br 2 F 2 No Yes!

Why Carbon? ? l Found in Nature ( ranked 17 th in crust) l Element l Compound l Found in all living matter l Found in body tissue l Found in food l Found in fuels (coal, wood, petroleum)

Forms of Carbon l Term = Allotrope Same element l Different bonding pattern l Different arrangement l • Carbon exists in 3 solid forms • Diamond • Graphite • Fullerene

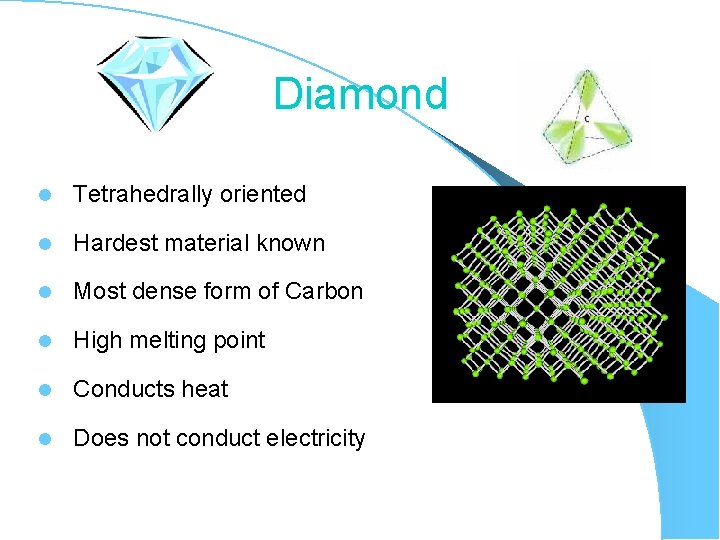
Diamond l Tetrahedrally oriented l Hardest material known l Most dense form of Carbon l High melting point l Conducts heat l Does not conduct electricity

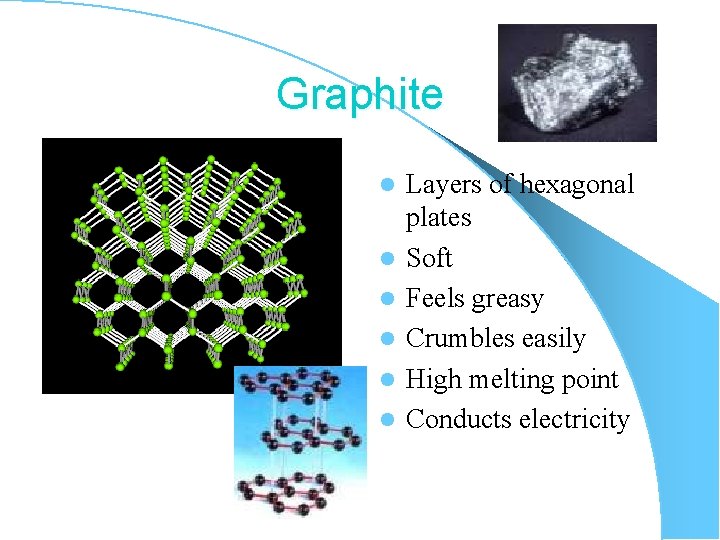
Graphite l l l Layers of hexagonal plates Soft Feels greasy Crumbles easily High melting point Conducts electricity

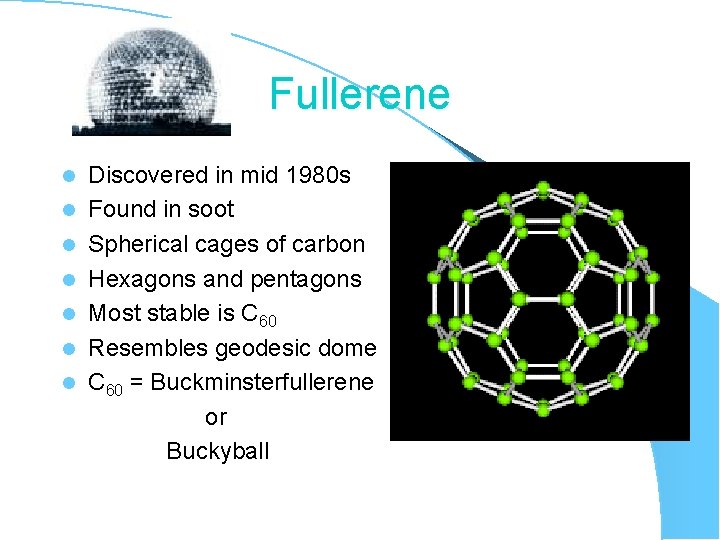
Fullerene l l l l Discovered in mid 1980 s Found in soot Spherical cages of carbon Hexagons and pentagons Most stable is C 60 Resembles geodesic dome C 60 = Buckminsterfullerene or Buckyball

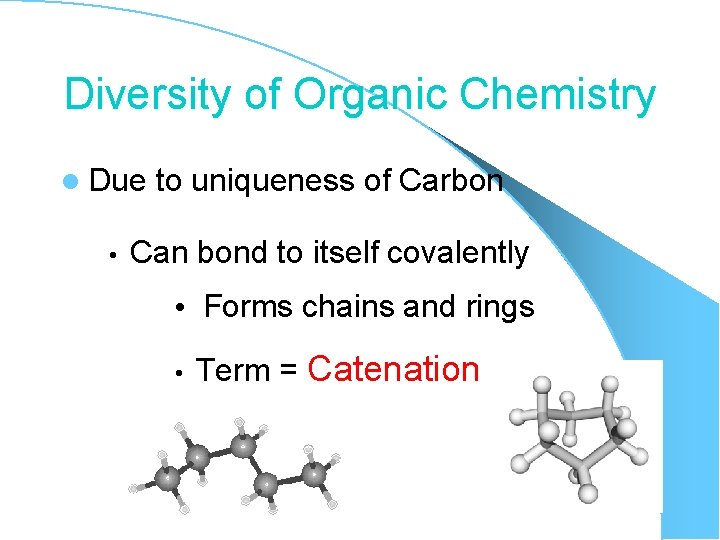
Diversity of Organic Chemistry l Due • to uniqueness of Carbon Can bond to itself covalently • Forms chains and rings • Term = Catenation

Carbon bonds to elements l Carbon readily bonds to : – – – H O N S Halogens l Cl, Br, F, I l Hydrocarbons – Simplest organic compounds – Only contain Carbon and Hydrogen (Cx. Hy)

Formulas Written representations of a compound using letters (and sometimes numbers) Molecular Formula Indicates l # of atoms l Types of atoms Example: C 8 H 18

Structural Formula Indicates • • • # of atoms Type of atoms Bonding Arrangement • Shiloh and Dione were here ☻

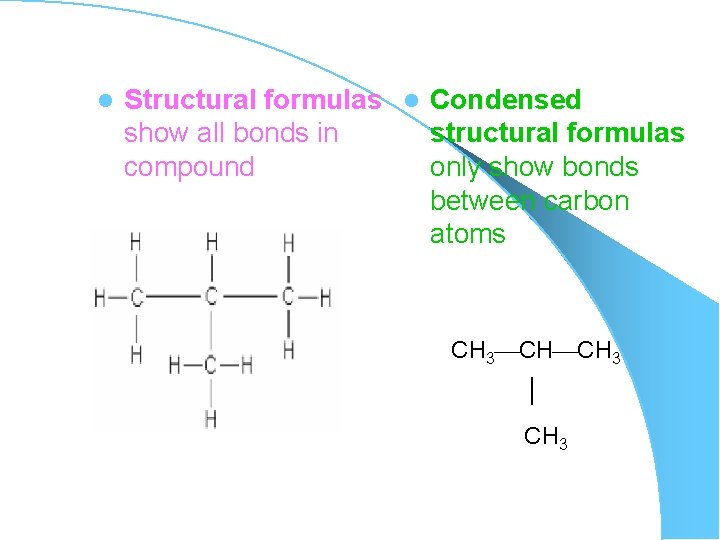
l Structural formulas l Condensed show all bonds in structural formulas compound only show bonds between carbon atoms CH 3 CH CH 3

Arrangement of Atoms l Compounds that As # of carbon atoms goes up have: l # of isomers goes up – Same molecular – C 8 18 isomers formula – C 9 35 isomers – Different structure – C 10 75 isomers or arrangement – C 40 – Called ISOMERS 69, 491, 178, 805, 831 l

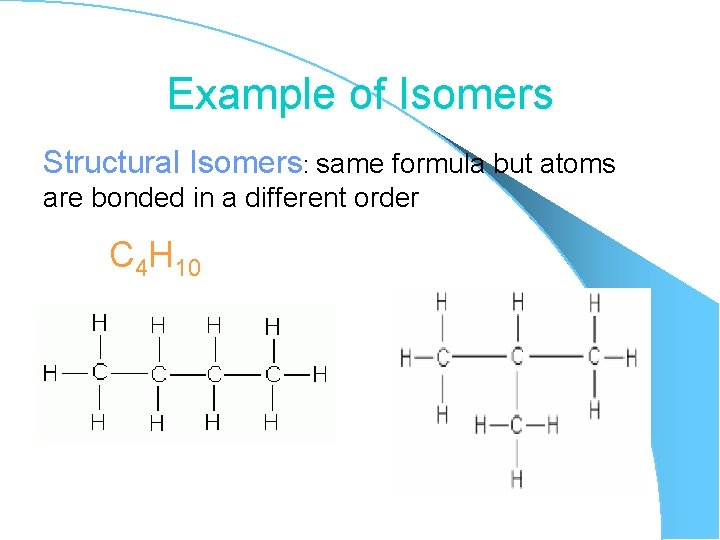
Example of Isomers Structural Isomers: same formula but atoms are bonded in a different order C 4 H 10

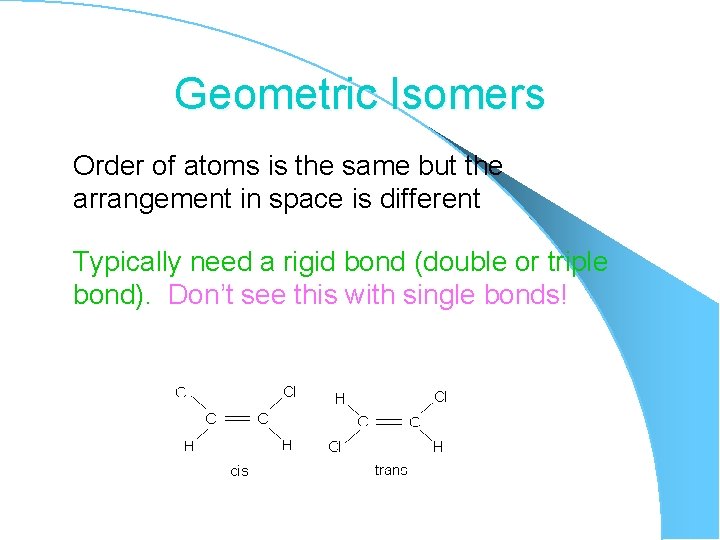
Geometric Isomers Order of atoms is the same but the arrangement in space is different Typically need a rigid bond (double or triple bond). Don’t see this with single bonds!

We are going to study: l Alkanes l Ethers l Alkenes l Esters l Alkynes l Aldehydes l Aromatics l Ketones l Alcohols l Amines