Organic Chemistry and Biochemistry Organic Chemistry o Organic
Organic Chemistry and Biochemistry

Organic Chemistry o Organic Chemistry - the branch of chemistry that deals with carbon compounds n n n n All living organisms Food (proteins, carbohydrates, lipids) Fuels (ethanol, propane) Wood and paper products Plastics (polymers) Soaps and detergents (lipids + water+ Na. OH) Cosmetics Medicines

Carbon Atom o Central to all organic compounds o Usually forms four covalent bonds 2 2 n Electron structure 1 s 2 s 2 p n Four valence electrons 2 o Carbon-carbon bonds are formed because carbon atoms can easily share electrons (covalent bonding) with other carbon atoms
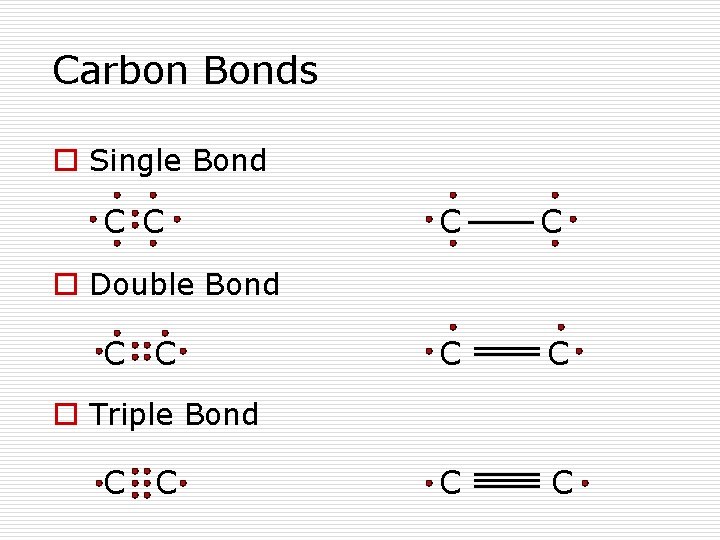
Carbon Bonds o Single Bond C C C C o Double Bond C C o Triple Bond C C

Hydrocarbons o Hydrocarbons - compounds composed entirely of carbon and hydrogen atoms n Saturated hydrocarbons – have only single bonds between carbon atoms (alkanes) n Unsaturated hydrocarbons – contain a double or a triple bond between two carbon atoms (alkenes/alkynes)

Hydrocarbons o Alkanes – saturated hydrocarbons with only single covalent bonds o Alkenes – unsaturated hydrocarbons with at least one double bond; no triple bonds; single bonds are also allowed o Alkynes – unsaturated hydrocarbons with at least one triple bond; single and double bonds are also allowed
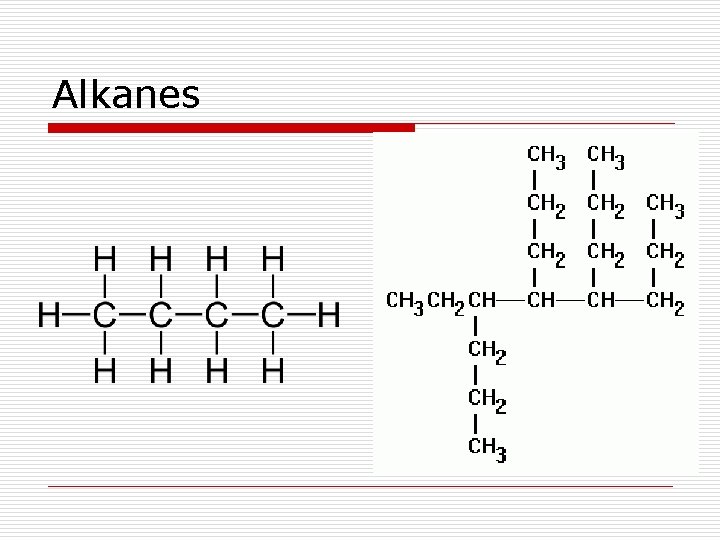
Alkanes
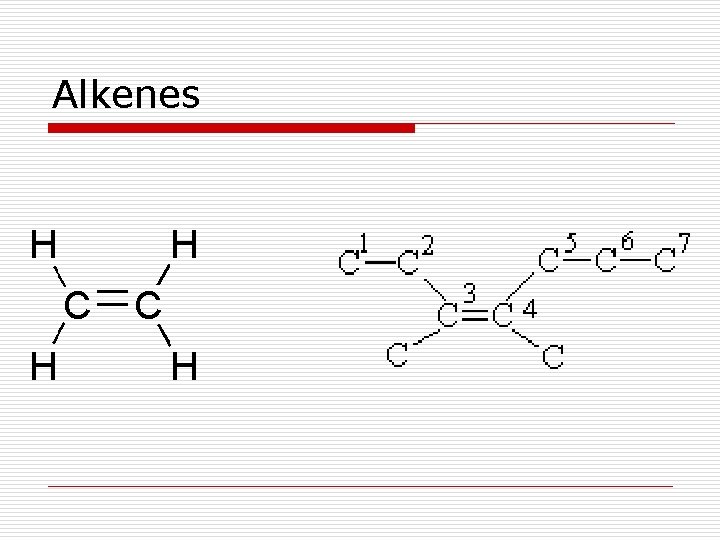
Alkenes
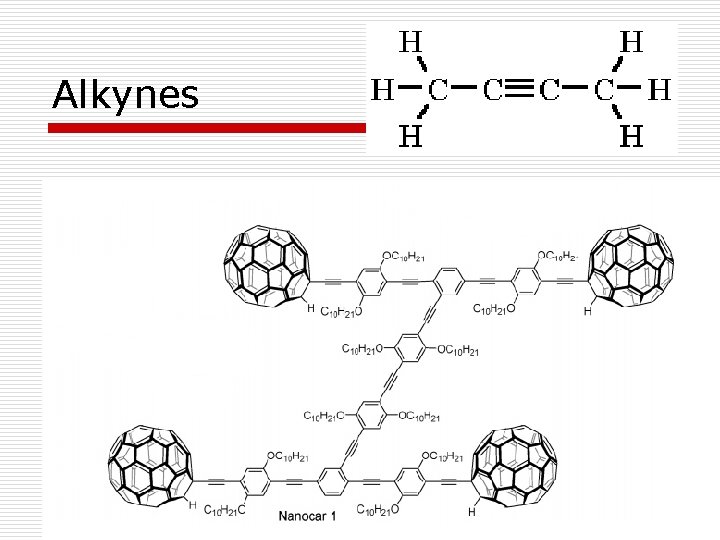
Alkynes
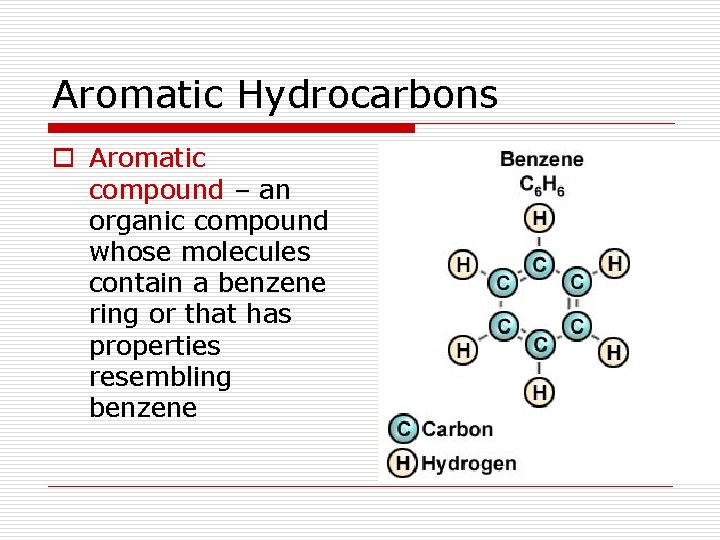
Aromatic Hydrocarbons o Aromatic compound – an organic compound whose molecules contain a benzene ring or that has properties resembling benzene
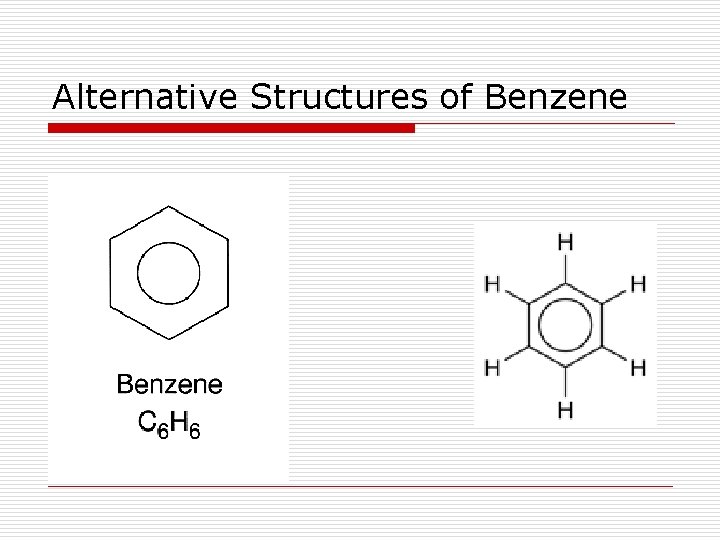
Alternative Structures of Benzene
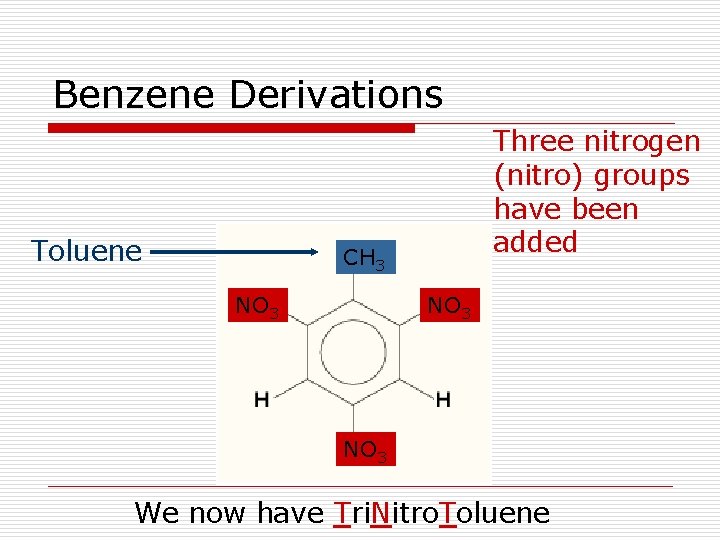
Benzene Derivations Toluene Three nitrogen (nitro) groups have been added CH 3 NO 3 We now have Tri. Nitro. Toluene

Poor Guy…

Hydrocarbon Derivatives o Hydrocarbon Derivatives – compounds that can be synthesized from a hydrocarbon o Hydrocarbon derivatives can contain other elements such as oxygen, nitrogen, or a halogen

Four Classes of Hydrocarbon Derivatives o o Alcohols Aldehydes Ketones Esters

Four Classes of Hydrocarbon Derivatives o 1. Alcohols – organic compounds whose molecules contain a hydroxyl group (-OH) covalently bonded to a saturated carbon atom n Example: ethanol n R - OH

Four Classes of Hydrocarbon Derivatives o 2. Aldehydes – organic compounds whose molecules contain a carbon double-bonded to an oxygen atom (at least one H is also attached to the C atom) n Example: formaldehyde

Four Classes of Hydrocarbon Derivatives o 3. Ketones – (similar to aldehydes) organic compounds whose molecules contain a carbon atom double-bonded to an oxygen atom (no H atoms are directly attached to the C atom) = O H 3 C – CH 3 Acetone

Four Classes of Hydrocarbon Derivatives o 4. Esters – organic compounds whose molecules contain a carbon atom double-bonded to an oxygen atom and single-bonded to another oxygen atom (this second oxygen atom will have at least one other atom attached)
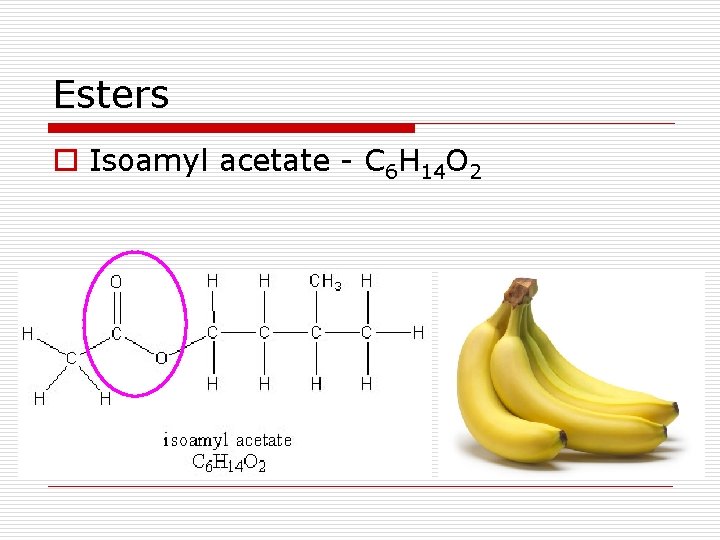
Esters o Isoamyl acetate - C 6 H 14 O 2
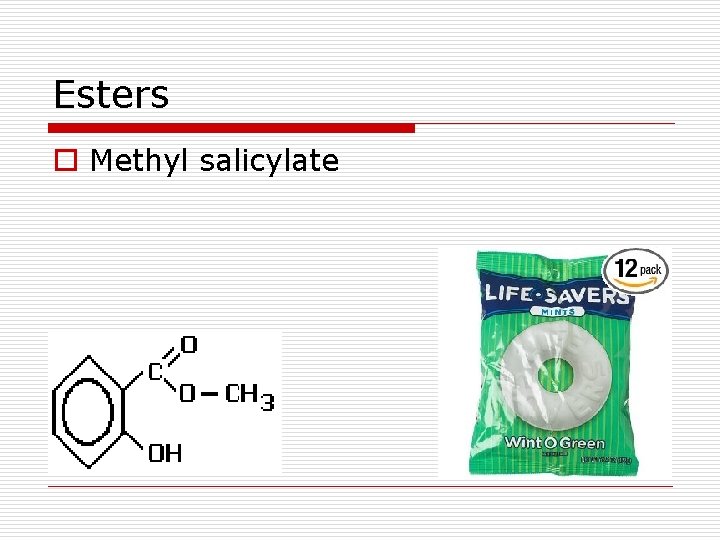
Esters o Methyl salicylate

Isomers o Isomers – compounds having identical molecular formulas but different structural formulas o Pentane – C 5 H 12 (molecular formula)
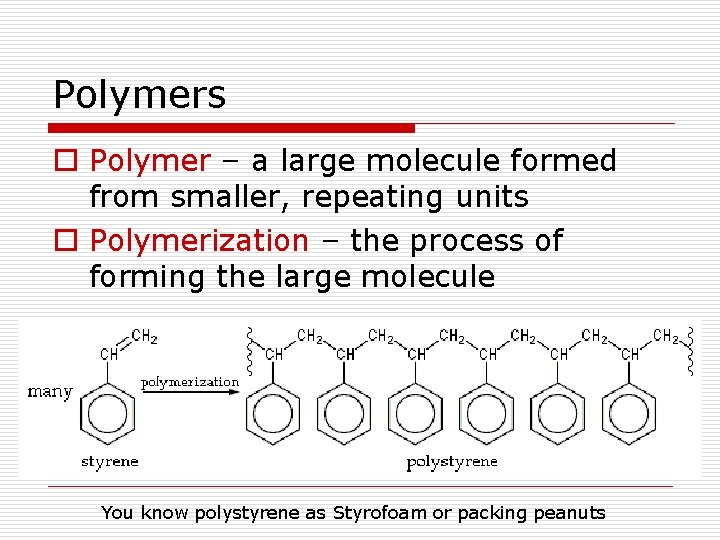
Polymers o Polymer – a large molecule formed from smaller, repeating units o Polymerization – the process of forming the large molecule You know polystyrene as Styrofoam or packing peanuts

Biochemistry o Biochemistry – the branch of chemistry concerned with the chemical reactions occurring in living organisms n n Growth Digestion Metabolism Reproduction

Four Major Classes of Biomolecules o o Carbohydrates Lipids Proteins Nucleic Acids

Carbohydrates o Carbohydrate – a polyhydroxy aldehyde or polyhydroxy ketone n Examples: sugar, starch, cellulose

Lipids o Lipids – organic compounds found in living organisms that are water insoluable; they ARE soluable in most alcohols; lipids are fats, oils, and steroids

Proteins o Proteins – polymers consisting of amino acids linked together n Amino acids – the building blocks of all proteins o Example: Tryptophan – the amino acid in turkey that has been linked to making people sleepy
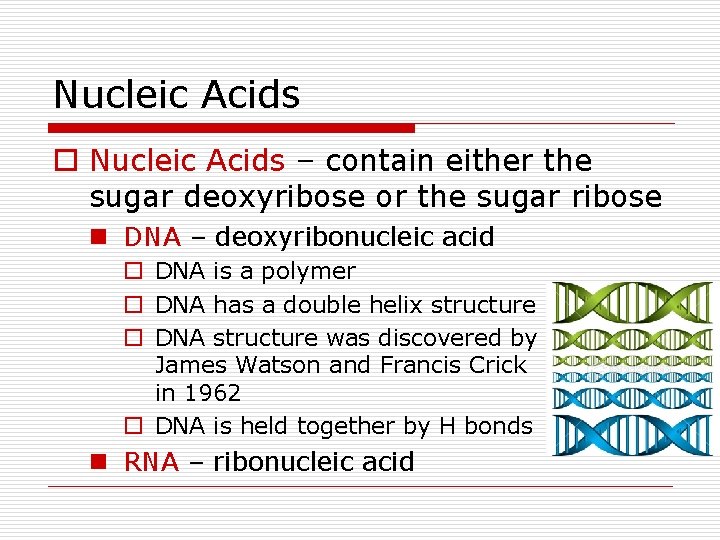
Nucleic Acids o Nucleic Acids – contain either the sugar deoxyribose or the sugar ribose n DNA – deoxyribonucleic acid o DNA is a polymer o DNA has a double helix structure o DNA structure was discovered by James Watson and Francis Crick in 1962 o DNA is held together by H bonds n RNA – ribonucleic acid
- Slides: 29