Organic Chemistry Introduction Brainstorming Activity Organic Chemistry Organic

Organic Chemistry Introduction

Brainstorming Activity

Organic Chemistry • Organic compounds are everywhere • It is defined as the chemistry of the carbon compound • Hydrogen atoms typically accompany the carbon (hydrocarbon) • Over 8 million organic compounds

Organic Compounds in real life worksheet
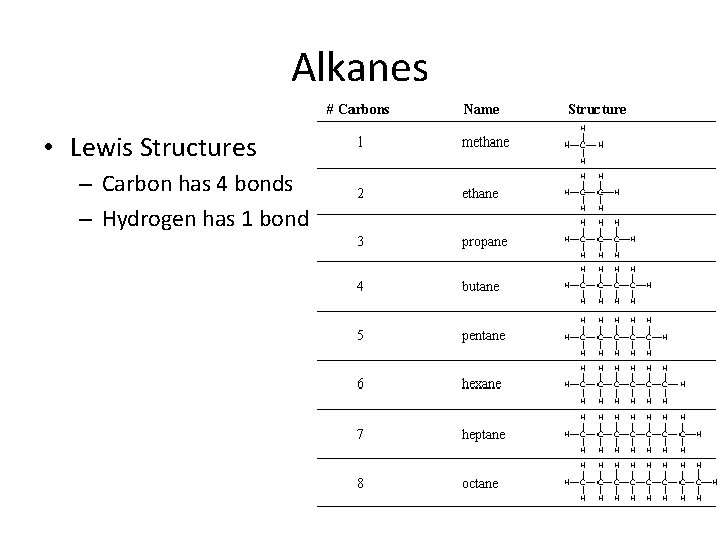
Alkanes • Lewis Structures – Carbon has 4 bonds – Hydrogen has 1 bond
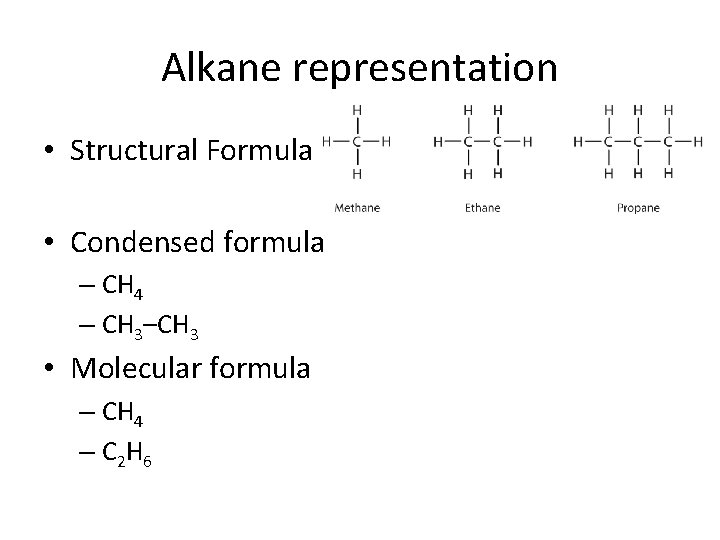
Alkane representation • Structural Formula • Condensed formula – CH 4 – CH 3–CH 3 • Molecular formula – CH 4 – C 2 H 6

What is a way to remember the molecular formula?

Straight Chained Alkanes • All alkane’s end in –ane • Saturated = each carbon has the maximum number of hydrogen's attached • Geometry of an alkane is not straight – It is bent! – To make sure all hydrogens are as far away from every other hydrogen – 109° between every single H-C-H bond – Tetrahedron – The bonds can rotate freely in an alkane
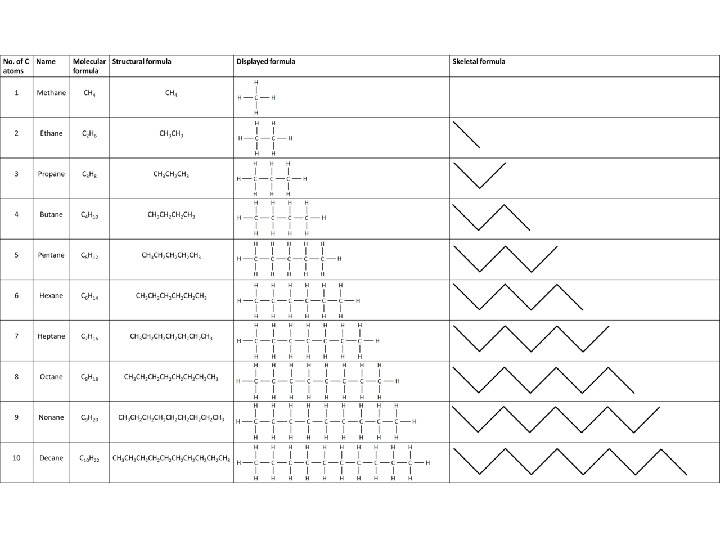

Branches • Hydrocarbons aren’t just straight chained alkanes • They can have branches! • We call these side branches, alkyl groups – The end of alkyl groups end in –yl • Branches can be as long as they want wherever they want • -CH 3 CH 2 CH 3 is a butyl branch
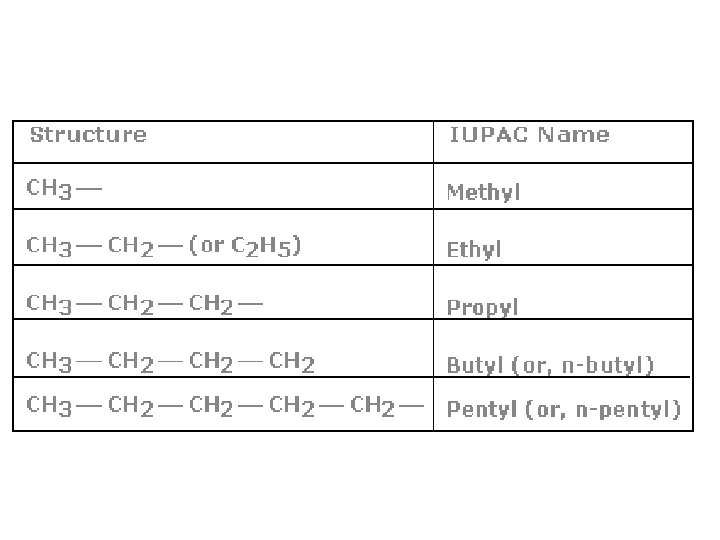

How do we name the branches? • Name the longest chain possible – This is the parent chain • Number our parent chain from 1 – Can number from any direction • You want to make sure your branches have the lowest combined set of numbers. • List your alkyl groups and their numbers. • Put them in alphabetical order based on alkyl group – Ethyl before methyl • Name them by # of where they are, then a dash, then the alkyl group, then the alkane

Naming • • • #-alkylalkane 2 -methylpentane 3 -ethyl-2 -methyloctane 3, 3 -dimethylhexane 2, 4 -dimethylheptane 2, 3, 4 -triethylnonane

How carbons bond Carbon has 4 bonds Carbon without any other carbon is CH 4 Carbon with 1 carbon attached to it becomes CH 3 Carbon with 2 carbons attached to it becomes CH 2 • Carbon with 3 carbons attached to it becomes CH • Carbon with 4 carbons attached to it becomes C • •
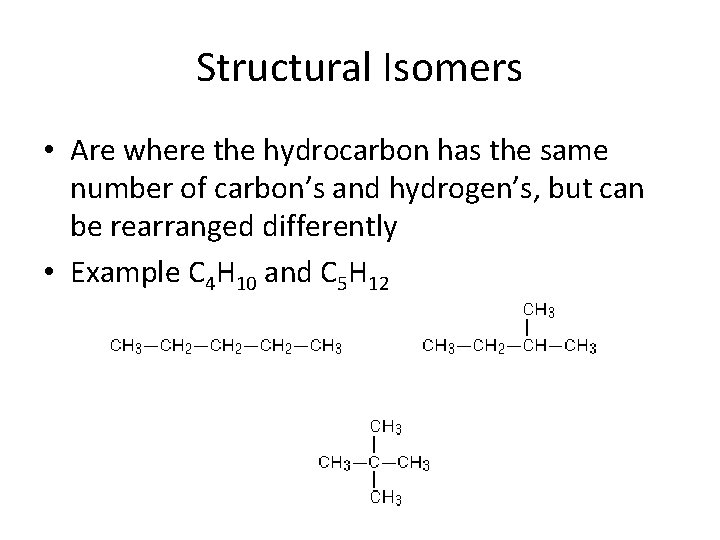
Structural Isomers • Are where the hydrocarbon has the same number of carbon’s and hydrogen’s, but can be rearranged differently • Example C 4 H 10 and C 5 H 12

Properties of Alkanes • Structural isomers each have their own properties based on arrangement • Properties also vary based on chain length – Methane to propane are gas at room temperature – Pentane to C 15 H 32 are liquid at room temperature – C 16 H 34 and longer are solid at room temperature
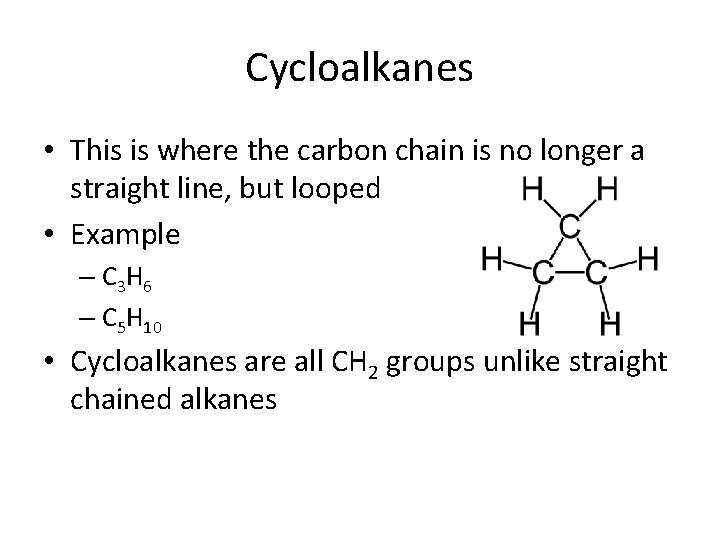
Cycloalkanes • This is where the carbon chain is no longer a straight line, but looped • Example – C 3 H 6 – C 5 H 10 • Cycloalkanes are all CH 2 groups unlike straight chained alkanes

Naming Cycloalkanes • We name all our cycloalkanes the same, except we add cyclo to the front – Cyclopropane – Cyclobutane – Cyclopentane • What if we have branches? – We follow the same naming pattern – Except, if we have 1 branch, we do not need to put a number – If we have more than 1 branch, we add numbers where the first branch alphabetically is 1 and we go around to form the lowest number possible

Alkyl Halides • They are like alkyl groups, but are halogens • Same naming system has halides when they are part of a branch • Fluorine = fluro • Chlorine = chloro • Bromine = bromo • Iodine = iodo • Example – 2 -fluropentane

Alkyl Halides • Alkyl halides have special properties based on their branch and number • They can be unreactive and inert such as fluoro halides – Teflon is a fluoro halide and is unreactive and inert

Multiple Bonded Carbon chains • Double bonded carbon-carbon is called Alkene – Their names end in -ene – C=C • Triple bonded carbon-carbon is called Alkyne – Their names end in –yne – C≡C
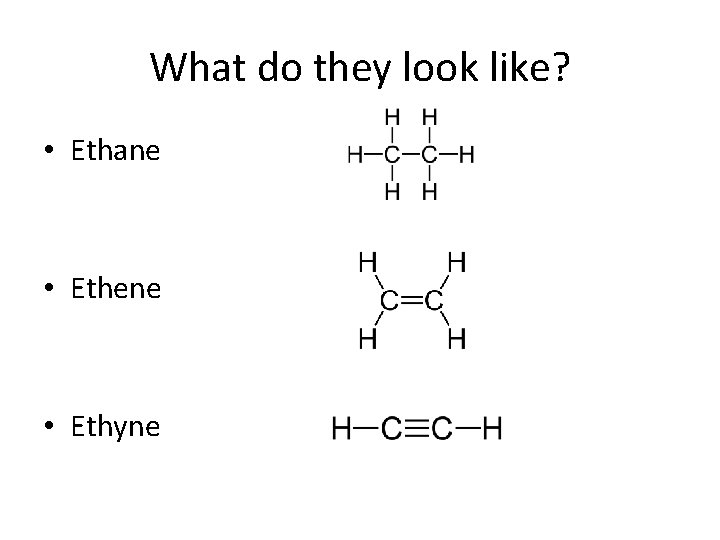
What do they look like? • Ethane • Ethene • Ethyne

Unsaturated • Because these carbons do not have full number of hydrogen’s around each carbon, we say they are unsaturated

To name multiple bonded carbon chains • We number the carbon chain so that our double bonded is the lowest number possible • We name it based on the first carbon of the double bond and its number • 3 -pentene • 4 -heptyne • To write the condense structure, do it like usual, except we put a double or triple bond where it is

Geometry of alkene and alkyne • The geometry of multiple bonded carbons is a planar/flat molecule • This is due to no rotation around the bond and how the bonds are bonded • Very rigid
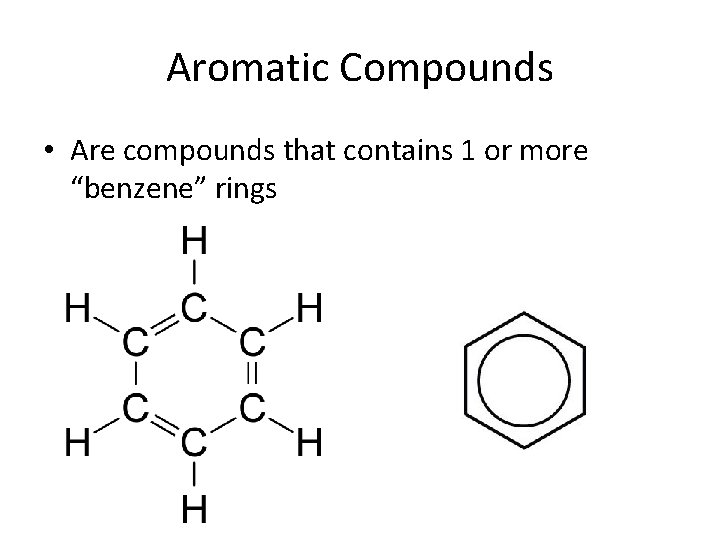
Aromatic Compounds • Are compounds that contains 1 or more “benzene” rings

Aromatic Compounds • Aromatic comes from the word aroma • They smell, sometimes good, sometimes bad • We name aromatic compounds the same as we do with cyclic molecules • The ending is benzene • Example – 1, 2 -methylbenzene

Functional Groups • Specific groups of atoms that exist in molecules that gives the molecule special properties and reactivity • They are what turns organic compounds into alcohols, acids, bases, soluble, insoluble, explosive, or smelly
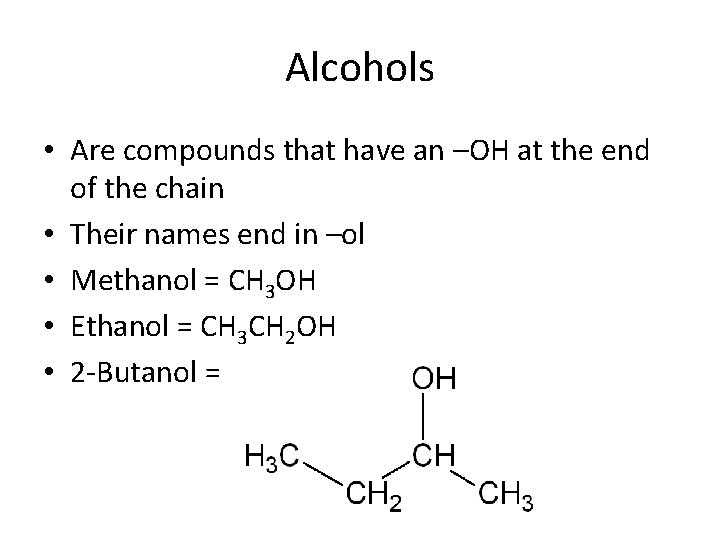
Alcohols • Are compounds that have an –OH at the end of the chain • Their names end in –ol • Methanol = CH 3 OH • Ethanol = CH 3 CH 2 OH • 2 -Butanol =

Properties of Alcohols • The –OH group makes the organic compound soluble in water up to a certain length of the carbon chain • Shorter chains are soluble – Up to propanol – Longer chains such as pentanol are insoluble in water due to the carbon chain being non-polar • Alcohols are poisonous! – Ethanol is just less poisonous, but still is.
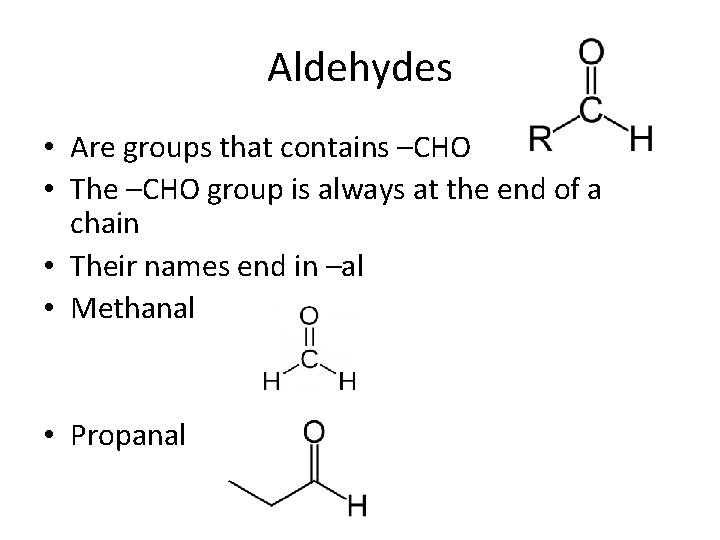
Aldehydes • Are groups that contains –CHO • The –CHO group is always at the end of a chain • Their names end in –al • Methanal • Propanal
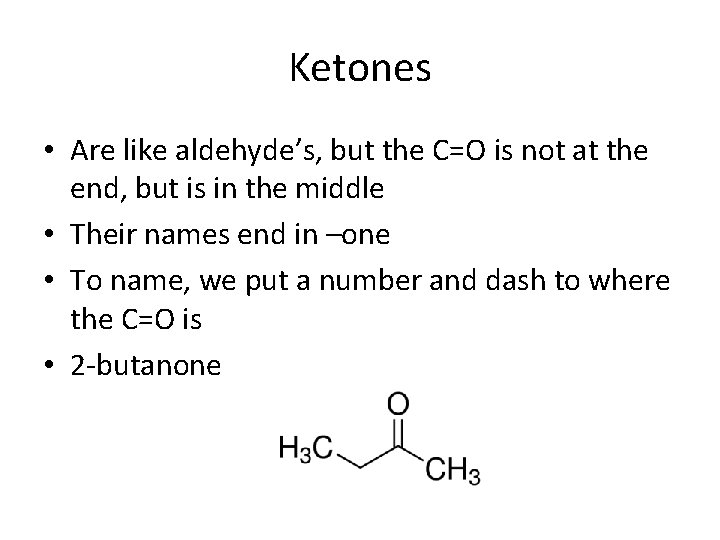
Ketones • Are like aldehyde’s, but the C=O is not at the end, but is in the middle • Their names end in –one • To name, we put a number and dash to where the C=O is • 2 -butanone

Ethers • Are anesthetic compounds that can be used in hospitals to put people to sleep • They are where the oxygen splits the carbon chain • To name, we separate the two carbon chains into their own carbon chain groups • We name the SHORTER chain with the ending –oxy
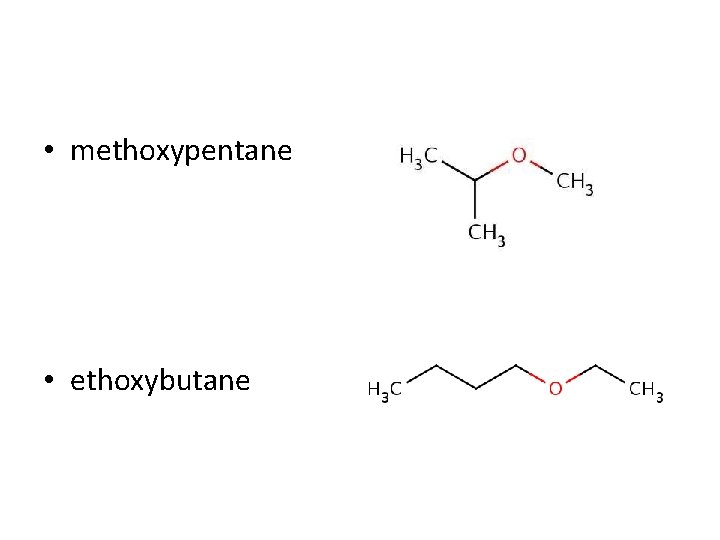
• methoxypentane • ethoxybutane
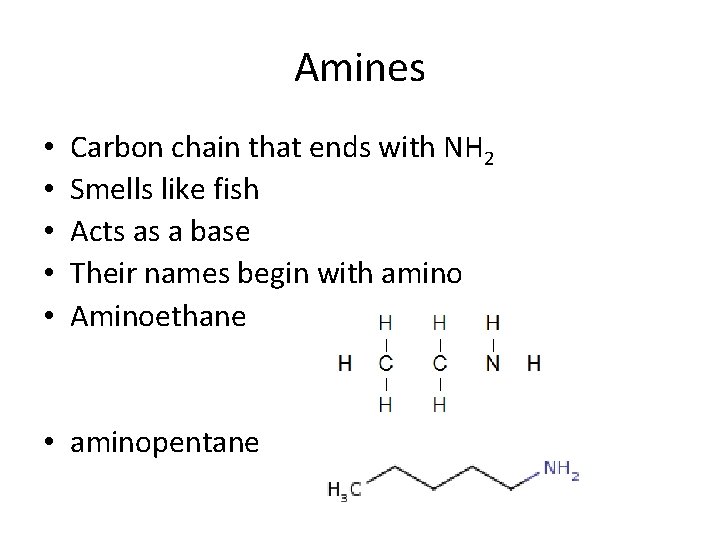
Amines • • • Carbon chain that ends with NH 2 Smells like fish Acts as a base Their names begin with amino Aminoethane • aminopentane
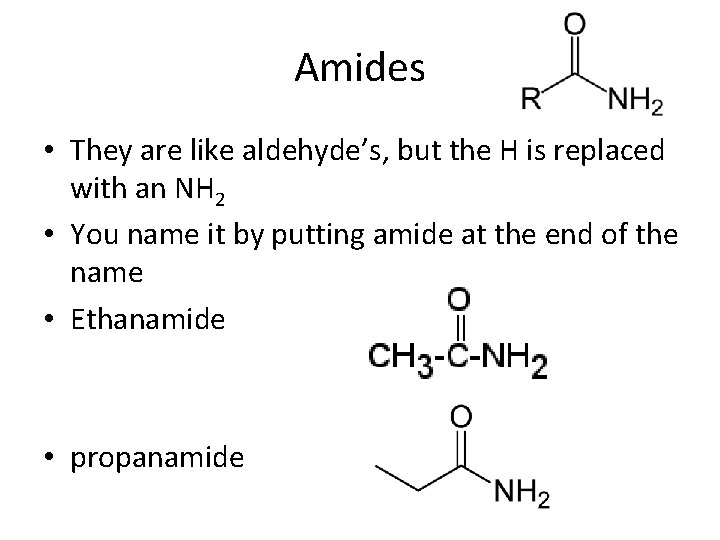
Amides • They are like aldehyde’s, but the H is replaced with an NH 2 • You name it by putting amide at the end of the name • Ethanamide • propanamide
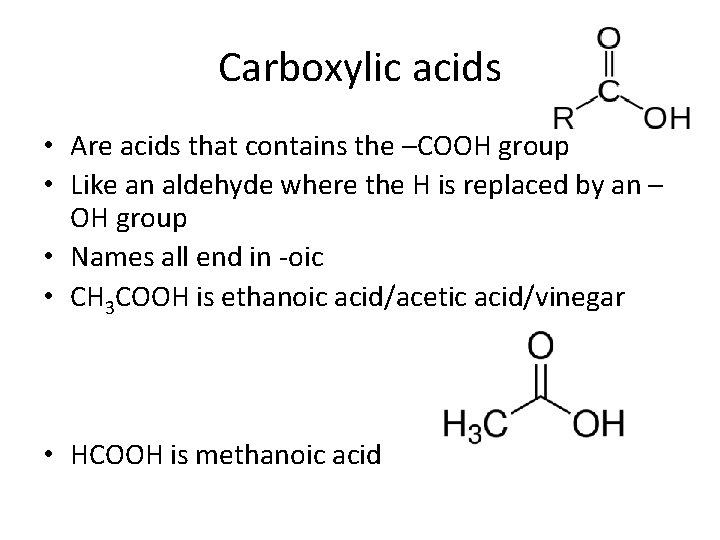
Carboxylic acids • Are acids that contains the –COOH group • Like an aldehyde where the H is replaced by an – OH group • Names all end in -oic • CH 3 COOH is ethanoic acid/acetic acid/vinegar • HCOOH is methanoic acid
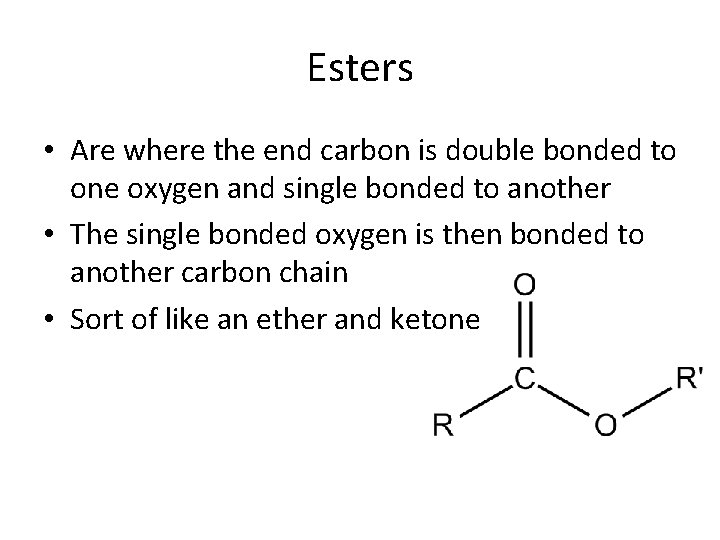
Esters • Are where the end carbon is double bonded to one oxygen and single bonded to another • The single bonded oxygen is then bonded to another carbon chain • Sort of like an ether and ketone
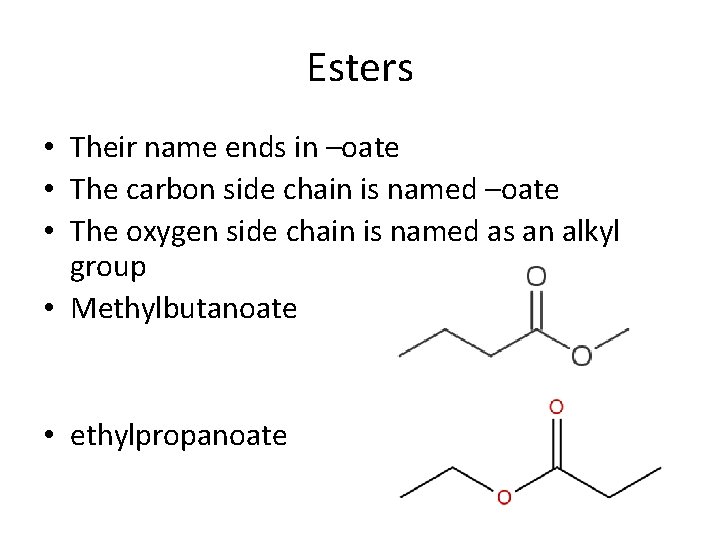
Esters • Their name ends in –oate • The carbon side chain is named –oate • The oxygen side chain is named as an alkyl group • Methylbutanoate • ethylpropanoate

Summary Sheet

Homework • • Page 242 #38 (a, c, e odd) #39 (a, b, e, p, q, r) #44 (a, c, e odd)
- Slides: 42