Organic Chemistry William H Brown Christopher S Foote

Organic Chemistry William H. Brown Christopher S. Foote Brent L. Iverson 11 -1

Ethers & Epoxides Chapter 11 11 -2

Structure u The functional group of an ether is an oxygen atom bonded to two carbon atoms • in dialkyl ethers, oxygen is sp 3 hybridized with bond angles of approximately 109. 5°. • in dimethyl ether, the C-O-C bond angle is 110. 3° 11 -3

Structure • in other ethers, the ether oxygen is bonded to an sp 2 hybridized carbon • in ethyl vinyl ether, for example, the ether oxygen is bonded to one sp 3 hybridized carbon and one sp 2 hybridized carbon 11 -4
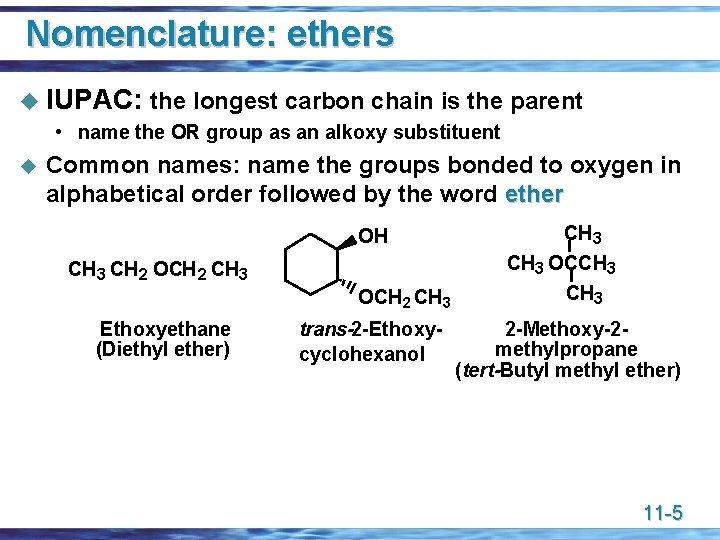
Nomenclature: ethers u IUPAC: the longest carbon chain is the parent • name the OR group as an alkoxy substituent u Common names: name the groups bonded to oxygen in alphabetical order followed by the word ether OH CH 3 OCCH 3 CH 2 OCH 2 CH 3 Ethoxyethane (Diethyl ether) CH 3 trans-2 -Ethoxycyclohexanol CH 3 2 -Methoxy-2 methylpropane (tert-Butyl methyl ether) 11 -5

Nomenclature: ethers u Although cyclic ethers have IUPAC names, their common names are more widely used • IUPAC: prefix ox- shows oxygen in the ring • the suffixes -irane, irane -etane, etane -olane, olane and -ane show three, four, five, and six atoms in a saturated ring 11 -6

Physical Properties u Although ethers are polar compounds, only weak dipole-dipole attractive forces exist between their molecules in the pure liquid state 11 -7

Physical Properties u Boiling points of ethers are • lower than alcohols of comparable MW • close to those of hydrocarbons of comparable MW u Ethers are hydrogen bond acceptors • they are more soluble in H 2 O than are hydrocarbons 11 -8

Preparation of Ethers u Williamson ether synthesis: SN 2 displacement of halide, tosylate, or mesylate by alkoxide ion 11 -9

Preparation of Ethers • yields are highest with methyl and 1° halides, • lower with 2° halides (competing -elimination) • reaction fails with 3° halides ( -elimination only) 11 -10

Preparation of Ethers u Acid-catalyzed dehydration of alcohols • diethyl ether and several other ethers are made on an industrial scale this way • a specific example of an SN 2 reaction in which a poor leaving group (OH-) is converted to a better one (H 2 O) 11 -11

Preparation of Ethers • Step 1: proton transfer gives an oxonium ion • Step 2: nucleophilic displacement of H 2 O by the OH group of the alcohol gives a new oxonium ion 11 -12

Preparation of Ethers Step 3: proton transfer to solvent completes the reaction 11 -13

Preparation of Ethers u Acid-catalyzed addition of alcohols to alkenes • yields are highest using an alkene that can form a stable carbocation • and using methanol or a 1° alcohol that is not prone to undergo acid-catalyzed dehydration 11 -14

Preparation of Ethers • Step 1: protonation of the alkene gives a carbocation • Step 2: reaction of the carbocation (an electrophile) with the alcohol (a nucleophile) gives an oxonium ion 11 -15

Preparation of Ethers Step 3: proton transfer to solvent completes the reaction 11 -16

Cleavage of Ethers u Ethers are cleaved by HX to an alcohol and a haloalkane • cleavage requires both a strong acid and a good nucleophile; therefore, the use of concentrated HI (57%) and HBr (48%) • cleavage by concentrated HCl (38%) is less effective, primarily because Cl- is a weaker nucleophile in water than either I- or Br- 11 -17

Cleavage of Ethers u. A dialkyl ether is cleaved to two moles of haloalkane 11 -18

Cleavage of Ethers • Step 1: proton transfer to the oxygen atom of the ether gives an oxonium ion • Step 2: nucleophilic displacement on the 1° carbon gives a haloalkane and an alcohol • the alcohol is then converted to an haloalkane by another SN 2 reaction 11 -19

Cleavage of Ethers u 3°, allylic, and benzylic ethers are particularly sensitive to cleavage by HX • tert-butyl ethers are cleaved by HCl at room temp • in this case, protonation of the ether oxygen is followed by C-O cleavage to give the tert-butyl cation 11 -20

Oxidation of Ethers u Ethers react with O 2 at a C-H bond adjacent to the ether oxygen to give hydroperoxides • reaction occurs by a radical chain mechanism u Hydroperoxide: a compound containing the OOH group 11 -21

Silyl Ethers as Protecting Groups u When dealing with compounds containing two or more functional groups, it is often necessary to protect one of them (to prevent its reaction) while reacting at the other • suppose you wish to carry out this transformation 11 -22

Silyl Ethers as Protecting Groups • the new C-C bond can be formed by alkylation of an alkyne anion • the OH group, however, is more acidic (p. Ka 16 -18) than the terminal alkyne (p. Ka 25) • treating the compound with one mole of Na. NH 2 will give the alkoxide anion rather than the alkyne anion 11 -23

Silyl Ethers as Protecting Groups u. A protecting group must • add easily to the sensitive group • be resistant to the reagents used to transform the unprotected functional group(s) • be removed easily to regenerate the original functional group u In this chapter, we discuss trimethylsilyl (TMS) and other trialkylsilyl ethers as OH protecting groups 11 -24

Silyl Ethers as Protecting Groups u Silicon is in Group 4 A of the Periodic Table, immediately below carbon • like carbon, it also forms tetravalent compounds such as the following 11 -25

Silyl Ethers as Protecting Groups u An -OH group can be converted to a silyl ether by treating it with a trialkylsilyl chloride in the presence of a 3° amine 11 -26

Silyl Ethers as Protecting Groups • replacement of one of the methyl groups of the TMS group by tert-butyl gives a tert-butyldimethylsilyl (TBDMS) group, which is considerably more stable than the TMS group • other common silyl protecting groups include the TES and TIPS groups 11 -27

Silyl Ethers as Protecting Groups • silyl ethers are unaffected by most oxidizing and reducing agents, and are stable to most nonaqueous acids and bases • the TBDMS group is stable in aqueous solution within the p. H range 2 to 12, which makes it one of the most widely used -OH protecting groups • silyl blocking groups are most commonly removed by treatment with fluoride ion, generally in the form of tetrabutylammonium fluoride 11 -28

Silyl Ethers as Protecting Groups • we can use the TMS group as a protecting group in the conversion of 4 -pentyn-1 -ol to 4 -heptyn-1 -ol 11 -29
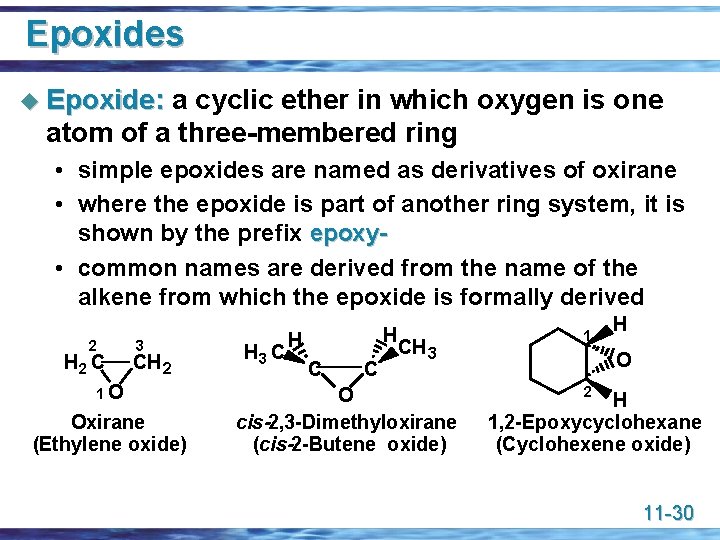
Epoxides u Epoxide: a cyclic ether in which oxygen is one atom of a three-membered ring • simple epoxides are named as derivatives of oxirane • where the epoxide is part of another ring system, it is shown by the prefix epoxy • common names are derived from the name of the alkene from which the epoxide is formally derived 2 H 2 C 3 CH 2 1 O Oxirane (Ethylene oxide) H 3 C H H C C CH 3 O cis-2, 3 -Dimethyloxirane (cis-2 -Butene oxide) 1 H O 2 H 1, 2 -Epoxycyclohexane (Cyclohexene oxide) 11 -30

Synthesis of Epoxides u Ethylene oxide, one of the few epoxides manufactured on an industrial scale, is prepared by air oxidation of ethylene 2 CH 2 = CH 2 + O 2 Ag 2 H 2 C CH 2 O Oxirane (Ethylene oxide) 11 -31
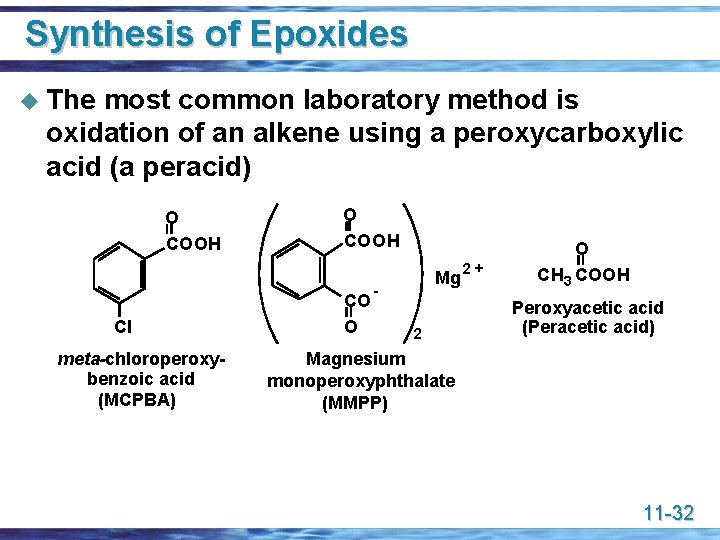
Synthesis of Epoxides u The most common laboratory method is oxidation of an alkene using a peroxycarboxylic acid (a peracid) O COOH Cl meta-chloroperoxybenzoic acid (MCPBA) O COOH CO O Mg 2 2+ O CH 3 COOH Peroxyacetic acid (Peracetic acid) Magnesium monoperoxyphthalate (MMPP) 11 -32

Synthesis of Epoxides u Epoxidation of cyclohexene 11 -33

Synthesis of Epoxides u Epoxidation is stereospecific: • epoxidation of cis-2 -butene gives only cis-2, 3 dimethyloxirane • epoxidation of trans-2 -butene gives only trans-2, 3 dimethyloxirane 11 -34

Synthesis of Epoxides u. A mechanism for alkene epoxidation must take into account that the reaction • takes place in nonpolar solvents, which means that no ions are involved • is stereospecific with retention of the alkene configuration, which means that even though the pi bond is broken, at no time is there free rotation about the remaining sigma bond 11 -35

Synthesis of Epoxides u. A mechanism for alkene epoxidation 11 -36

Synthesis of Epoxides u Epoxides are can also be synthesized via halohydrins • the second step is an internal SN 2 reaction 11 -37

Synthesis of Epoxides • halohydrin formation is both regioselective and stereoselective; for alkenes that show cis, trans isomerism, it is also stereospecific (Section 6. 3 F) • conversion of a halohydrin to an epoxide is stereoselective u Problem: account for the fact that conversion of cis-2 butene to an epoxide by the halohydrin method gives only cis-2, 3 -dimethyloxirane H H 3 C H 1. Cl 2 , H 2 O C C CH 3 2. Na. OH, H O 2 cis-2 -Butene H 3 C H CH 3 O cis-2, 3 -Dimethyloxirane 11 -38

Synthesis of Epoxides u Sharpless epoxidation • stereospecific and enantioselective 11 -39

Reactions of Epoxides u Ethers are not normally susceptible to attack by nucleophiles u Because of the strain associated with the threemembered ring, epoxides readily undergo a variety of ring-opening reactions 11 -40

Reactions of Epoxides u Acid-catalyzed ring opening • in the presence of an acid catalyst, such as sulfuric acid, epoxides are hydrolyzed to glycols O + H 2 O Oxirane (Ethylene oxide) H+ HO OH 1, 2 -Ethanediol (Ethylene glycol) 11 -41

Reactions of Epoxides Step 1: proton transfer to oxygen gives a bridged oxonium ion intermediate Step 2: backside attack by water (a nucleophile) on the oxonium ion (an electrophile) opens the ring Step 3: proton transfer to solvent completes the reaction 11 -42

Reactions of Epoxides u Attack of the nucleophile on the protonated epoxide shows anti stereoselectivity • hydrolysis of an epoxycycloalkane gives a trans-1, 2 diol 11 -43

Reactions of Epoxides u Compare the stereochemistry of the glycols formed by these two methods 11 -44

Epoxides • the value of epoxides is the variety of nucleophiles that will open the ring and the combinations of functional groups that can be prepared from them 11 -45
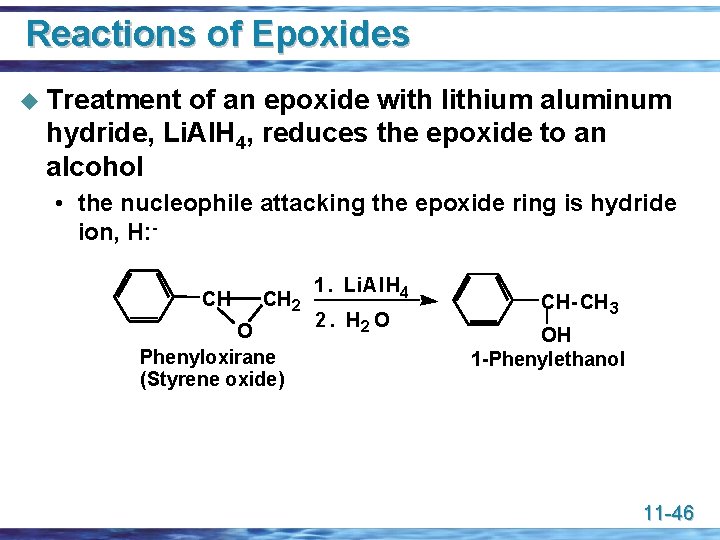
Reactions of Epoxides u Treatment of an epoxide with lithium aluminum hydride, Li. Al. H 4, reduces the epoxide to an alcohol • the nucleophile attacking the epoxide ring is hydride ion, H: CH CH 2 O Phenyloxirane (Styrene oxide) 1. Li. Al. H 4 2. H 2 O CH- CH 3 OH 1 -Phenylethanol 11 -46

Ethylene Oxide • ethylene oxide is a valuable building block for organic synthesis because each of its carbons has a functional group 11 -47

Ethylene Oxide • part of the local anesthetic procaine is derived from ethylene oxide • the hydrochloride salt of procaine is marketed under the trade name Novocaine 11 -48

Epichlorohydrin u The epoxide epichlorohydrin is also a valuable building block because each of its three carbons contains a reactive functional group • epichlorohydrin is synthesized from propene 11 -49

Epichlorohydrin • the characteristic structural feature of a product derived from epichlorohydrin is a three-carbon unit with -OH on the middle carbon, and a carbon, nitrogen, oxygen, or sulfur nucleophile on the two end carbons 11 -50

Epichlorohydrin • an example of a compound containing the threecarbon skeleton of epichlorohydrin is nadolol, a adrenergic blocker with vasodilating activity 11 -51

Crown Ethers u Crown ether: a cyclic polyether derived from ethylene glycol or a substituted ethylene glycol • the parent name is crown, preceded by a number describing the size of the ring and followed by the number of oxygen atoms in the ring 11 -52

Crown Ethers u The diameter of the cavity created by the repeating oxygen atoms is comparable to the diameter of alkali metal cations • 18 -crown-6 provides very effective solvation for K+ 11 -53

Thioethers u The sulfur analog of an ether • IUPAC name: select the longest carbon chain as the parent and name the sulfur-containing substituent as an alkylsulfanyl group • common name: list the groups bonded to sulfur followed by the word sulfide 11 -54

Nomenclature u Disulfide: contains an -S-S group • IUPAC name: select the longest carbon chain as the parent and name the disulfide-containing substituent as an alkyldisulfanyl group • Common name: list the groups bonded to sulfur and add the word disulfide 11 -55

Preparation of Sulfides u Symmetrical sulfides: treat one mole of Na 2 S with two moles of a haloalkane 11 -56

Preparation of Sulfides u Unsymmetrical sulfides: convert a thiol to its sodium salt and then treat this salt with an alkyl halide (a variation on the Williamson ether synthesis) 11 -57

Oxidation Sulfides u Sulfides can be oxidized to sulfoxides and sulfones by the proper choice of experimental conditions 11 -58
Ethers & Epoxides End of Chapter 11 11 -59
- Slides: 59