Organic Chemistry Second Edition David Klein Chapter 11












- Slides: 33

Organic Chemistry Second Edition David Klein Chapter 11 Radical Reactions Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2 e

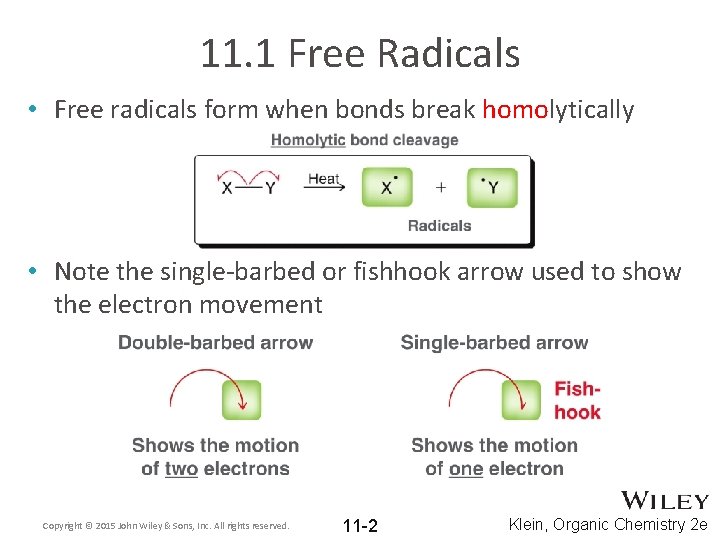
11. 1 Free Radicals • Free radicals form when bonds break homolytically • Note the single-barbed or fishhook arrow used to show the electron movement Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -2 Klein, Organic Chemistry 2 e

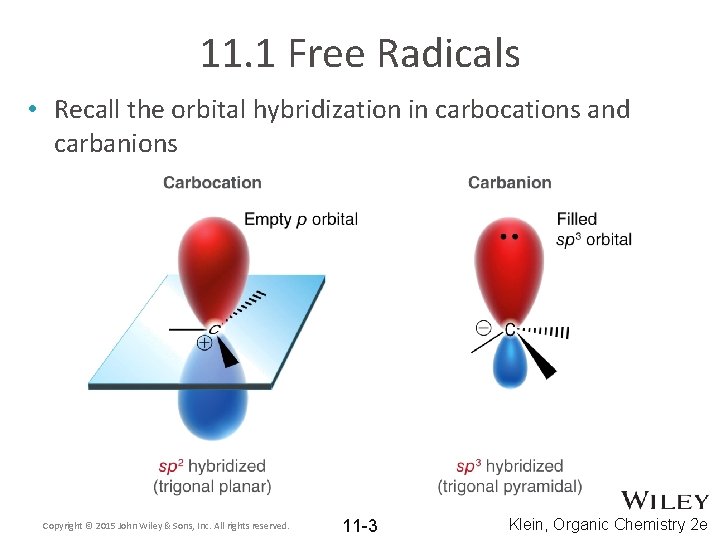
11. 1 Free Radicals • Recall the orbital hybridization in carbocations and carbanions Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -3 Klein, Organic Chemistry 2 e

11. 1 Free Radicals • Free radicals can be thought of as sp 2 hybridized or quickly interconverting sp 3 hybridized sp 2 hybridized Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -4 Klein, Organic Chemistry 2 e

11. 1 Free Radical Stability • Free radicals do not have a formal charge but are unstable because of an incomplete octet • Groups that can push (donate) electrons toward the free radical will help to stabilize it. WHY? HOW? Consider hyperconjugation Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -5 Klein, Organic Chemistry 2 e

11. 1 Free Radical Resonance • Drawing resonance for free radicals using fishhook arrows to show electron movement • Remember, for resonance, the arrows don’t ACTUALLY show electron movement. WHY? • Draw the resonance hybrid for an allyl radical • HOW and WHY does resonance affect the stability of the free radical? Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -6 Klein, Organic Chemistry 2 e

11. 1 Free Radical Resonance • The benzylic radical is a hybrid that consists of 4 contributors • Draw the remaining contributors • Draw the hybrid Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -7 Klein, Organic Chemistry 2 e

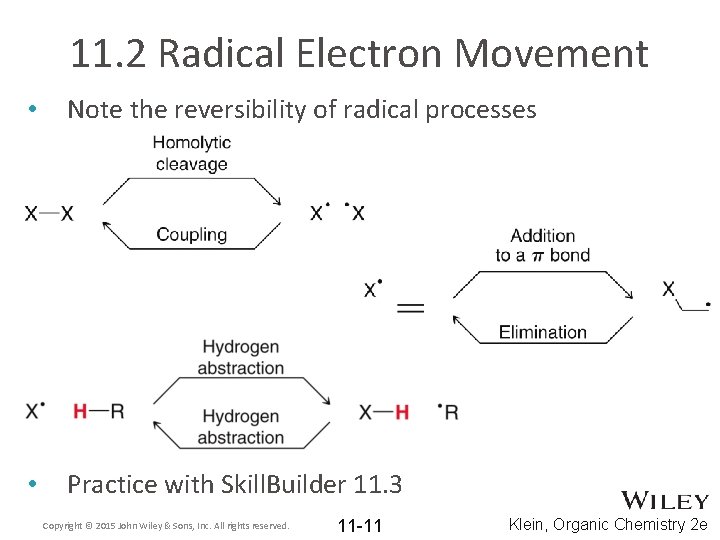
11. 2 Radical Electron Movement • Free radical electron movement is quite different from electron movement in ionic reactions • For example, free radicals don’t undergo rearrangement • There are SIX key arrow-pushing patterns that we will discuss Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -8 Klein, Organic Chemistry 2 e

11. 2 Radical Electron Movement 1. Homolytic Cleavage: initiated by light or heat 2. Addition to a pi bond 3. Hydrogen abstraction: NOT the same as proton transfer 4. Halogen abstraction Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -9 Klein, Organic Chemistry 2 e

11. 2 Radical Electron Movement 5. Elimination: the radical from the α carbon is pushed toward the β carbon to eliminate a group on the β carbon (reverse of addition to a pi bond) – The –X group is NOT a leaving group. WHY? 6. Coupling: the reverse of homolytic cleavage • Note that radical electron movement generally involves 2 or 3 fishhook arrows Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -10 Klein, Organic Chemistry 2 e

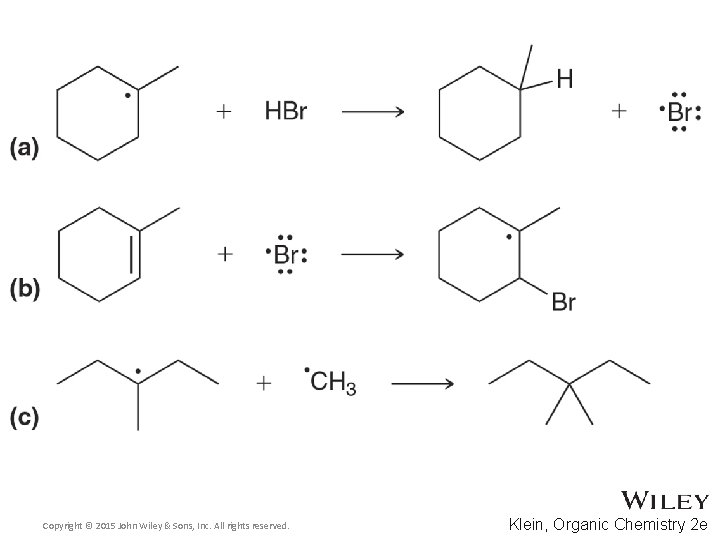
11. 2 Radical Electron Movement • Note the reversibility of radical processes • Practice with Skill. Builder 11. 3 Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -11 Klein, Organic Chemistry 2 e

Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2 e

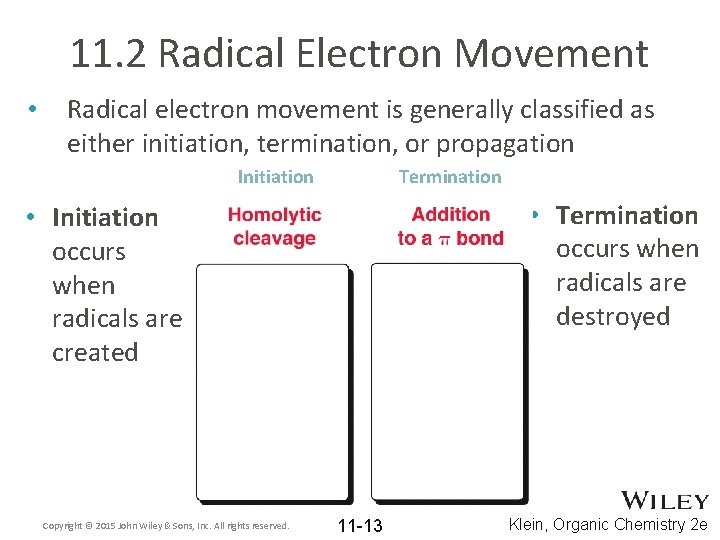
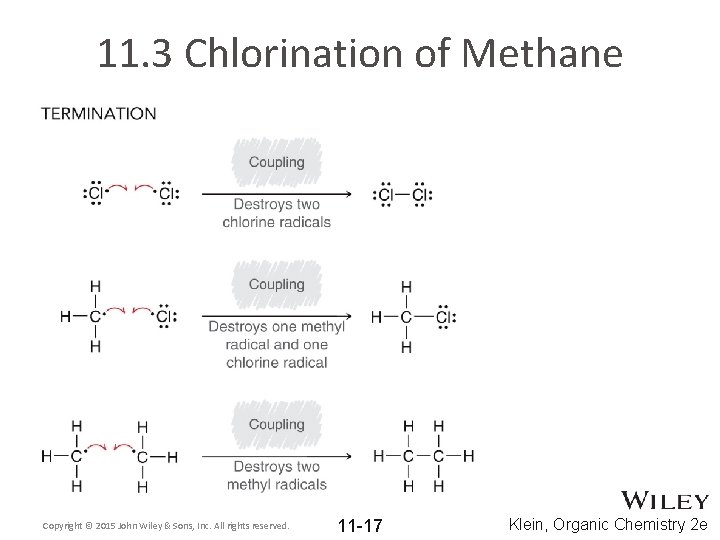
11. 2 Radical Electron Movement • Radical electron movement is generally classified as either initiation, termination, or propagation Initiation Termination • Termination occurs when radicals are destroyed • Initiation occurs when radicals are created Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -13 Klein, Organic Chemistry 2 e

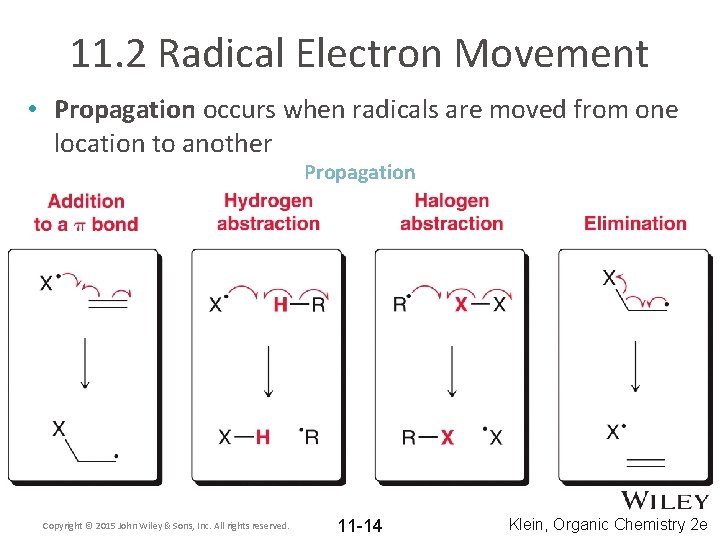
11. 2 Radical Electron Movement • Propagation occurs when radicals are moved from one location to another Propagation Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -14 Klein, Organic Chemistry 2 e

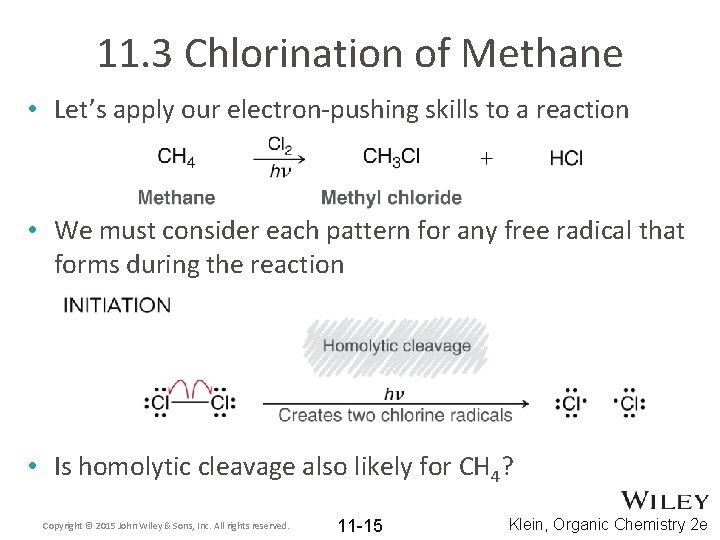
11. 3 Chlorination of Methane • Let’s apply our electron-pushing skills to a reaction • We must consider each pattern for any free radical that forms during the reaction • Is homolytic cleavage also likely for CH 4? Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -15 Klein, Organic Chemistry 2 e

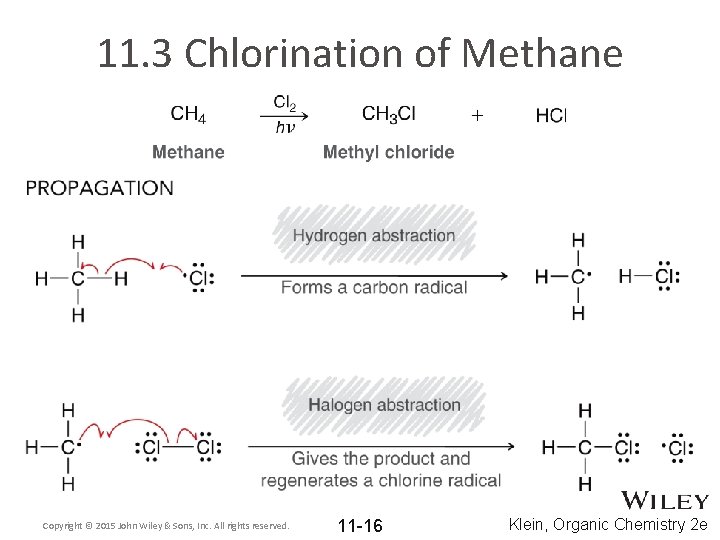
11. 3 Chlorination of Methane Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -16 Klein, Organic Chemistry 2 e

11. 3 Chlorination of Methane Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -17 Klein, Organic Chemistry 2 e

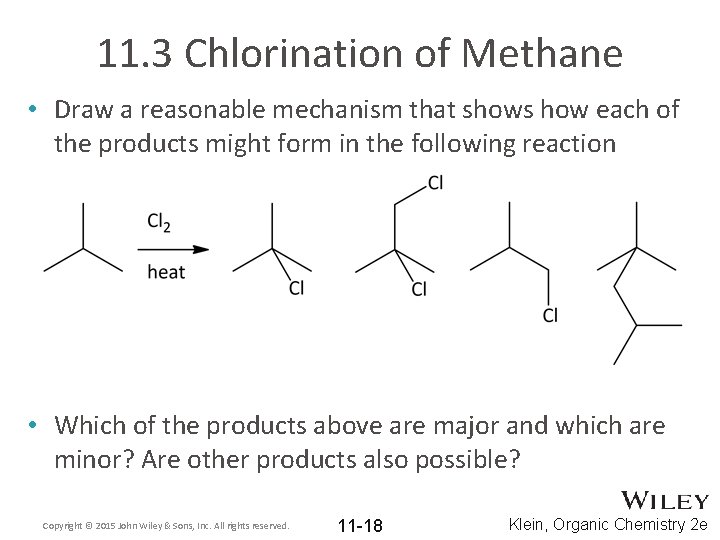
11. 3 Chlorination of Methane • Draw a reasonable mechanism that shows how each of the products might form in the following reaction • Which of the products above are major and which are minor? Are other products also possible? Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -18 Klein, Organic Chemistry 2 e

Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2 e

11. 7 Allylic Halogenation • When an allylic hydrogen is abstracted, it leaves behind an allylic free radical that is stabilized by resonance • Based on the high selectivity of bromination that we discussed, you might expect bromination to occur as shown below Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -20 Klein, Organic Chemistry 2 e

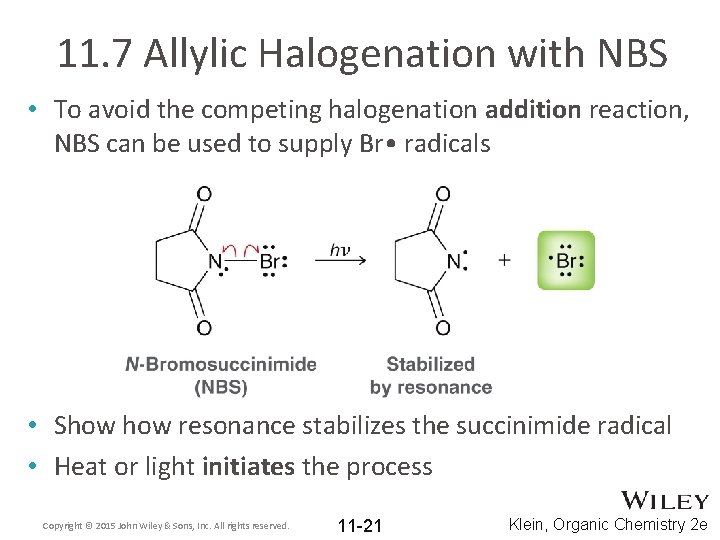
11. 7 Allylic Halogenation with NBS • To avoid the competing halogenation addition reaction, NBS can be used to supply Br • radicals • Show resonance stabilizes the succinimide radical • Heat or light initiates the process Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -21 Klein, Organic Chemistry 2 e

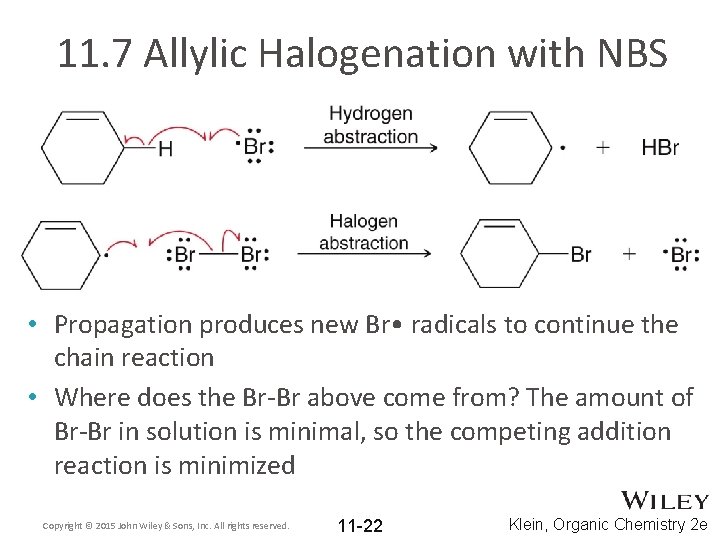
11. 7 Allylic Halogenation with NBS • Propagation produces new Br • radicals to continue the chain reaction • Where does the Br-Br above come from? The amount of Br-Br in solution is minimal, so the competing addition reaction is minimized Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -22 Klein, Organic Chemistry 2 e

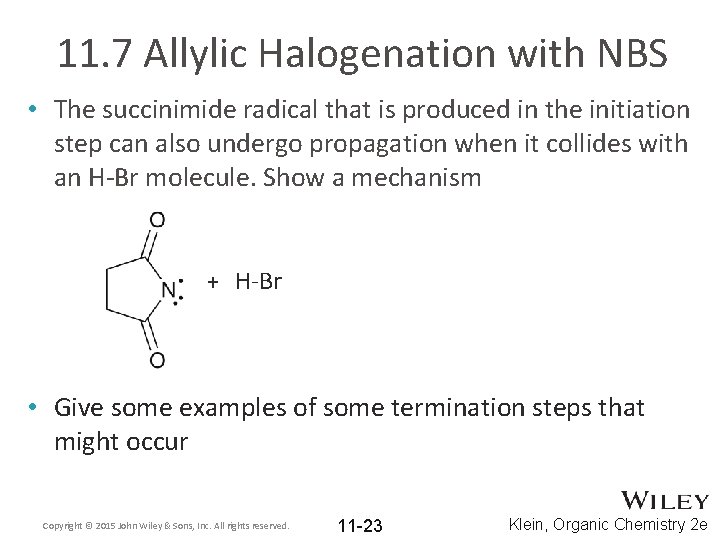
11. 7 Allylic Halogenation with NBS • The succinimide radical that is produced in the initiation step can also undergo propagation when it collides with an H-Br molecule. Show a mechanism + H-Br • Give some examples of some termination steps that might occur Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -23 Klein, Organic Chemistry 2 e

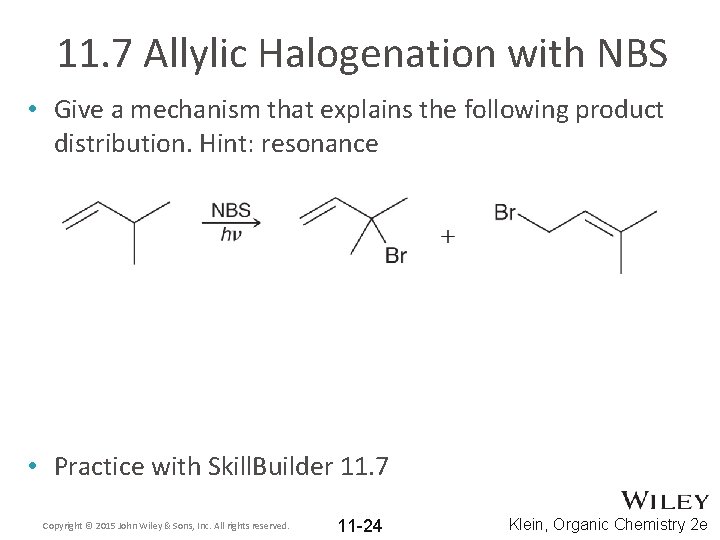
11. 7 Allylic Halogenation with NBS • Give a mechanism that explains the following product distribution. Hint: resonance • Practice with Skill. Builder 11. 7 Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -24 Klein, Organic Chemistry 2 e

Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2 e

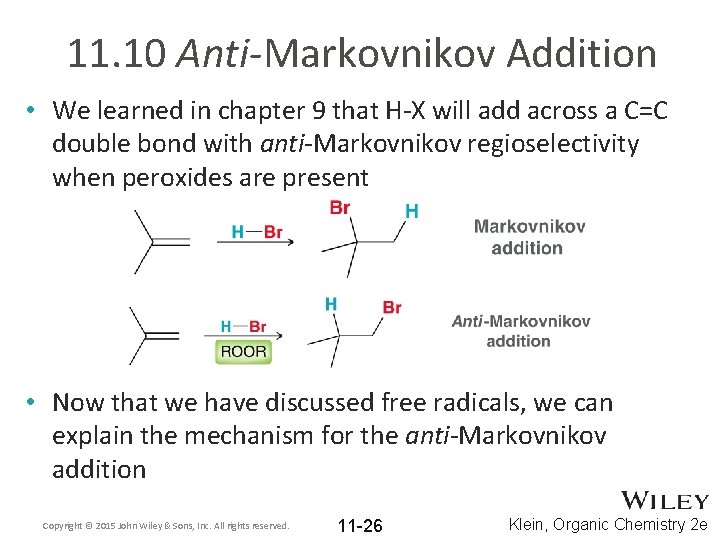
11. 10 Anti-Markovnikov Addition • We learned in chapter 9 that H-X will add across a C=C double bond with anti-Markovnikov regioselectivity when peroxides are present • Now that we have discussed free radicals, we can explain the mechanism for the anti-Markovnikov addition Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -26 Klein, Organic Chemistry 2 e

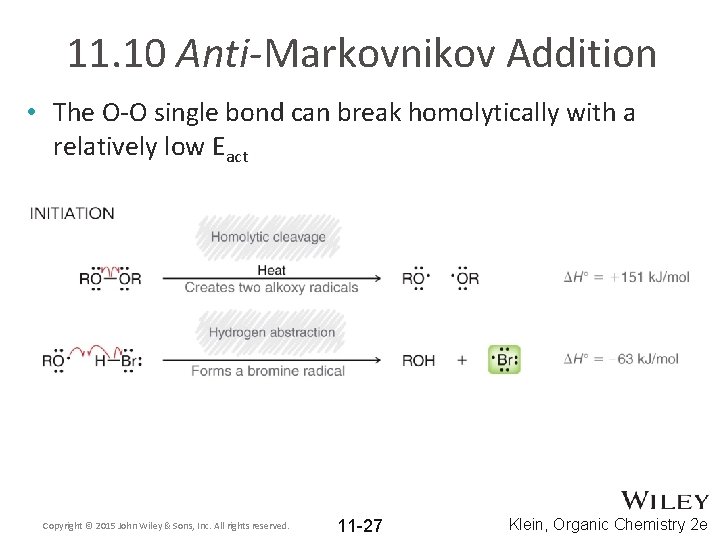
11. 10 Anti-Markovnikov Addition • The O-O single bond can break homolytically with a relatively low Eact Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -27 Klein, Organic Chemistry 2 e

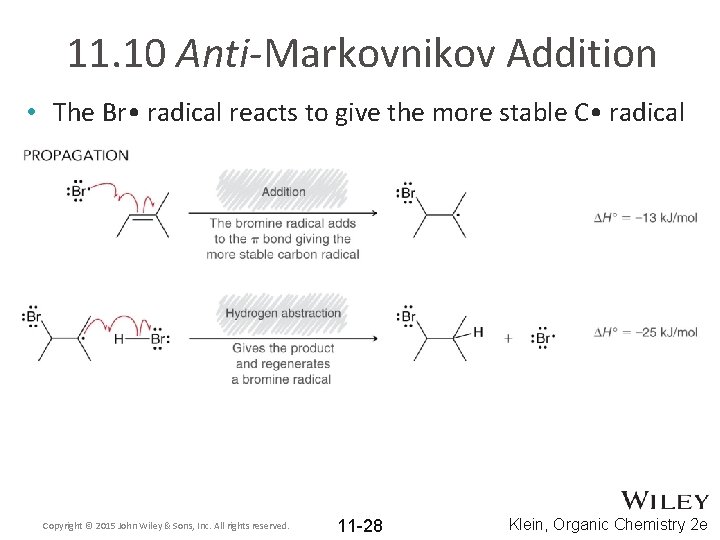
11. 10 Anti-Markovnikov Addition • The Br • radical reacts to give the more stable C • radical Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -28 Klein, Organic Chemistry 2 e

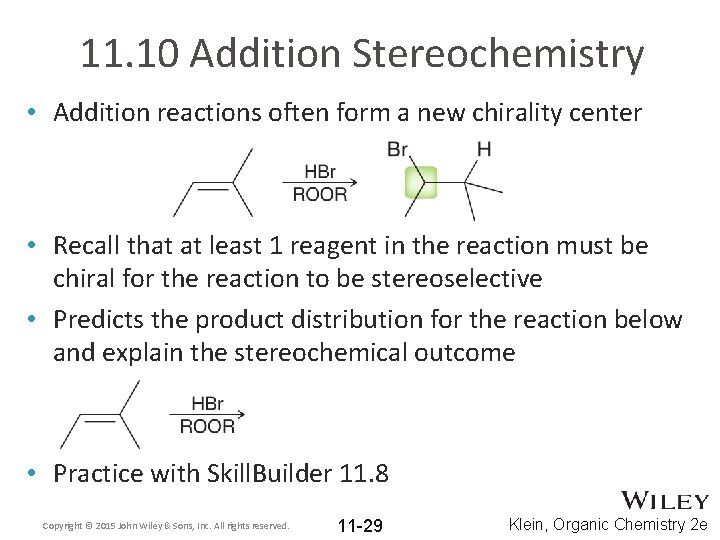
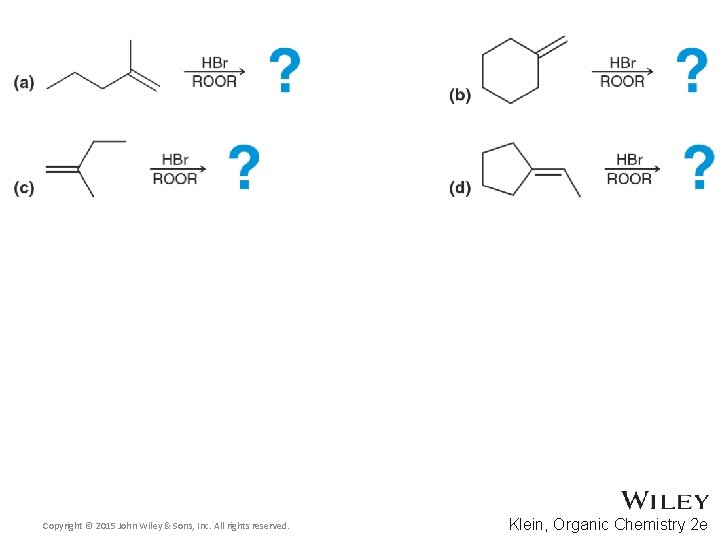
11. 10 Addition Stereochemistry • Addition reactions often form a new chirality center • Recall that at least 1 reagent in the reaction must be chiral for the reaction to be stereoselective • Predicts the product distribution for the reaction below and explain the stereochemical outcome • Practice with Skill. Builder 11. 8 Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -29 Klein, Organic Chemistry 2 e

Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. Klein, Organic Chemistry 2 e

11. 13 Synthetic Utility of Halogenation • Radical chlorination and bromination are both useful processes • Recall that bromination is more selective. WHY? • Temperature can be used to help avoid polysubstitution. HOW? Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -31 Klein, Organic Chemistry 2 e

11. 13 Synthetic Utility of Halogenation • Chlorination can be useful with highly symmetrical substrates • It is difficult to avoid polysubstitution. WHY? • The synthetic utility of halogenation is limited – Chlorination is difficult to control – Bromination requires a substrate with 1 site that is significantly more reactive than all others Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -32 Klein, Organic Chemistry 2 e

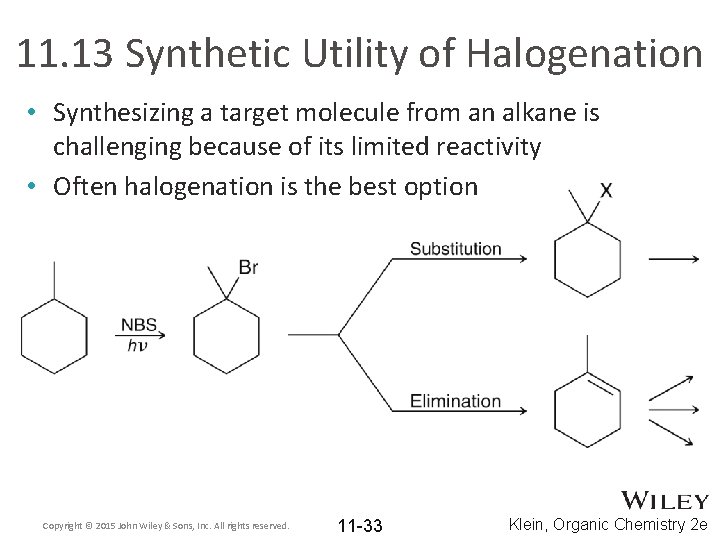
11. 13 Synthetic Utility of Halogenation • Synthesizing a target molecule from an alkane is challenging because of its limited reactivity • Often halogenation is the best option Copyright © 2015 John Wiley & Sons, Inc. All rights reserved. 11 -33 Klein, Organic Chemistry 2 e