Organic Chemistry Hydrocarbons Grade 10 Organic Chemistry Is

Organic Chemistry Hydrocarbons Grade 10

Organic Chemistry • Is the study of carbon-containing compounds except carbon dioxide, carbon monoxide and carbonates. • What is made from organic materials? –All living organisms. –All petroleum products. –Most drugs and medicines.

Hydrocarbons • Hydrocarbons are organic substances that are made up of carbon and hydrogen only. • Which of the following is/are hydrocarbon? CH 4 C 2 H 5 OH CO 2 C 6 H 6

Alkanes • Alkanes are hydrocarbons obtained directly from crude oil. • Alkanes are saturated hydrocarbons with a general formula C n. H 2 n+2 • Saturated means : –the molecule has no C=C double bonds, –only carbon-carbon single bonds –the maximum number of atoms. – no atoms can add to it.

Finding the general formula of Alkanes • The general formula is C n H 2 n+2 • n is the number of carbon atoms. • For n=1 C 1 H 2(1)+2 CH 4 • For n=2 C 2 H 2(2)+2 C 2 H 6 • For n=3 C 3 H 2(3)+2 C 3 H 8 • For n=4 C 4 H 2(4)+2 C 4 H 10 • For n=5 C 5 H 2(5)+2 C 5 H 12

Objective 3. 1 Explain series. the term homologous

Homologous series • A homologous series is a family of compounds which has : v Same general formula. v Similar chemical properties v Successive members differ by CH 2 v Gradual change in their physical properties melting/boiling points, solubility etc. v. Alkanes belong to the same homologous series.

Naming alkanes • The name of the alkane starts with a part that indicates the number of carbon atom and ends with –ane • When n=1 meth+ane = methane • When n=2 eth+ane = ethane • When n=3 prop+ane= propane • When n=4 but+ane = butane • When n=5 pent+ane= pentane
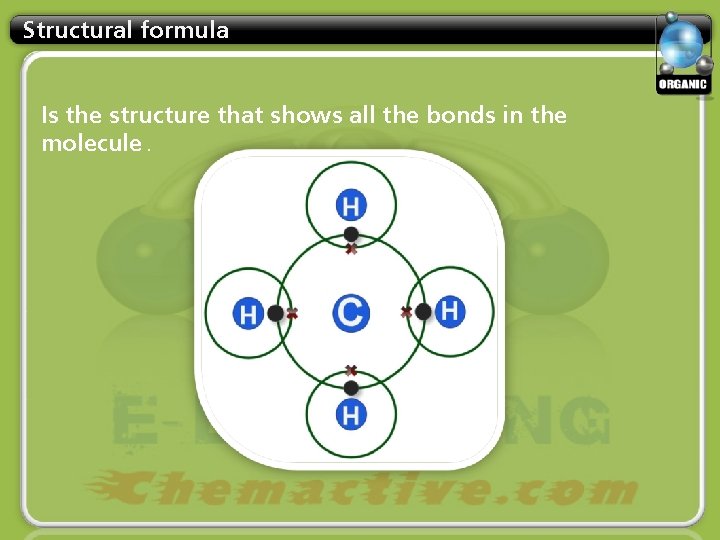
Structural formula Is the structure that shows all the bonds in the molecule.
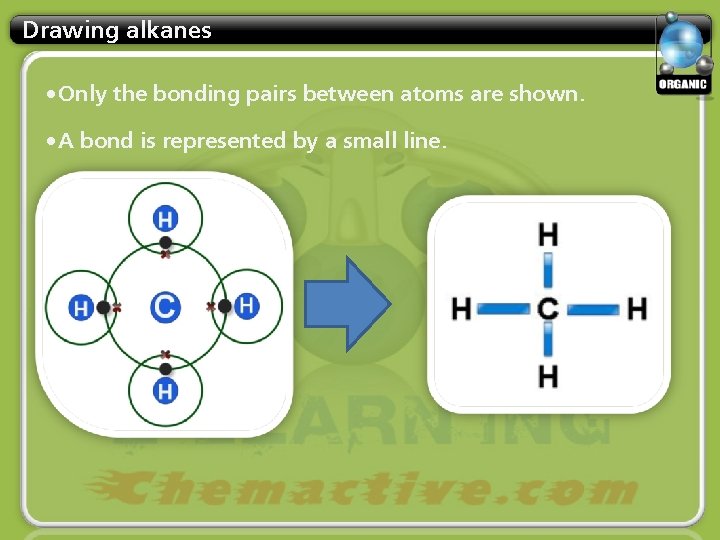
Drawing alkanes • Only the bonding pairs between atoms are shown. • A bond is represented by a small line.
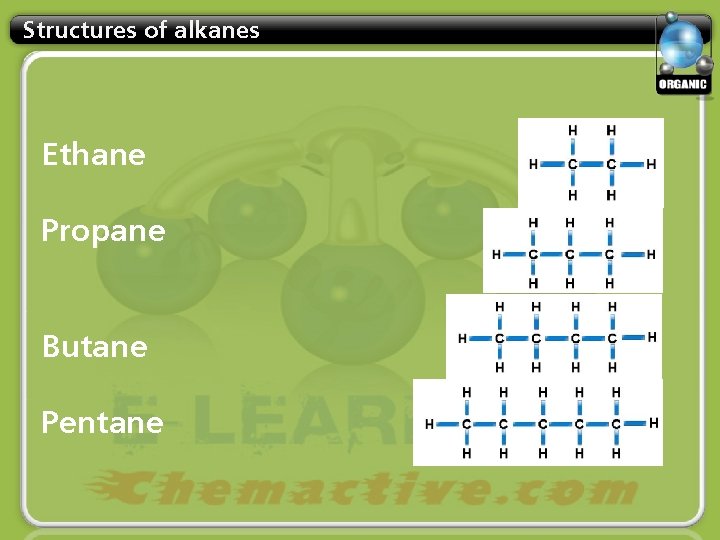
Structures of alkanes Ethane Propane Butane Pentane

Alkenes • These are obtained from the cracking of alkanes. • They are unsaturated hydrocarbons • They form a homologous series with a general formula C n. H 2 n • Unsaturated means : –the molecule has C=C double bonds. – we can add atoms to it.

Finding the general formula of Alkenes • The general formula is C n H 2 n • n is the number of carbon atoms. • For n=1 C 1 H 2(1) CH 2 NOT FOUND • For n=2 C 2 H 2(2) C 2 H 4 • For n=3 C 3 H 2(3) C 3 H 6 • For n=4 C 4 H 2(4) C 4 H 8 • For n=5 C 5 H 2(5) C 5 H 10

Naming alkenes • The name of the alkene starts with a part that indicates the number of carbon atom and ends with –ene • When n=1 meth+ene = methene • When n=2 eth+ene = ethene • When n=3 prop+ene= propene • When n=4 but+ene = butene • When n=5 pent+ene= pentene

Structures of alkanes Ethene Propene Butene Pentene
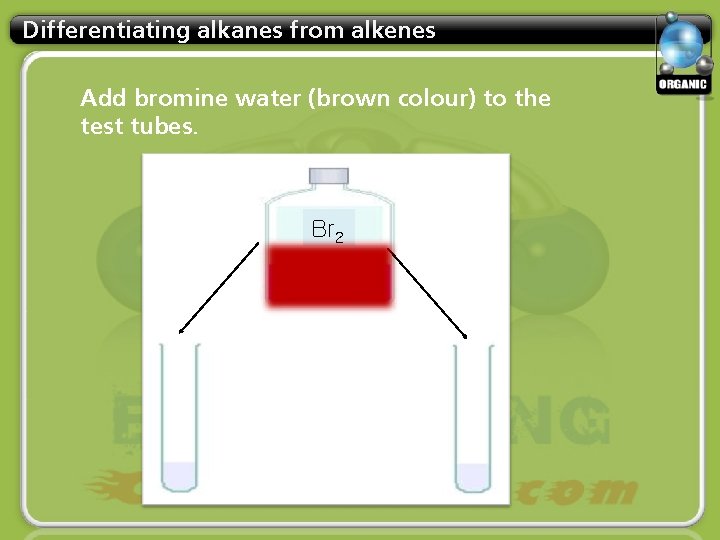
Differentiating alkanes from alkenes Add bromine water (brown colour) to the test tubes. Br 2

Differentiating alkanes from alkenes The colour will disappear in one tube only. Which tube is it?

Differentiating alkanes from alkenes • Keep in mind that the brown colour is due to the presence of Br-Br. . When Bromine is added to an alkene, the Br-Br bond will break and the double bond in the alkene will break too. . An addition reaction will occur and the product formed will not contain Br-Br. . This means the brown colour will disappear.
- Slides: 18