Solomons Organic Chemistry Ver 9 Chapter 22 Carbohydrates

Solomons, Organic Chemistry, Ver. 9 Chapter 22 Carbohydrates Chapter 22
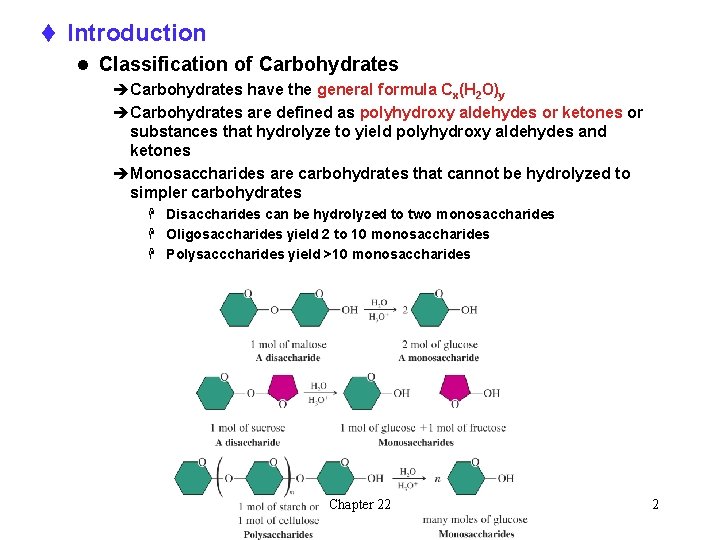
t Introduction l Classification of Carbohydrates èCarbohydrates have the general formula Cx(H 2 O)y èCarbohydrates are defined as polyhydroxy aldehydes or ketones or substances that hydrolyze to yield polyhydroxy aldehydes and ketones èMonosaccharides are carbohydrates that cannot be hydrolyzed to simpler carbohydrates H Disaccharides can be hydrolyzed to two monosaccharides H Oligosaccharides yield 2 to 10 monosaccharides H Polysacccharides yield >10 monosaccharides Chapter 22 2

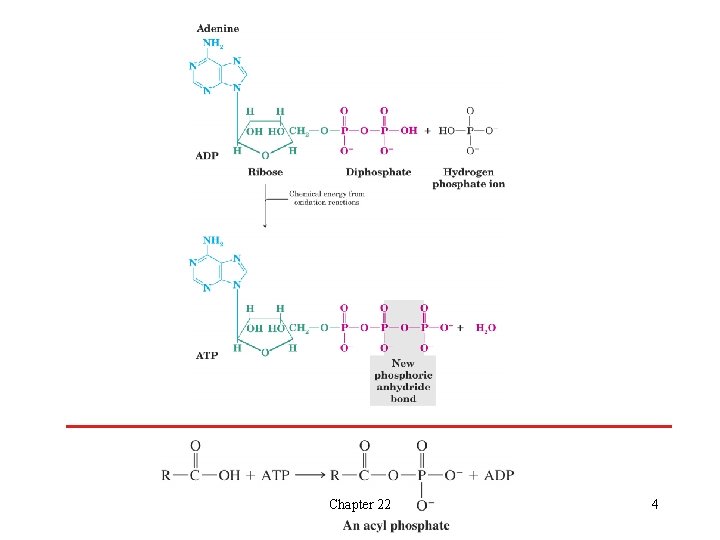
l Photosynthesis and Carbohydrate Metabolism èCarbohydrates are synthesized in plants by photosynthesis H Light from the sun is absorbed by chlorophyll and this is converted to the energy necessary to biosynthesize carbohydrates èCarbohydrates act as a repository of solar energy H The energy is released when animals or plants metabolize carbohydrates èMuch of the energy released by oxidation of glucose is trapped in the molecule adenosine triphosphate (ATP) H The phosphoric anhydride bond formed when adenosine triphosphate (ADP) is phosphorylated to make ATP is the repository of this energy H This chemical energy is released when ATP is hydrolyzed or a new anhydride linkage is created Chapter 22 3
Chapter 22 4
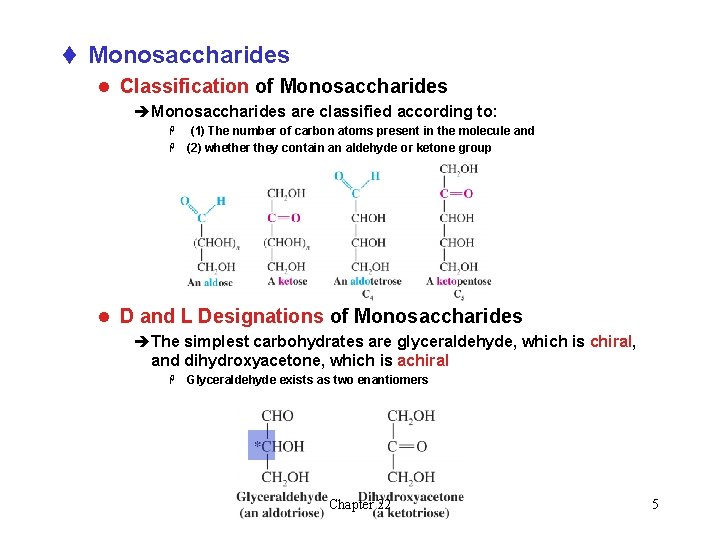
t Monosaccharides l Classification of Monosaccharides èMonosaccharides are classified according to: (1) The number of carbon atoms present in the molecule and H (2) whether they contain an aldehyde or ketone group H l D and L Designations of Monosaccharides èThe simplest carbohydrates are glyceraldehyde, which is chiral, and dihydroxyacetone, which is achiral H Glyceraldehyde exists as two enantiomers Chapter 22 5
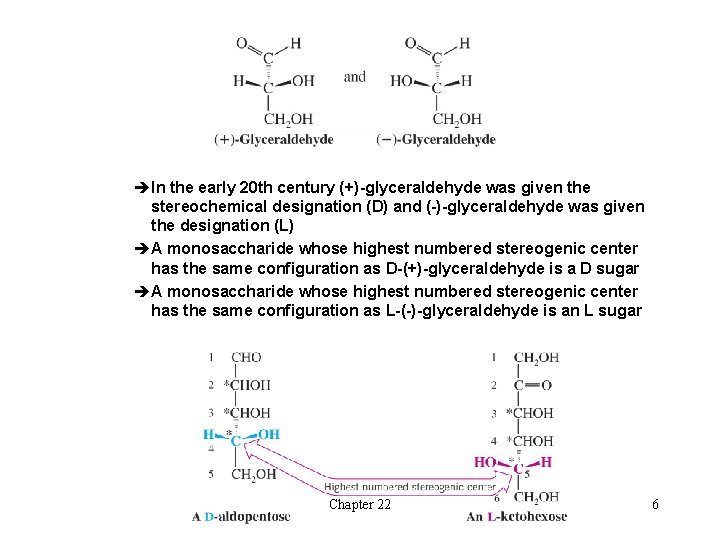
èIn the early 20 th century (+)-glyceraldehyde was given the stereochemical designation (D) and (-)-glyceraldehyde was given the designation (L) èA monosaccharide whose highest numbered stereogenic center has the same configuration as D-(+)-glyceraldehyde is a D sugar èA monosaccharide whose highest numbered stereogenic center has the same configuration as L-(-)-glyceraldehyde is an L sugar Chapter 22 6

l Structural Formulas for Monosaccharides èFischer projections are used to represent stereochemistry in carbohydrates èIn Fischer projections horizontal lines are understood to project out of the plane toward the reader and vertical lines are understood to project behind the plane H A Fischer projection cannot be removed from the plane of the paper or turned 90 o and still represent the molecule accurately èGlucose exists primarily in two cyclic hemiacetal forms that are diastereomers of each other H The cyclic hemiacetal forms interconvert via the open-chain form H The cyclic hemiacetals differ only in configuration at C 1 and are called anomers H The carbon at which their configurations differ is called the anomeric carbon H The a-anomer has the C 1 hydroxyl trans to the -CH 2 OH group H The b-anomer has the C 1 hydroxyl cis to the -CH 2 OH group èThe flat cyclic representation of carbohydrates is called a Haworth formula H Cyclic glucose actually exists in the chair form Chapter 22 7
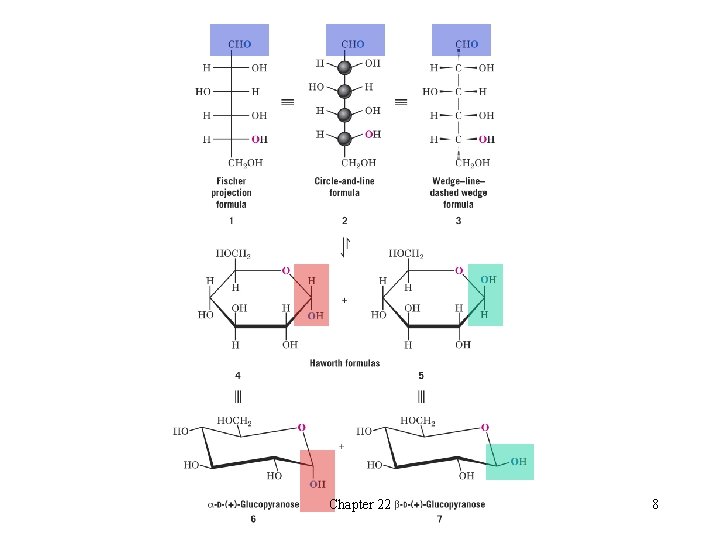
Chapter 22 8
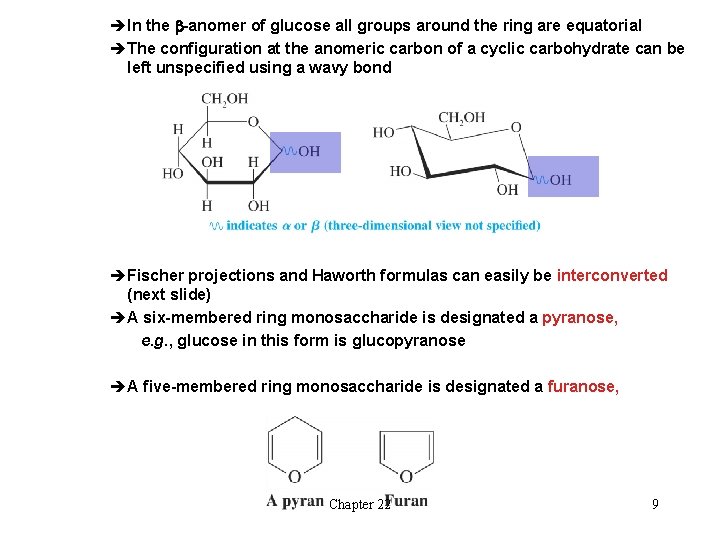
èIn the b-anomer of glucose all groups around the ring are equatorial èThe configuration at the anomeric carbon of a cyclic carbohydrate can be left unspecified using a wavy bond èFischer projections and Haworth formulas can easily be interconverted (next slide) èA six-membered ring monosaccharide is designated a pyranose, e. g. , glucose in this form is glucopyranose èA five-membered ring monosaccharide is designated a furanose, Chapter 22 9
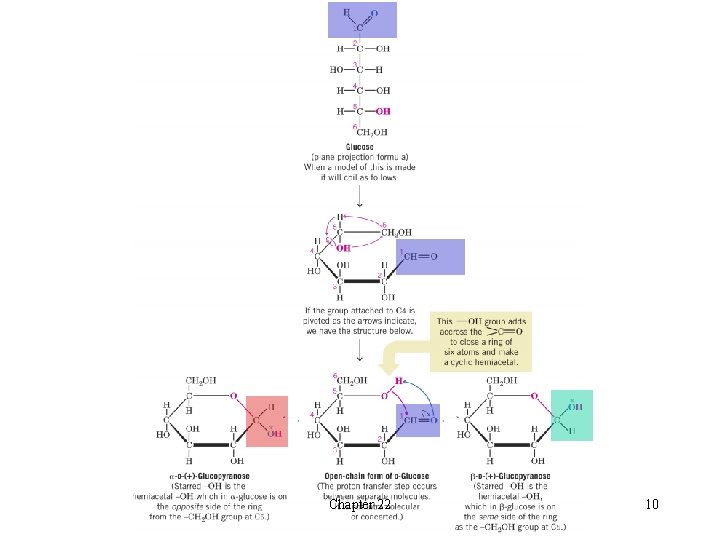
Chapter 22 10

t Mutarotation èThe a- and b-forms of glucose can be isolated separately H Pure a-glucose has a specific rotation of +112 o H Pure b-glucose has a specific rotation of +18. 7 o èWhen either form of glucose is allowed to stand in aqueous solution, the specific rotation of the solution slowly changes to +52. 7 o H It does not matter whether one starts with pure a- or b-glucose èMutarotation is the change in optical rotation as an equilibrium mixture of anomers forms èMutarotation of glucose results in an equilibrium mixture of 36% a-glucose and 64% b-glucose H The more stable b-glucose form predominates H A very small amount of the open-chain form exists in this equilibrium Chapter 22 11
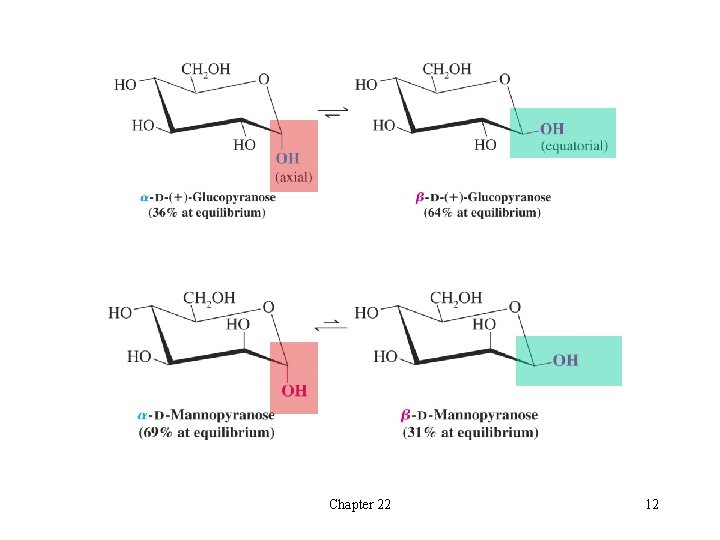
Chapter 22 12
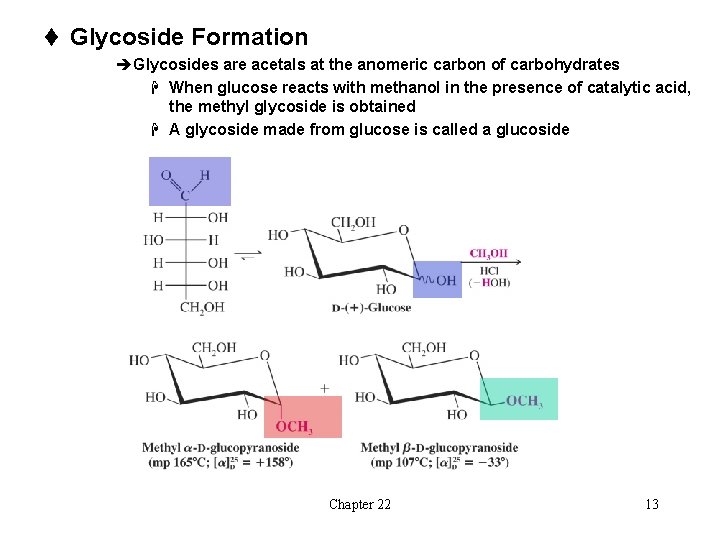
t Glycoside Formation èGlycosides are acetals at the anomeric carbon of carbohydrates H When glucose reacts with methanol in the presence of catalytic acid, the methyl glycoside is obtained H A glycoside made from glucose is called a glucoside Chapter 22 13
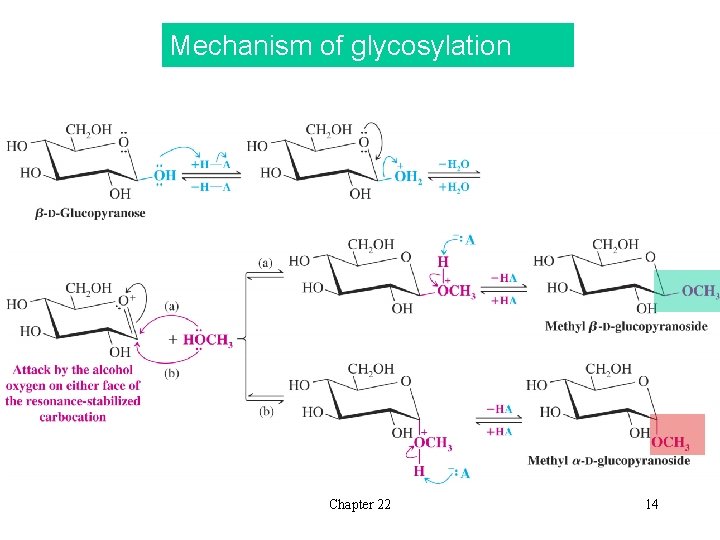
Mechanism of glycosylation Chapter 22 14
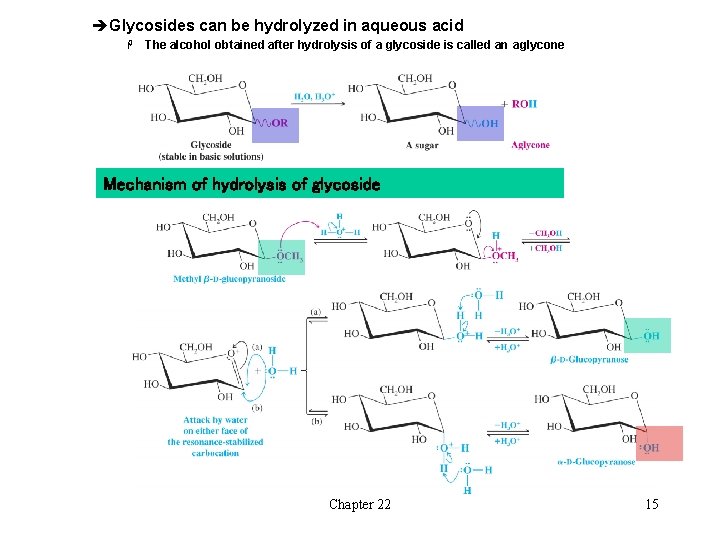
èGlycosides can be hydrolyzed in aqueous acid H The alcohol obtained after hydrolysis of a glycoside is called an aglycone Mechanism of hydrolysis of glycoside Chapter 22 15

t Other Reactions of Monosaccharides l Enolization, Tautomerization and Isomerization èMonosaccharides dissolved in aqueous base will isomerize by a series of keto-enol tautomerizations H A solution of D-glucose containing calcium hydroxide will form several products, including D-fructose and D-mannose èA monosaccharide can be protected from keto-enol tautomerization by conversion to a glycoside Chapter 22 16
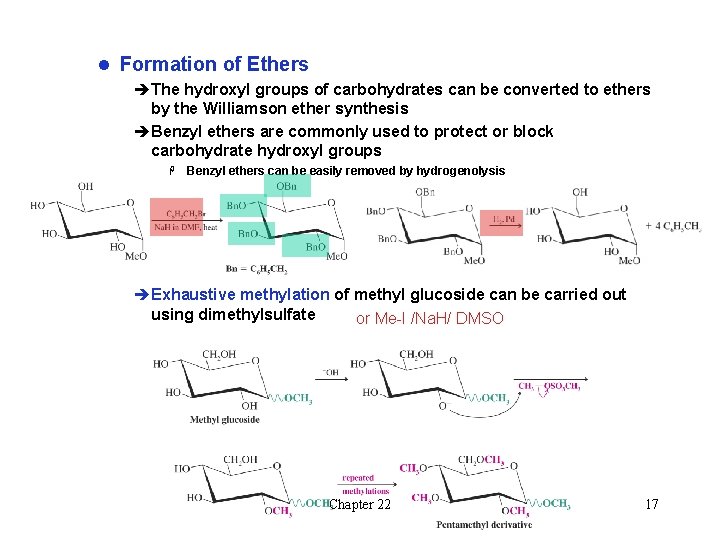
l Formation of Ethers èThe hydroxyl groups of carbohydrates can be converted to ethers by the Williamson ether synthesis èBenzyl ethers are commonly used to protect or block carbohydrate hydroxyl groups H Benzyl ethers can be easily removed by hydrogenolysis èExhaustive methylation of methyl glucoside can be carried out using dimethylsulfate or Me-I /Na. H/ DMSO Chapter 22 17
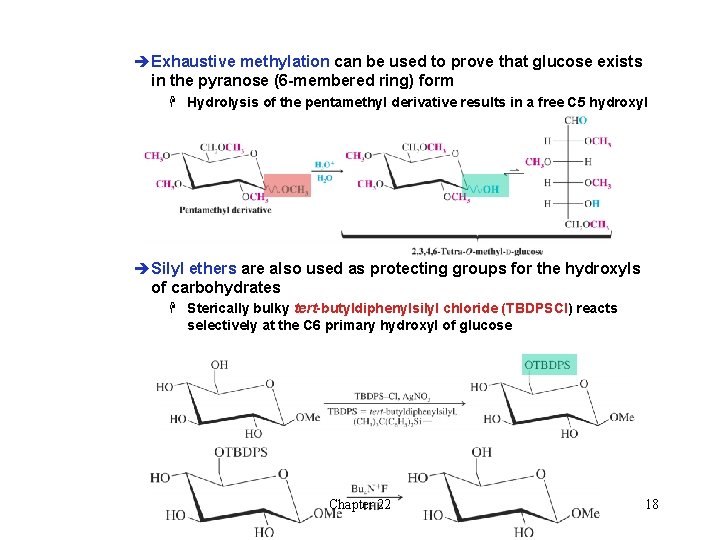
èExhaustive methylation can be used to prove that glucose exists in the pyranose (6 -membered ring) form H Hydrolysis of the pentamethyl derivative results in a free C 5 hydroxyl èSilyl ethers are also used as protecting groups for the hydroxyls of carbohydrates H Sterically bulky tert-butyldiphenylsilyl chloride (TBDPSCl) reacts selectively at the C 6 primary hydroxyl of glucose Chapter 22 18

l Conversion to Esters èCarbohydrates react with acetic anhydride in the presence of weak base to convert all hydroxyl groups to acetate esters l Conversion to Cyclic Acetals èCarbohydrates form cyclic acetals with acetone selectively between cis-vicinal hydroxyl groups H Cyclic acetals from reaction with acetone are called acetonides or isopropylidenates Chapter 22 19

t Oxidation Reactions of Monosaccharides l Benedict’s or Tollens’ Reagents: Reducing Sugars èAldoses and ketoses give positive tests when treated with Tollens’ solution or Benedict’s reagent H Tollens’ reagent [Ag(NH 3)2 OH] gives a silver mirror when Ag+ is reduced to Ag 0 H Benedict’s reagent (an alkaline solution of cupric citrate complex) gives a brick red precipitate of Cu 2 O H In basic solution a ketose can be converted to an aldose that can then react with Tollens’ or Benedicts’s reagent Chapter 22 20
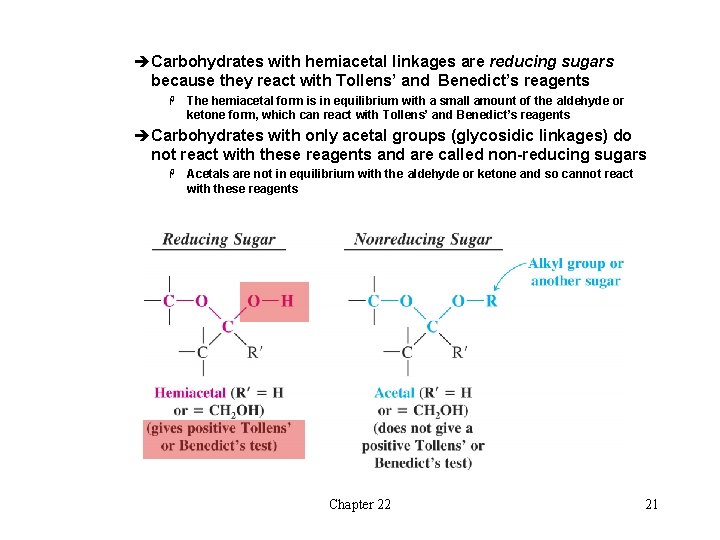
èCarbohydrates with hemiacetal linkages are reducing sugars because they react with Tollens’ and Benedict’s reagents H The hemiacetal form is in equilibrium with a small amount of the aldehyde or ketone form, which can react with Tollens’ and Benedict’s reagents èCarbohydrates with only acetal groups (glycosidic linkages) do not react with these reagents and are called non-reducing sugars H Acetals are not in equilibrium with the aldehyde or ketone and so cannot react with these reagents Chapter 22 21
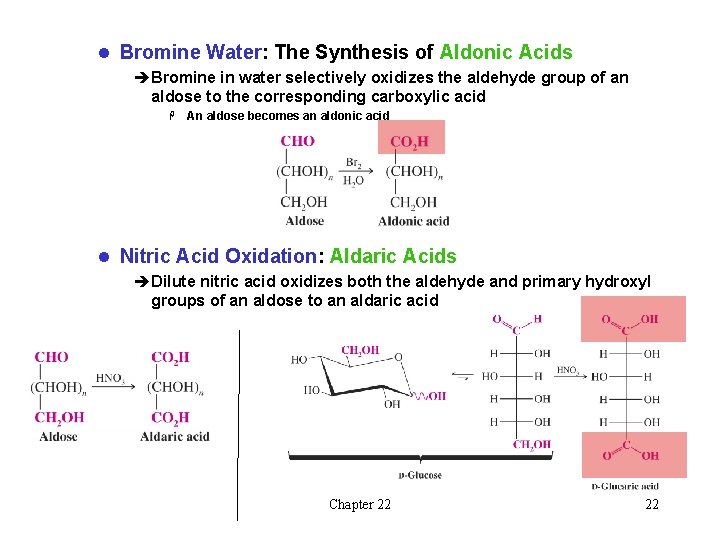
l Bromine Water: The Synthesis of Aldonic Acids èBromine in water selectively oxidizes the aldehyde group of an aldose to the corresponding carboxylic acid H An aldose becomes an aldonic acid l Nitric Acid Oxidation: Aldaric Acids èDilute nitric acid oxidizes both the aldehyde and primary hydroxyl groups of an aldose to an aldaric acid Chapter 22 22
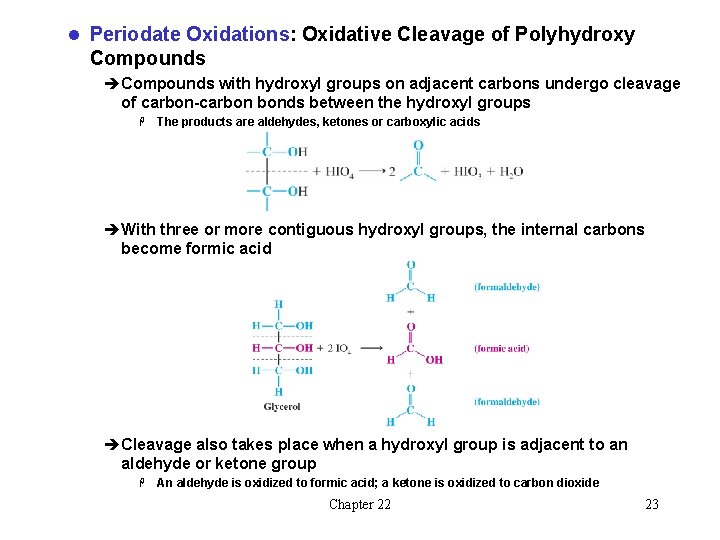
l Periodate Oxidations: Oxidative Cleavage of Polyhydroxy Compounds èCompounds with hydroxyl groups on adjacent carbons undergo cleavage of carbon-carbon bonds between the hydroxyl groups H The products are aldehydes, ketones or carboxylic acids èWith three or more contiguous hydroxyl groups, the internal carbons become formic acid èCleavage also takes place when a hydroxyl group is adjacent to an aldehyde or ketone group H An aldehyde is oxidized to formic acid; a ketone is oxidized to carbon dioxide Chapter 22 23
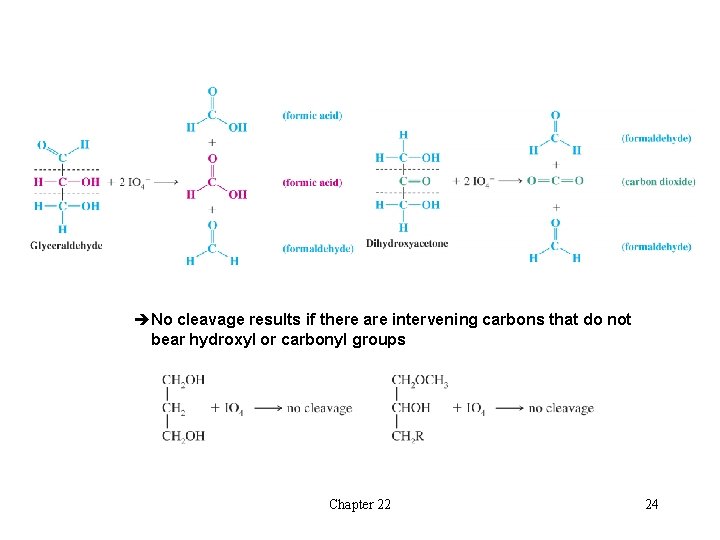
èNo cleavage results if there are intervening carbons that do not bear hydroxyl or carbonyl groups Chapter 22 24
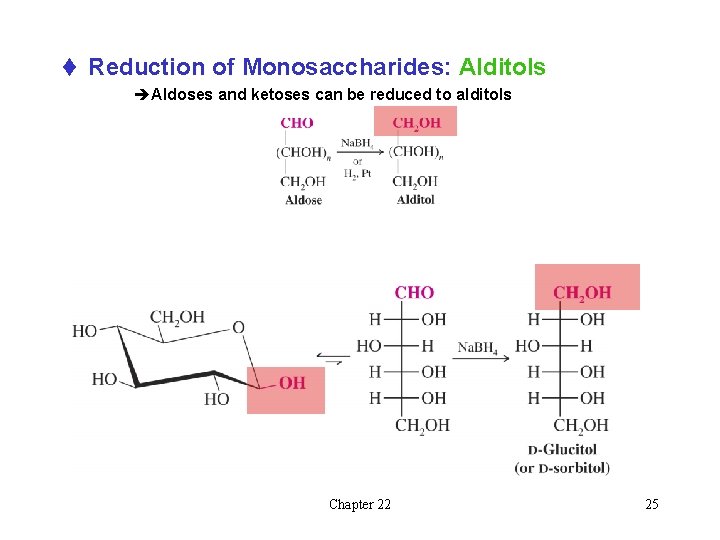
t Reduction of Monosaccharides: Alditols èAldoses and ketoses can be reduced to alditols Chapter 22 25
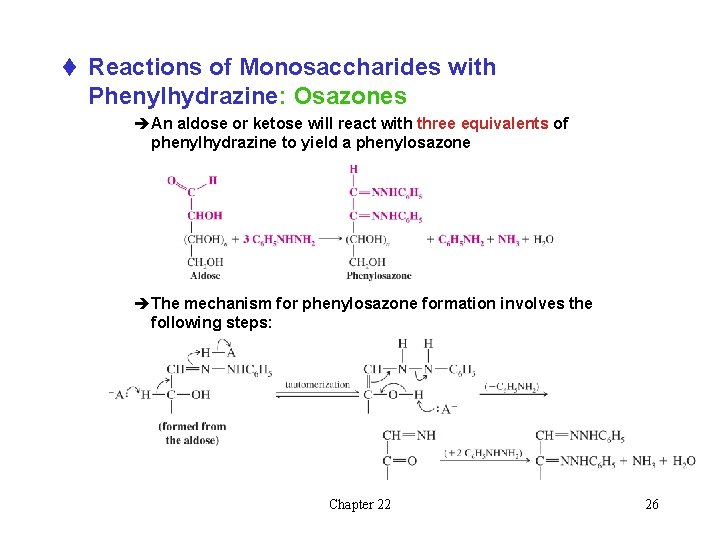
t Reactions of Monosaccharides with Phenylhydrazine: Osazones èAn aldose or ketose will react with three equivalents of phenylhydrazine to yield a phenylosazone èThe mechanism for phenylosazone formation involves the following steps: Chapter 22 26
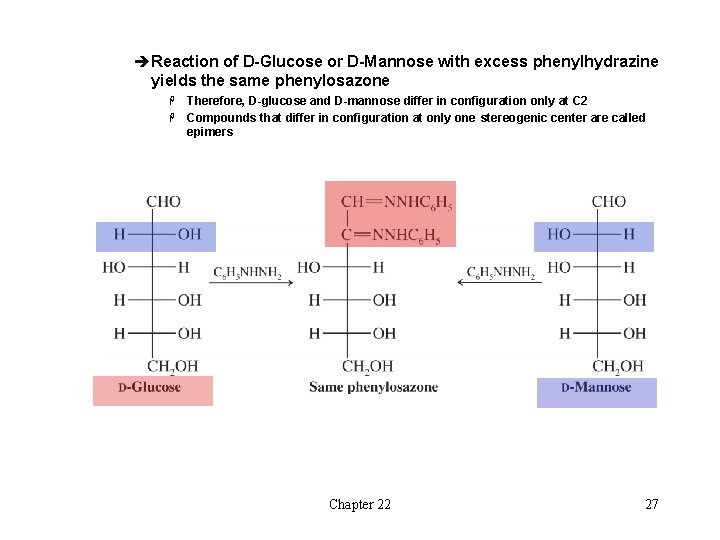
èReaction of D-Glucose or D-Mannose with excess phenylhydrazine yields the same phenylosazone Therefore, D-glucose and D-mannose differ in configuration only at C 2 H Compounds that differ in configuration at only one stereogenic center are called epimers H Chapter 22 27
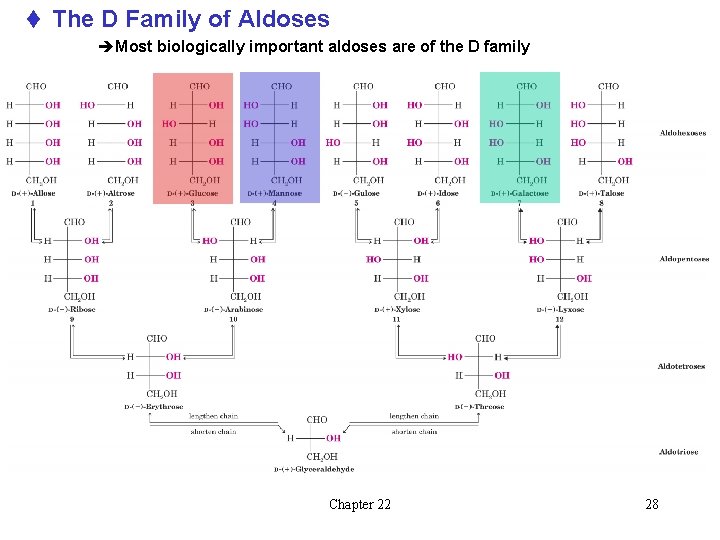
t The D Family of Aldoses èMost biologically important aldoses are of the D family Chapter 22 28
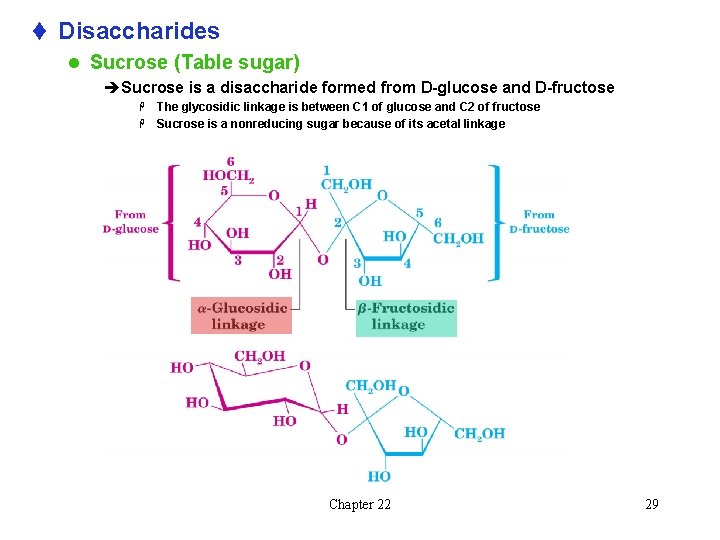
t Disaccharides l Sucrose (Table sugar) èSucrose is a disaccharide formed from D-glucose and D-fructose The glycosidic linkage is between C 1 of glucose and C 2 of fructose H Sucrose is a nonreducing sugar because of its acetal linkage H Chapter 22 29
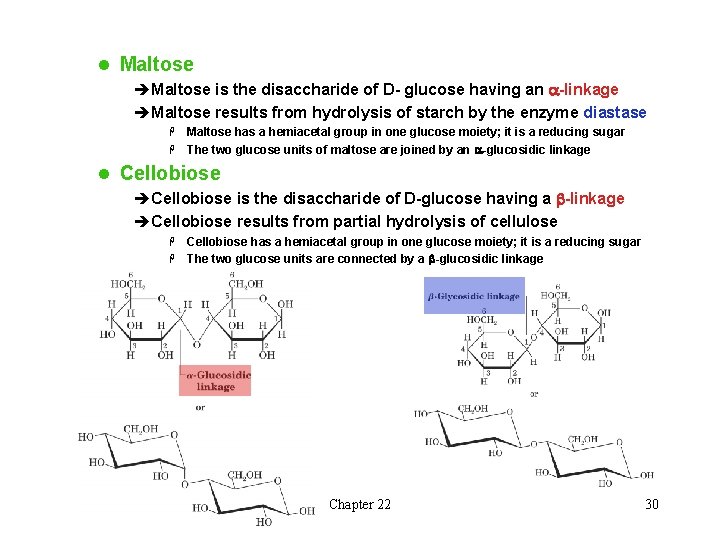
l Maltose èMaltose is the disaccharide of D- glucose having an a-linkage èMaltose results from hydrolysis of starch by the enzyme diastase Maltose has a hemiacetal group in one glucose moiety; it is a reducing sugar H The two glucose units of maltose are joined by an a-glucosidic linkage H l Cellobiose èCellobiose is the disaccharide of D-glucose having a b-linkage èCellobiose results from partial hydrolysis of cellulose Cellobiose has a hemiacetal group in one glucose moiety; it is a reducing sugar H The two glucose units are connected by a b-glucosidic linkage H Chapter 22 30
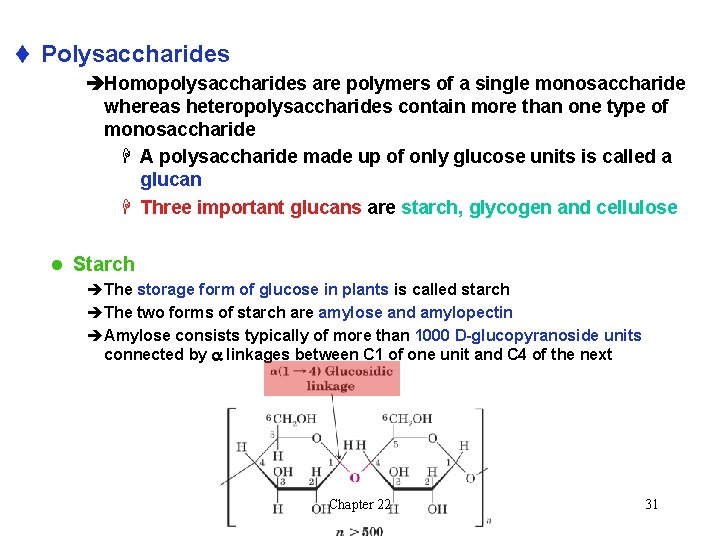
t Polysaccharides èHomopolysaccharides are polymers of a single monosaccharide whereas heteropolysaccharides contain more than one type of monosaccharide H A polysaccharide made up of only glucose units is called a glucan H Three important glucans are starch, glycogen and cellulose l Starch èThe storage form of glucose in plants is called starch èThe two forms of starch are amylose and amylopectin èAmylose consists typically of more than 1000 D-glucopyranoside units connected by a linkages between C 1 of one unit and C 4 of the next Chapter 22 31
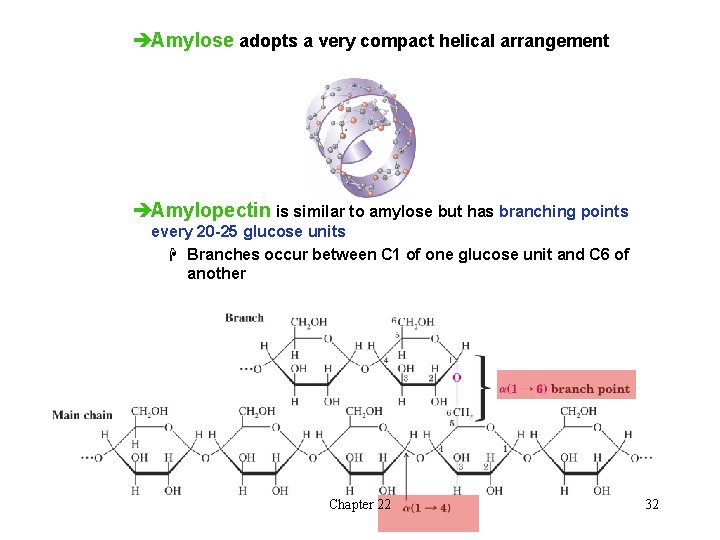
èAmylose adopts a very compact helical arrangement èAmylopectin is similar to amylose but has branching points every 20 -25 glucose units H Branches occur between C 1 of one glucose unit and C 6 of another Chapter 22 32

l Glycogen èGlycogen is the major carbohydrate storage molecule in animals èGlycogen is similar to amylopectin except that glycogen has far more branching H Branching occurs ever 10 -12 glucose units in glycogen èGlycogen is a very large polysaccharide H The large size of glycogen prevents if from leaving the storage cell H The storage of tens of thousands of glucose molecules into one molecule greatly relieves the osmotic problem for the storage cell (this would be caused by the attempted storage of many individual glucose molecules) H The highly branched nature of glycogen allows hydrolytic enzymes to have many chain ends from which glucose molecules can be hydrolyzed èGlucose is the source of “ready energy” for the body H Long chain fatty acids of triacylglycerols are used for long term energy storage Chapter 22 33
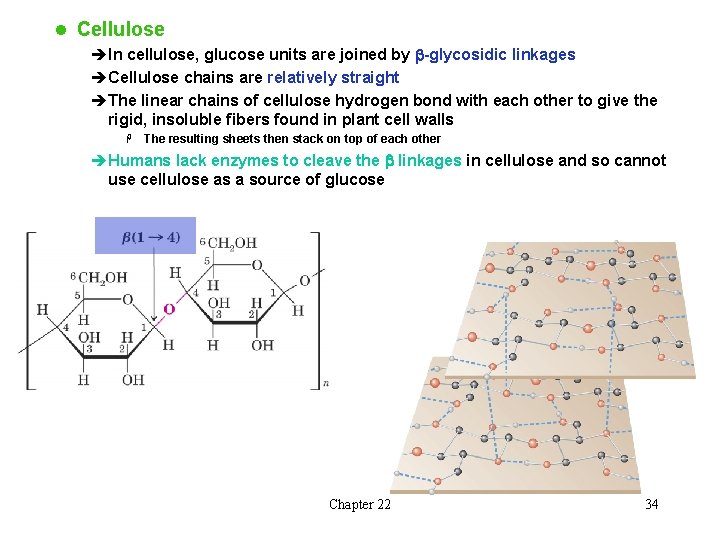
l Cellulose èIn cellulose, glucose units are joined by b-glycosidic linkages èCellulose chains are relatively straight èThe linear chains of cellulose hydrogen bond with each other to give the rigid, insoluble fibers found in plant cell walls H The resulting sheets then stack on top of each other èHumans lack enzymes to cleave the b linkages in cellulose and so cannot use cellulose as a source of glucose Chapter 22 34

l Glycolipids and Glycoproteins of the Cell Surface èGlycolipids and Glycoproteins are important for cell signaling and recognition èA, B, and O human blood groups are determined by glycoprotein antigens designated A, B, and H A antigen: Gal. NAc 1, 3 Gal L-Fuc 1, 2 A Blood type B Blood type O(H) Blood type Chapter 22 35
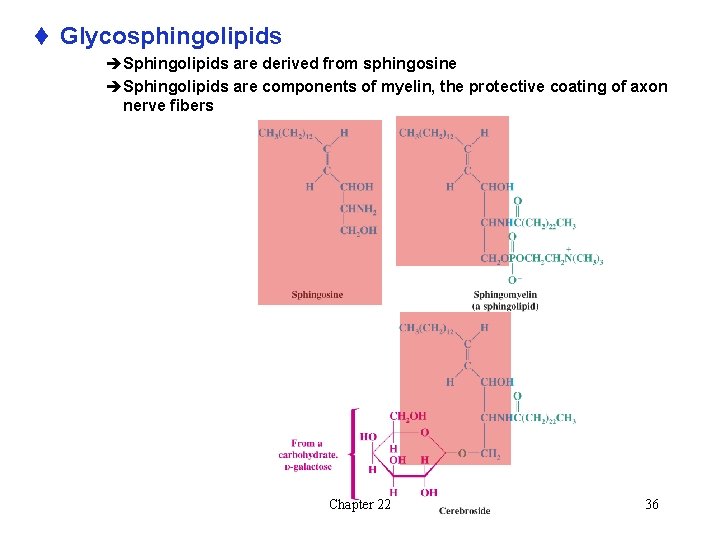
t Glycosphingolipids èSphingolipids are derived from sphingosine èSphingolipids are components of myelin, the protective coating of axon nerve fibers Chapter 22 36
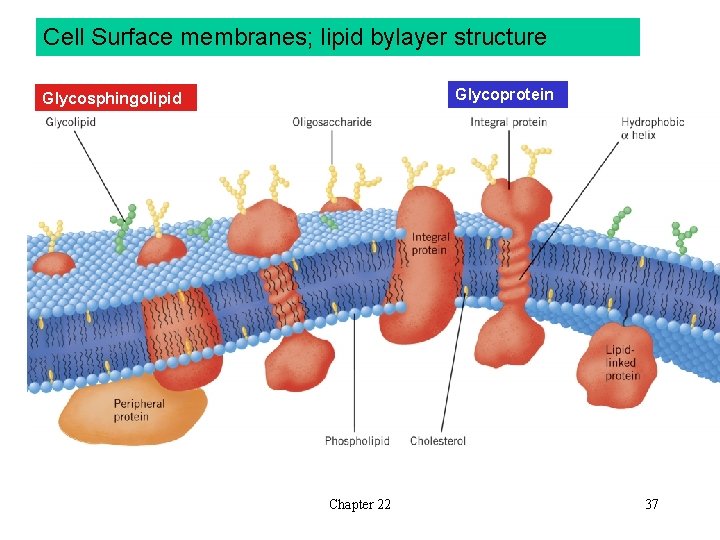
Cell Surface membranes; lipid bylayer structure Glycoprotein Glycosphingolipid Chapter 22 37

Cell Surface membranes Chapter 22 38
- Slides: 38