Organic Chemistry Hydrocarbon Naming Formula Writing Organic Chemistry
Organic Chemistry Hydrocarbon Naming & Formula Writing

Organic Chemistry • Study of carbon and most carbon containing compounds where carbon is bonded covalently • Number of carbon (organic) compounds far exceeds the number of inorganic compounds. . . Why? – Carbon has the ability to bond with other carbon atoms to form chains, rings, and networks.

Bonding in Carbon Atoms • Can form a bond with 4 other atoms – Organic compounds containing only single bonds are saturated – Organic compounds containing one or more double or triple covalent bonds are unsaturated
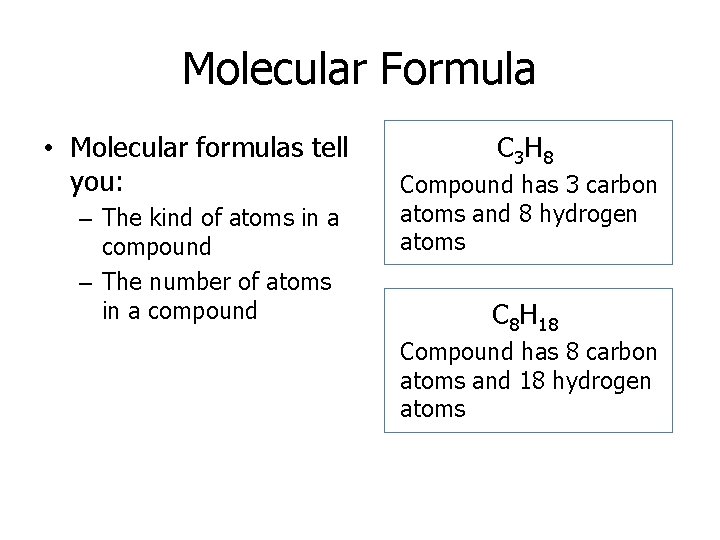
Molecular Formula • Molecular formulas tell you: – The kind of atoms in a compound – The number of atoms in a compound C 3 H 8 Compound has 3 carbon atoms and 8 hydrogen atoms C 8 H 18 Compound has 8 carbon atoms and 18 hydrogen atoms

Structural Formula • Structural formulas tell you: – the kind and number of atoms in a compound – the bonding patterns and approximate shapes of the molecules
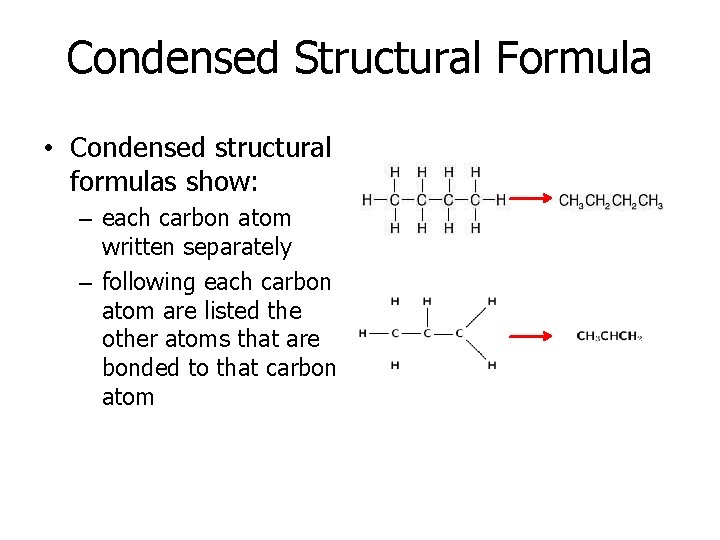
Condensed Structural Formula • Condensed structural formulas show: – each carbon atom written separately – following each carbon atom are listed the other atoms that are bonded to that carbon atom
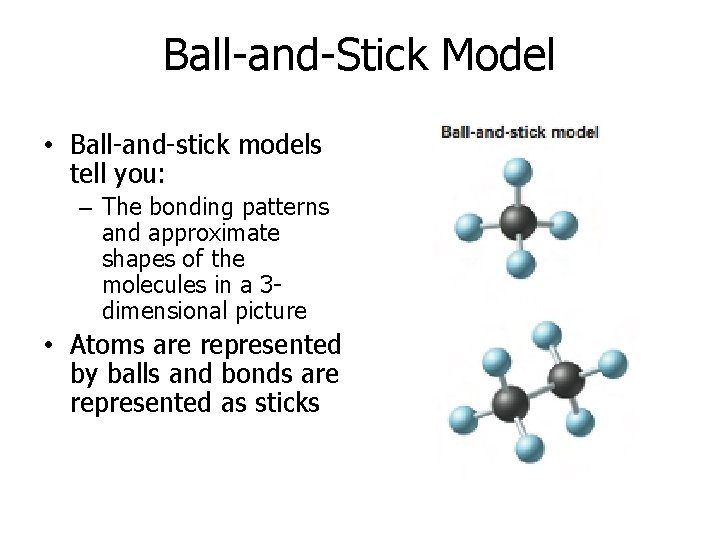
Ball-and-Stick Model • Ball-and-stick models tell you: – The bonding patterns and approximate shapes of the molecules in a 3 dimensional picture • Atoms are represented by balls and bonds are represented as sticks

Hydrocarbons • Organic compounds that contain only atoms of hydrogen and carbon • 3 organic compound series – Alkanes – Alkenes – Alkynes

Properties of Hydrocarbons 1. 2. 3. 4. 5. 6. Low melting points and low boiling points Poor conductors of heat and electricity Nonpolar – dissolve in nonpolar solvents React more slowly than ionic compounds Held together by Van der Waals forces Named according to IUPAC system
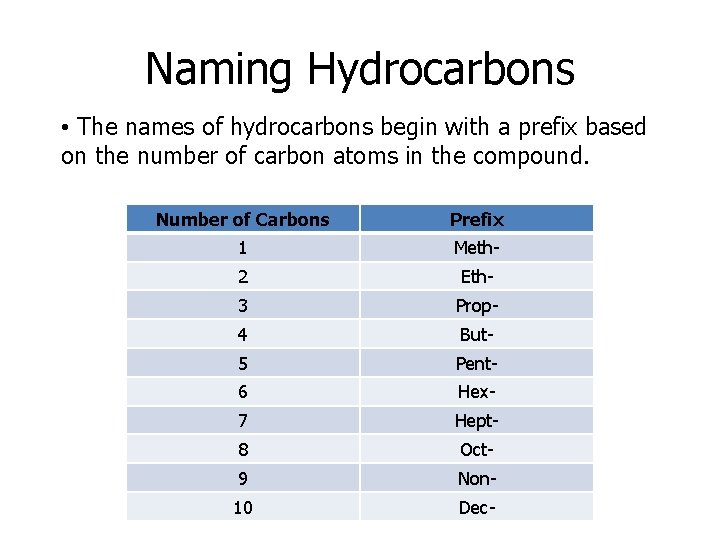
Naming Hydrocarbons • The names of hydrocarbons begin with a prefix based on the number of carbon atoms in the compound. Number of Carbons Prefix 1 Meth- 2 Eth- 3 Prop- 4 But- 5 Pent- 6 Hex- 7 Hept- 8 Oct- 9 Non- 10 Dec-

Alkanes • Hydrocarbons that contain only single bonds – saturated • • Release energy when burned Methane (CH 4), comprises about 90% of natural gas – used to heat homes
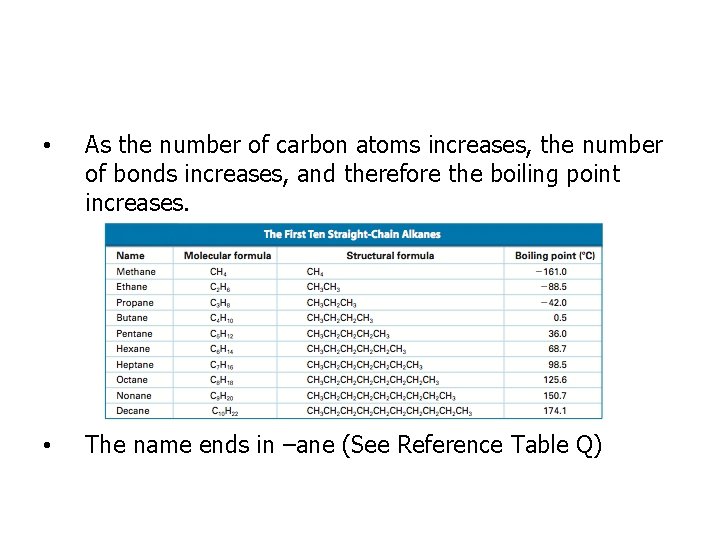
• As the number of carbon atoms increases, the number of bonds increases, and therefore the boiling point increases. • The name ends in –ane (See Reference Table Q)

Alkanes Cn. H(2 n+2) Number of Carbons Prefix Formula 1 Methane CH 4 2 3 4 5 Ethane Propane Butane Pentane C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12

Alkenes • Contains one double covalent bond – unsaturated • The first member of the alkene series is ethene, or ethylene • Name ends in –ene (See Reference Table Q) • Alkadienes have 2 double bonds and have the ending -diene

Alkenes Cn. H 2 n Number of Carbons 2 3 4 5 Prefix Ethene Propene Butene Pentene Formula C 2 H 4 C 3 H 6 C 4 H 8 C 5 H 10

Alkynes • Unsaturated hydrocarbons that contain one triple bond • Name ends in –yne (See Reference Table Q) Ethyne Propyne 5, 5 dimethyl 1 -Hexyne

Alkynes Cn. H(2 n-2) Number of Carbons Prefix Formula 2 Ethyne C 2 H 2 3 4 5 Propyne Butyne Pentyne C 3 H 4 C 4 H 6 C 5 H 8

Organic Chemistry Isomers & Naming Organic Compounds
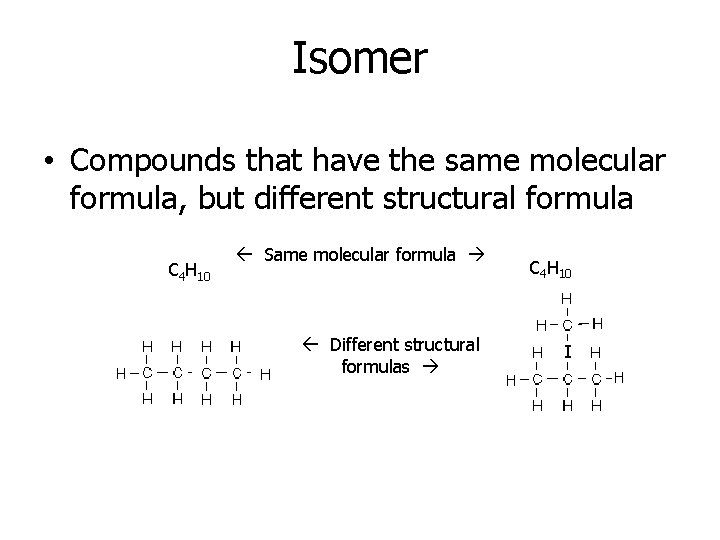
Isomer • Compounds that have the same molecular formula, but different structural formula C 4 H 10 Same molecular formula Different structural formulas C 4 H 10 I

• Different chemical and physical properties • As the number of carbon atoms increases, so does the number of possible isomers • It is the ability to form isomers that is largely responsible for the large number of organic compounds Worksheet - Isomers
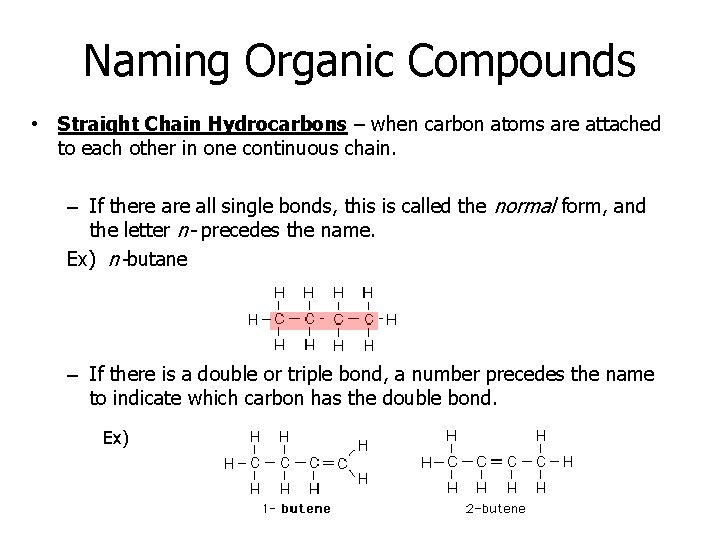
Naming Organic Compounds • Straight Chain Hydrocarbons – when carbon atoms are attached to each other in one continuous chain. – If there all single bonds, this is called the normal form, and the letter n- precedes the name. Ex) n-butane – If there is a double or triple bond, a number precedes the name to indicate which carbon has the double bond. Ex)
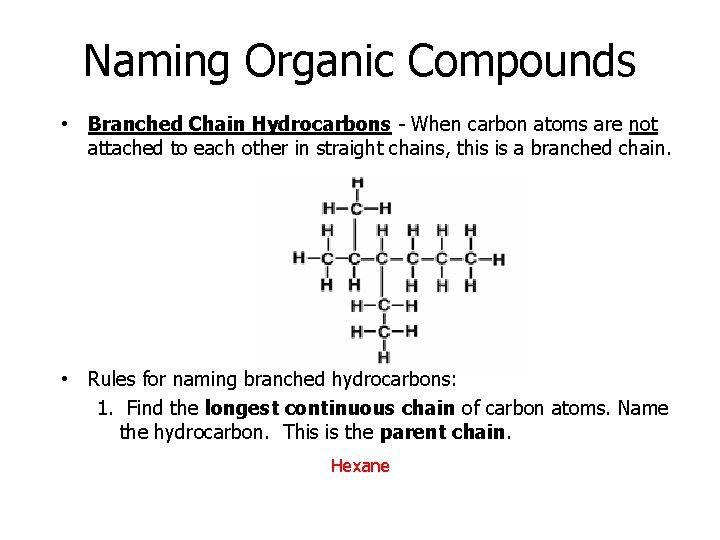
Naming Organic Compounds • Branched Chain Hydrocarbons - When carbon atoms are not attached to each other in straight chains, this is a branched chain. • Rules for naming branched hydrocarbons: 1. Find the longest continuous chain of carbon atoms. Name the hydrocarbon. This is the parent chain. Hexane
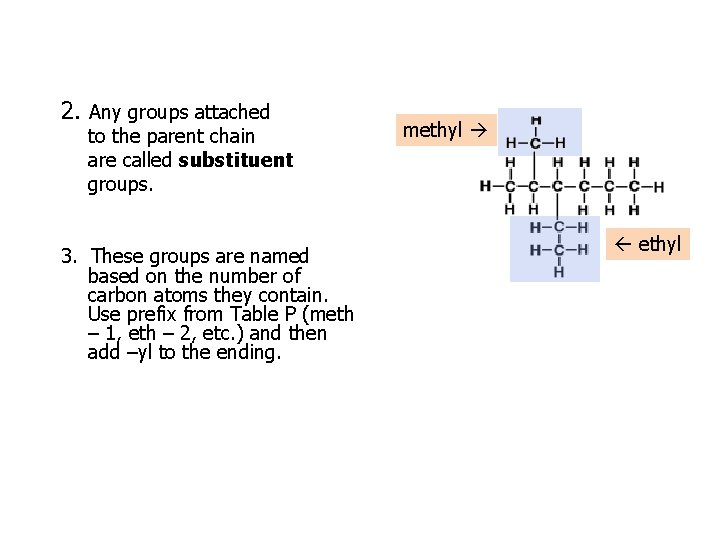
2. Any groups attached to the parent chain are called substituent groups. 3. These groups are named based on the number of carbon atoms they contain. Use prefix from Table P (meth – 1, eth – 2, etc. ) and then add –yl to the ending. methyl
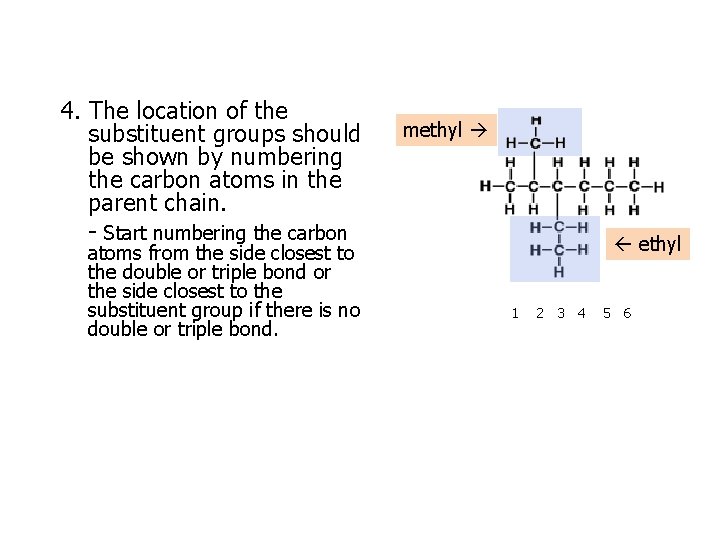
4. The location of the substituent groups should be shown by numbering the carbon atoms in the parent chain. - Start numbering the carbon atoms from the side closest to the double or triple bond or the side closest to the substituent group if there is no double or triple bond. methyl 1 2 3 4 5 6

5. If there is more than one of the same substituent group, a prefix is used. (di- 2, tri- 3, tetra- 4, etc. ) l For example, if there were 2 methyl groups, one on carbon-2, and one on carbon-3, you would combine the groups in the name 2, 3 -dimethyl. 6. We need to combine the substituent groups with the parent group in one long name. l Name the parent group l The substituent groups are listed before the parent group in alphabetical order. (when determining alphabetical order, disregard the prefixes di, tri, tetra, etc. ) 3 -ethyl-2 -methylhexane

Carbon Rings • Alkanes, alkenes and alkynes all contain carbon atoms in linear chains. • There also hydrocarbons arranged in rings.
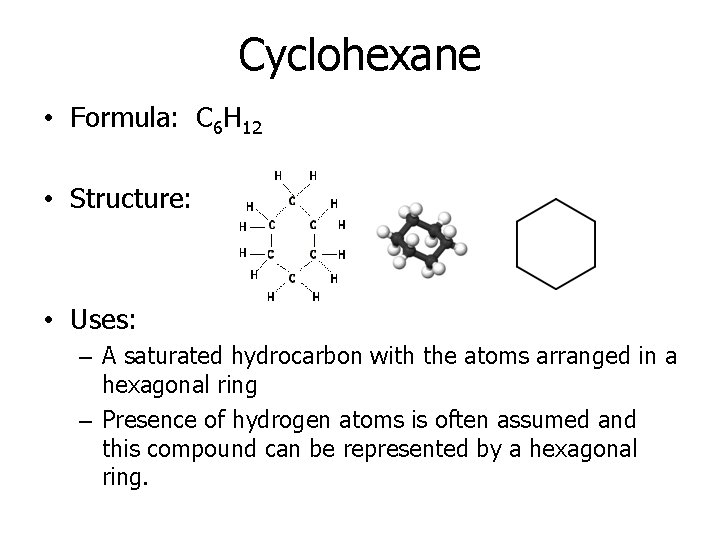
Cyclohexane • Formula: C 6 H 12 • Structure: • Uses: – A saturated hydrocarbon with the atoms arranged in a hexagonal ring – Presence of hydrogen atoms is often assumed and this compound can be represented by a hexagonal ring.
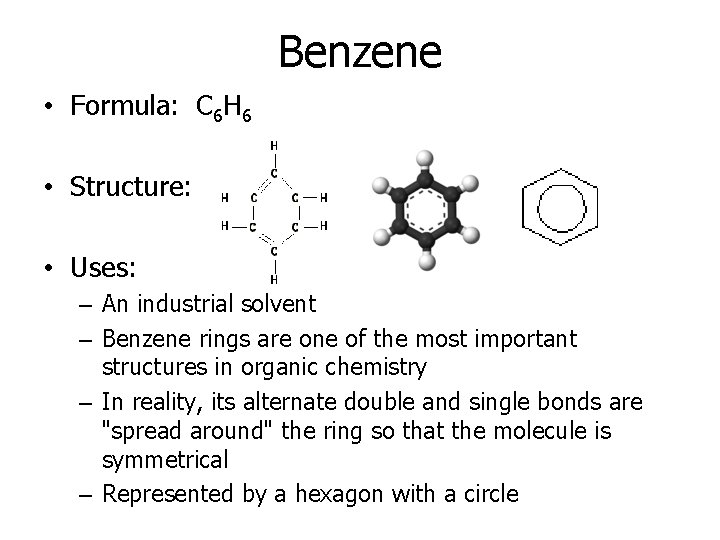
Benzene • Formula: C 6 H 6 • Structure: • Uses: – An industrial solvent – Benzene rings are one of the most important structures in organic chemistry – In reality, its alternate double and single bonds are "spread around" the ring so that the molecule is symmetrical – Represented by a hexagon with a circle
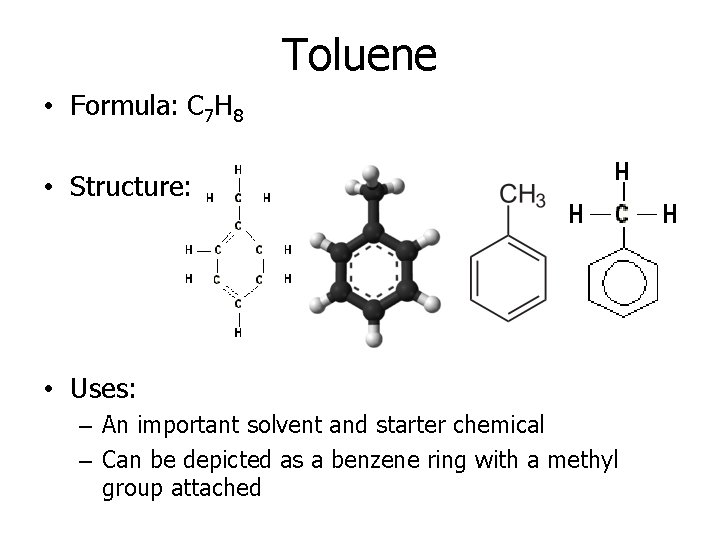
Toluene • Formula: C 7 H 8 • Structure: • Uses: – An important solvent and starter chemical – Can be depicted as a benzene ring with a methyl group attached
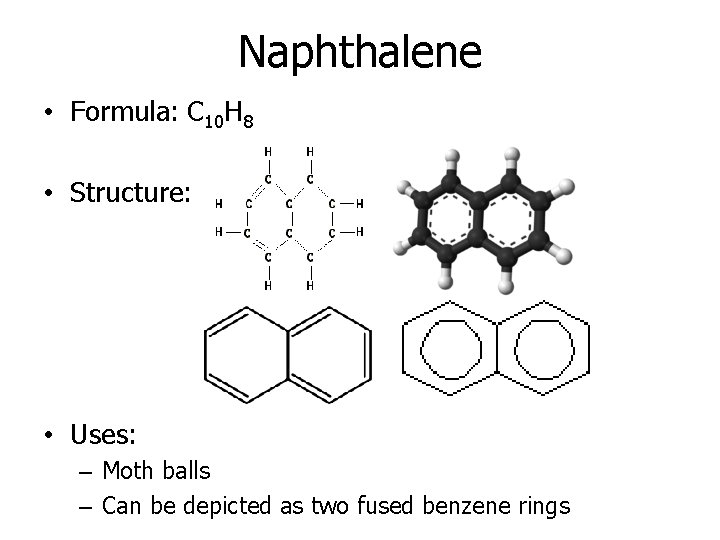
Naphthalene • Formula: C 10 H 8 • Structure: • Uses: – Moth balls – Can be depicted as two fused benzene rings
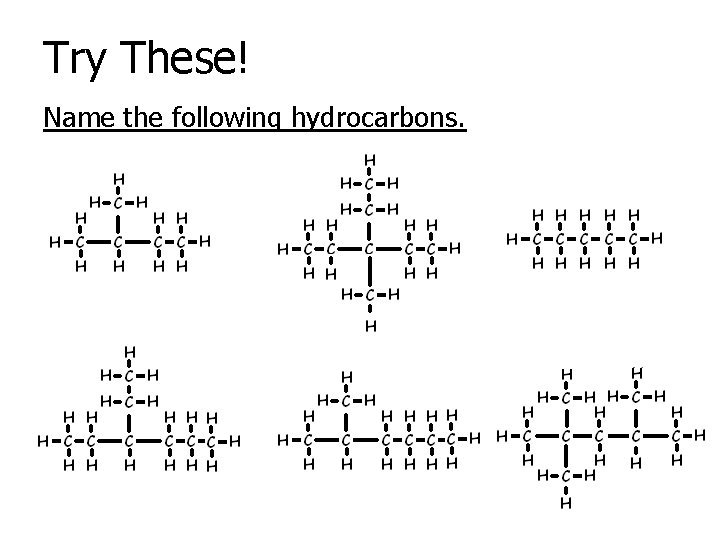
Try These! Name the following hydrocarbons.

Try These! Write the structural formula for the following hydrocarbons a)2, 3 -dimethyl-2 -pentene b)2 -methyl-4 -propyl-3 -heptene c) 4 -ethyl-2 -octyne d)2, 3, 4 -trimethylhexane

Organic Chemistry Organic Compounds with Functional Groups

Functional Groups • Functional groups – Groups of atoms that give an organic series its distinctive character – When oxygen or nitrogen atoms are added, the variety of organic compounds grows enormously – (See Table R on Chemistry Reference Tables)
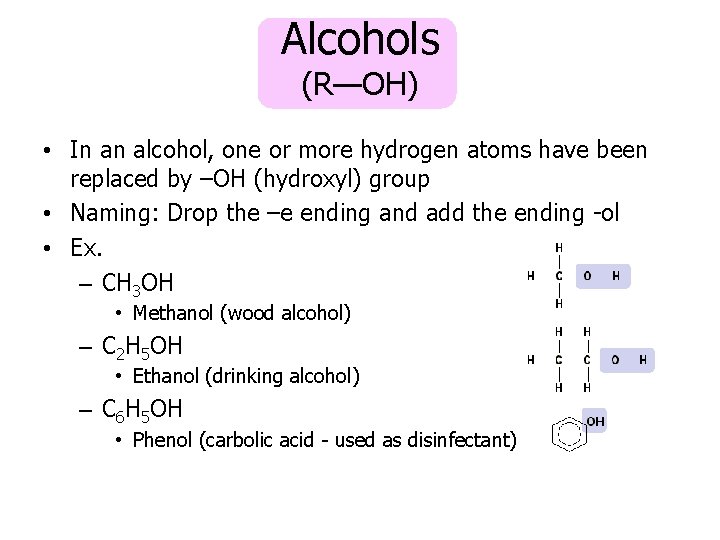
Alcohols (R—OH) • In an alcohol, one or more hydrogen atoms have been replaced by –OH (hydroxyl) group • Naming: Drop the –e ending and add the ending -ol • Ex. – CH 3 OH • Methanol (wood alcohol) – C 2 H 5 OH • Ethanol (drinking alcohol) – C 6 H 5 OH • Phenol (carbolic acid - used as disinfectant)

Classifying Alcohols • May be classified according to the number of –OH groups in the molecule – 1 –OH group per molecule (ending is –ol) – 2 –OH groups per molecule (ending is –diol) – 3 –OH groups per molecule (ending is –triol) • Primary alcohols have the –OH group bonded to a primary carbon atom (C bonded to 1 other C) • Secondary alcohols have the –OH group bonded to a secondary carbon atom (C bonded to 2 other C) • Tertiary alcohols have the –OH group bonded to a tertiary carbon atom (C bonded to 3 other C)

Ethers (R—O—R’) • Ethers have an O atom attached to two hydrocarbon chains (or rings) • Naming: Groups attached to ether are named in alphabetical order and are followed by the word ether • Ex. – (CH 3)2 O • Dimethyl Ether (a gas) – (C 2 H 5)2 O • Diethyl Ether (a liquid used as an anaesthetic)
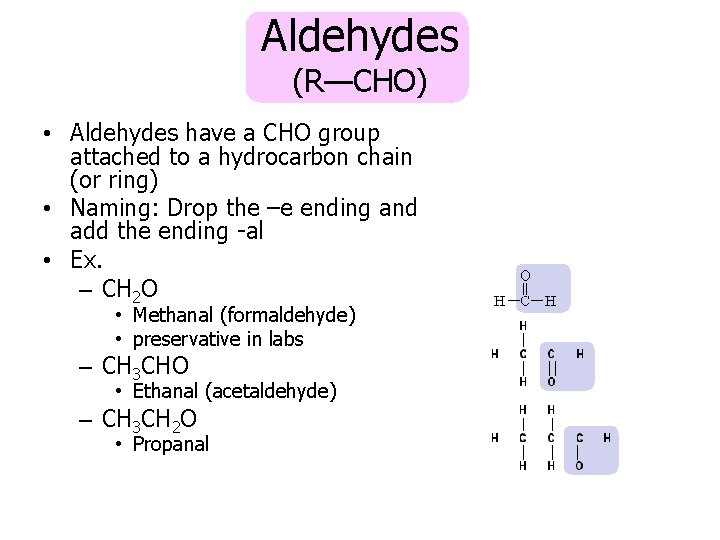
Aldehydes (R—CHO) • Aldehydes have a CHO group attached to a hydrocarbon chain (or ring) • Naming: Drop the –e ending and add the ending -al • Ex. – CH 2 O • Methanal (formaldehyde) • preservative in labs – CH 3 CHO • Ethanal (acetaldehyde) – CH 3 CH 2 O • Propanal
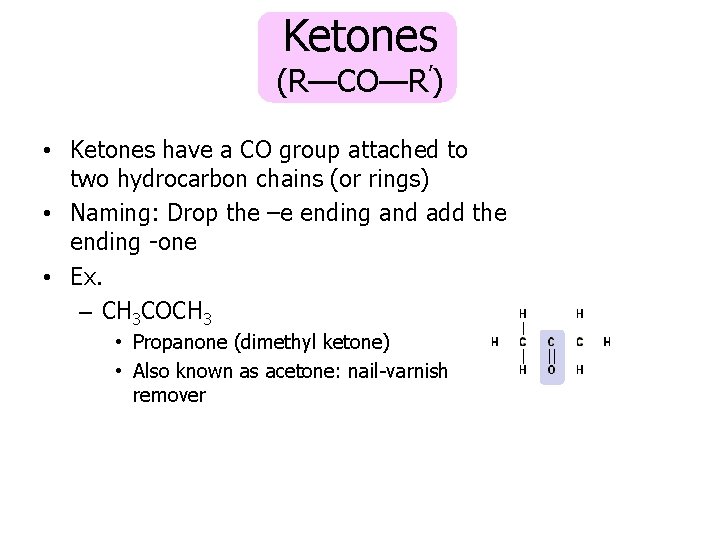
Ketones (R—CO—R’) • Ketones have a CO group attached to two hydrocarbon chains (or rings) • Naming: Drop the –e ending and add the ending -one • Ex. – CH 3 COCH 3 • Propanone (dimethyl ketone) • Also known as acetone: nail-varnish remover
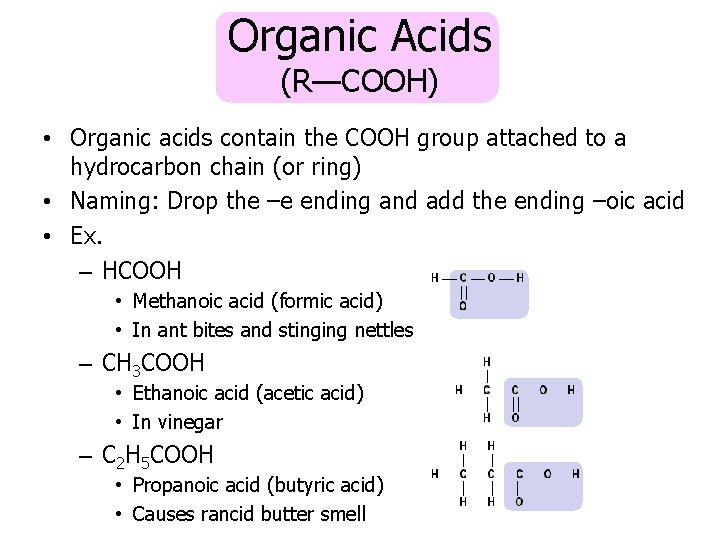
Organic Acids (R—COOH) • Organic acids contain the COOH group attached to a hydrocarbon chain (or ring) • Naming: Drop the –e ending and add the ending –oic acid • Ex. – HCOOH • Methanoic acid (formic acid) • In ant bites and stinging nettles – CH 3 COOH • Ethanoic acid (acetic acid) • In vinegar – C 2 H 5 COOH • Propanoic acid (butyric acid) • Causes rancid butter smell

Esters (R—COO-R’) • Esters contain the COO group attached to 2 hydrocarbon chains (or rings) • Formed from an organic acid an alcohol • They are usually very sweet smelling liquids used in perfumes or soaps • Naming: Change the –ol ending of the alcohol to –yl and change the –oic acid ending of the organic acid to -oate • Ex. – CH 3 COOCH 3 • Methyl ethanoate • Essence of pear drops
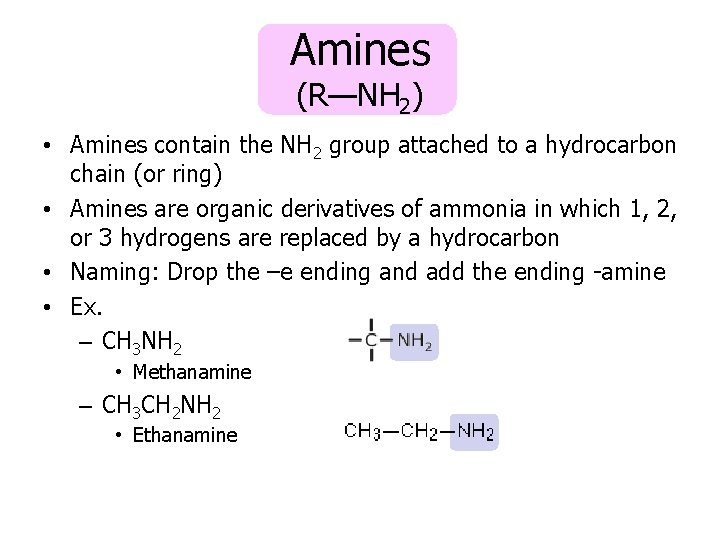
Amines (R—NH 2) • Amines contain the NH 2 group attached to a hydrocarbon chain (or ring) • Amines are organic derivatives of ammonia in which 1, 2, or 3 hydrogens are replaced by a hydrocarbon • Naming: Drop the –e ending and add the ending -amine • Ex. – CH 3 NH 2 • Methanamine – CH 3 CH 2 NH 2 • Ethanamine
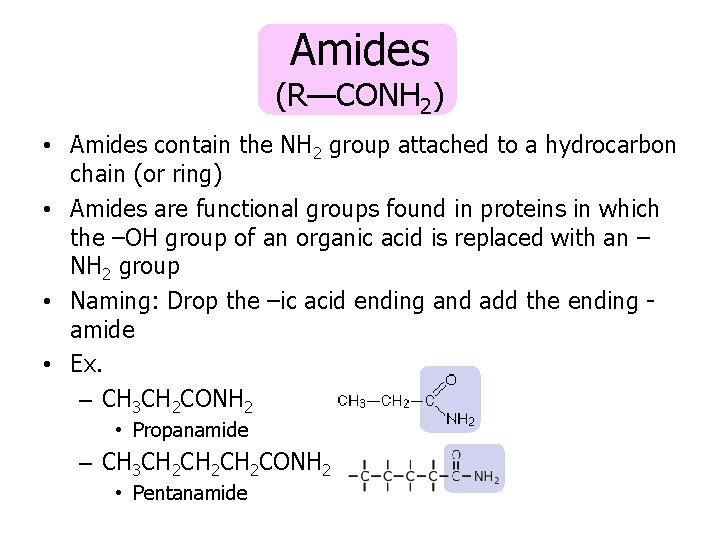
Amides (R—CONH 2) • Amides contain the NH 2 group attached to a hydrocarbon chain (or ring) • Amides are functional groups found in proteins in which the –OH group of an organic acid is replaced with an – NH 2 group • Naming: Drop the –ic acid ending and add the ending amide • Ex. – CH 3 CH 2 CONH 2 • Propanamide – CH 3 CH 2 CH 2 CONH 2 • Pentanamide

Combinations of Functional Groups • In the previous examples, each molecule has a single functional group • It is possible to have two or more functional groups on a molecule • These can be the same group or different • Ex. Same functional groups – (COOH)2 • Oxalic acid with two organic acid groups • Poison found in rhubarb leaves • Ex. Different functional groups – CH 2 OHCOOH • Hydroxymethanoic acid with a hydroxyl group and an organic acid group
- Slides: 44