Matter What is it Matter Anything that occupies






![Consider this……. . If an atom is a marker [if atom=marker] Then an element Consider this……. . If an atom is a marker [if atom=marker] Then an element](https://slidetodoc.com/presentation_image_h2/46cbb1cf654e2d7066b6448a492cc5d4/image-8.jpg)











![If I had a block of pure gold (woohoo!) [an element] OR a spoonful If I had a block of pure gold (woohoo!) [an element] OR a spoonful](https://slidetodoc.com/presentation_image_h2/46cbb1cf654e2d7066b6448a492cc5d4/image-20.jpg)
































- Slides: 56

Matter…. . What is it? Matter: Anything that occupies space and has mass

What is Matter? • It is: – Anything you can hold or touch – It has mass – It has volume – Us! – Our Earth • It is not: – Light – Sound – Electricity

Why does matter MATTER? Because we’re going to learn about chemistry…. And Chemistry = The study of matter and changes in matter This could be you!!

So what is matter made up of…. . ? At it’s most basic level……… Atoms! Atom: smallest unit of matter -made up of protons, electrons, neutrons -everything (classified as matter) is made up of atoms!

The specific number of p+, e-, & n’s give an atom its identity……. .

Identify an atom as what? (you may ask) An element! Element: a pure substance that cannot be broken down into simpler substances Iron

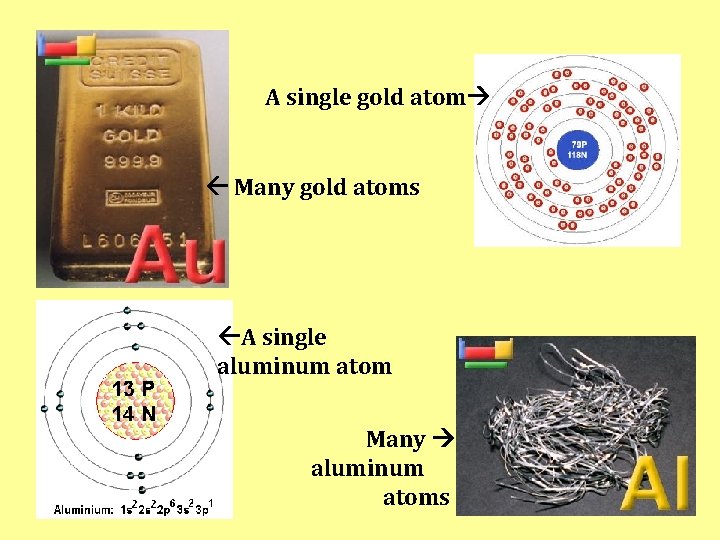
A single gold atom Many gold atoms A single aluminum atom Many aluminum atoms
![Consider this If an atom is a marker if atommarker Then an element Consider this……. . If an atom is a marker [if atom=marker] Then an element](https://slidetodoc.com/presentation_image_h2/46cbb1cf654e2d7066b6448a492cc5d4/image-8.jpg)
Consider this……. . If an atom is a marker [if atom=marker] Then an element could be a red marker [then element=red marker]

Now you try…… If an atom is a vegetable? Then an element could be a _______ If an atom is a pie? Then an element could be a _______

Science Theater!!!! What if I put a whole bunch of atoms together? . . If they’re the same, they’re an ELEMENT If they’re different, they’re a……………

Compound! Compound: a substance that is composed of atoms of 2 or more elements in definite proportions EX: Nylon is a compound made up of: Carbon Hydrogen (C 12 H 22 N 2 O 2)n Nitrogen Oxygen Every compound It is this specific ratio that makes it is unique and is nylon… if the elements were in a different from different ratio they would make up a the elements it completely different substance! contains

I showed Nylon as: (C 12 H 22 N 2 O 2) This is a representation/symbolic form of a compound…. Any idea what we call it?

Chemical Formula! Chemical Formula: symbolic representation of the components of a compound • made up of element symbols and numbers (subscripts) that display the ratio of atoms • Let’s look at our Nylon example:

INTERMISSION Take a lap around the room

Chemical Formulas • Let’s look at our Nylon example: C 12 H 22 N 2 O 2 Based on your prior Periodic Table knowledge, can you name the elements in this compound? Can you identify how many atoms of each element are in this compound?

C 12 H 22 N 2 O 2 Carbon : 12 Hydrogen : 22 Nitrogen : 2 Oxygen : 2

Element Symbols C 12 H 22 N 2 O 2 # of atoms in compound (of that element) Notice how the numbers come after the letter they’re describing

How many CARBON atoms are in methane? FINGERS UP! CH 4 How many HYDROGEN atoms are in methane? How many different elements are in methane?

How many different elements are in sodium bromide? FINGERS UP! Na. Br Sodium Bromine How many SODIUM (Na) atoms are in sodium bromide? How many BROMINE (Br) atoms are in sodium bromide?
![If I had a block of pure gold woohoo an element OR a spoonful If I had a block of pure gold (woohoo!) [an element] OR a spoonful](https://slidetodoc.com/presentation_image_h2/46cbb1cf654e2d7066b6448a492cc5d4/image-20.jpg)
If I had a block of pure gold (woohoo!) [an element] OR a spoonful of sugar [a compound] I would call these Pure Substances

Whereas, if I were in the ocean (i. e. salt water) [2 different compounds, specifically 2 different pure substances] I would call the ocean a…. MIXTURE

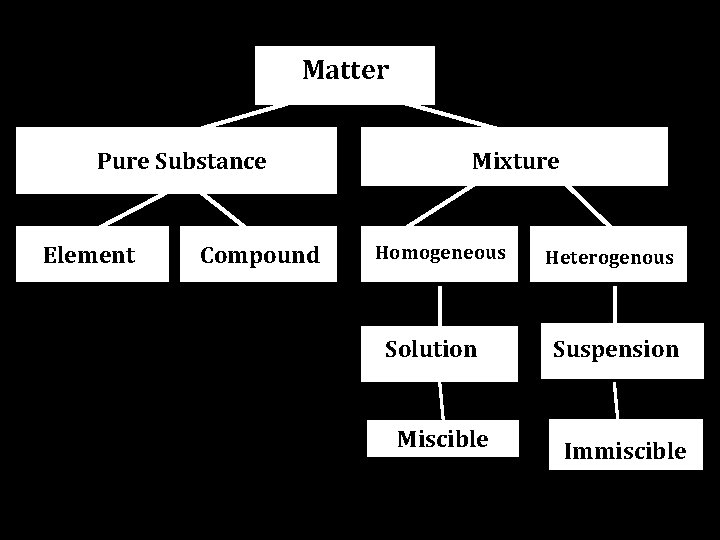
Pure substance: Any matter that has a FIXED composition and definite properties -Pure elements or compounds -EX. Gold, iron, salt, sugar Mixture: Combination of 2 or more substances in which the substances retain their distinct identities -EX. Salt water, balsamic and oil dressing

There are various types of mixtures, specifically 2 important ones…. Any ideas what they are? Homogeneous & Heterogeneous What’s the difference?

Types of Mixtures Homogeneous: Heterogeneous: Composition of mixture is the same throughout Composition is NOT uniform EX. Pennies=Copper+Zinc EX. Chex mix (YUM)

Types of Mixtures Homogeneous: When talking about liquids, it’s described as Heterogeneous: When talking about liquids, it’s described as MISCIBLE IMMISCIBLE (salt water) (balsamic and oil dressing)

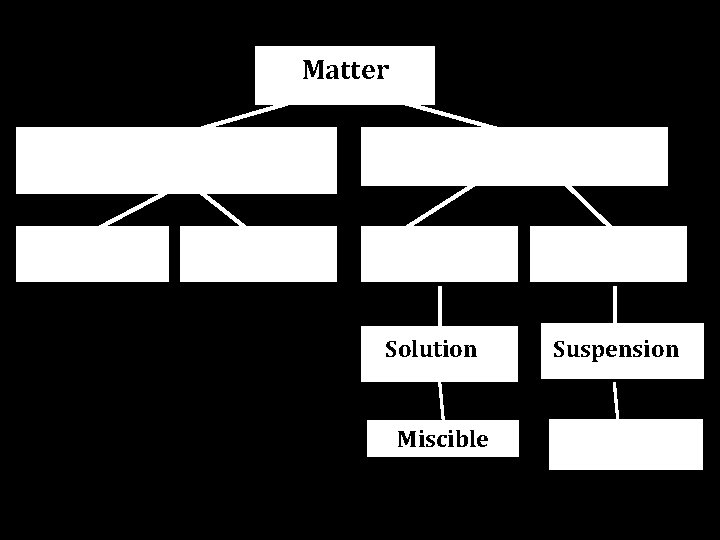
Matter Solution Miscible Suspension

Matter Pure Substance Element Compound Mixture Homogeneous Solution Miscible Heterogenous Suspension Immiscible

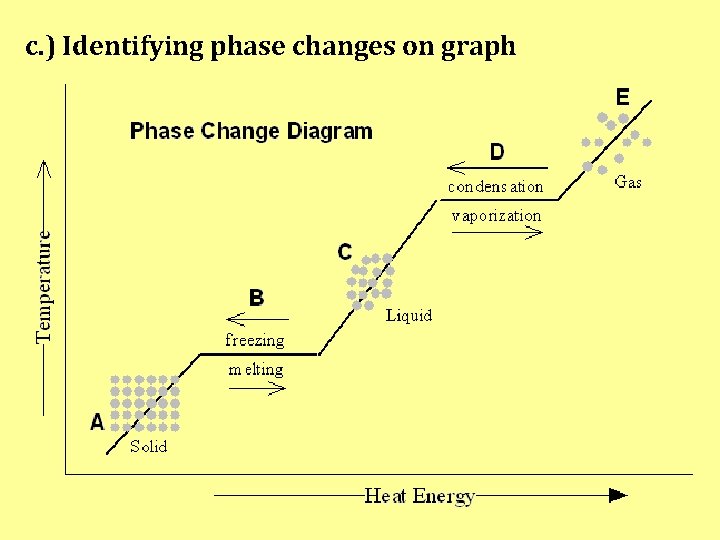
c. ) Identifying phase changes on graph

Over the course of the unit the words “Physical”, “Chemical” and “Properties” have come up a BUNCH! Let’s find out what they mean Take out your Notes

2 main types of PROPERTIES: a. ) Physical Properties: characteristics that can be observed or measured without changing the composition of the substance ex. ) texture, color, boiling pt, density Ex. ) Dirt=dark brown, grainy….

2 main types of PROPERTIES: b. ) Chemical Properties: characteristics that are only measurable and observable during a reaction; changing this type of property would change the identity of the substance ex. ) reactivity, p. H Sodium+water----->

**Physical and Chemical properties are IMPORTANT because they can help us identify different substances based on their properties** The more properties we can identify for a substance, the better we know the nature of that substance. These properties can then help us model the substance and thus understand how this substance will behave under various conditions.

For example, we know (based on many, many experiments) that water - has a boiling point of 100°C - has density of 1 g/m. L - is not flammable If I tested Mystery Liquid A & it…. . -boiled at 100°C -had a density of 1 g/m. L -did not light on fire when a match was What could you INFER based on these OBSERVATIONS?

If I tested Mystery Liquid A & it…. . -boiled at 100°C -had a density of 1 g/m. L -did not light on fire when a match was Then…. You could INFER that Mystery Liquid A is WATER based on these OBSERVATIONS

On the other hand, If I tested Mystery Liquid A & it…. . -boiled at 180°C -had a density of 0. 25 g/m. L -did not light on fire when a match was Could you INFER that Mystery Liquid A is water based on these OBSERVATIONS? NO (we’d have to compare our data to other known information and try to figure out what liquid it is)

2 main types of CHANGES: Physical Change: a change that does not alter the chemical composition of a substance [Δ that ≠ change chem. comp. of subst. ] ex. ) cutting paper, ice melting, dissolving sugar in water

Chemical Change: a process in which substances change into a different substance [Δ creates new subst. ] -chemical composition is altered: reaarangement of atoms bonds broken or new bonds formed [chem. comp. = new] ex. ) iron rusting (Iron turns into Iron oxide) baking bread (ingredients react and create gases that allow dough to rise)

There are 4 main indicators that a chemical change occurred: 1. ) Production and release of a gas 2. ) Formation of a precipitate (solid product) 3. ) Change of color (spontaneously) 4. ) Change in heat and light produced

Color 1. ) Physical Property 2. ) Chemical Property

Odor 1. ) Physical Property 2. ) Chemical Property

Reactivity 1. ) Physical Property 2. ) Chemical Property

Boiling Point 1. ) Physical Property 2. ) Chemical Property

Density 1. ) Physical Property 2. ) Chemical Property

Now let’s try it with physical and chemical CHANGES (∆’s)

Boiling of Water 1. ) Physical Change (∆) 2. ) Chemical Change (∆)

Tearing of Clothes 1. ) Physical ∆ 2. ) Chemical ∆

Rusting of a nail 1. ) Physical ∆ 2. ) Chemical ∆

Melting of Ice Cream 1. ) Physical ∆ 2. ) Chemical ∆

Metabolizing Food 1. ) Physical ∆ 2. ) Chemical ∆

Dissolving Kool-Aid in H 2 O 1. ) Physical ∆ 2. ) Chemical ∆

Wood burning in a fireplace 1. ) Physical ∆ 2. ) Chemical ∆

MOLECULES For the sake of our class, we’ll use compounds and molecules somewhat interchangeably but they, ultimately, are NOT THE SAME Write this down: Molecule ≠ Compound

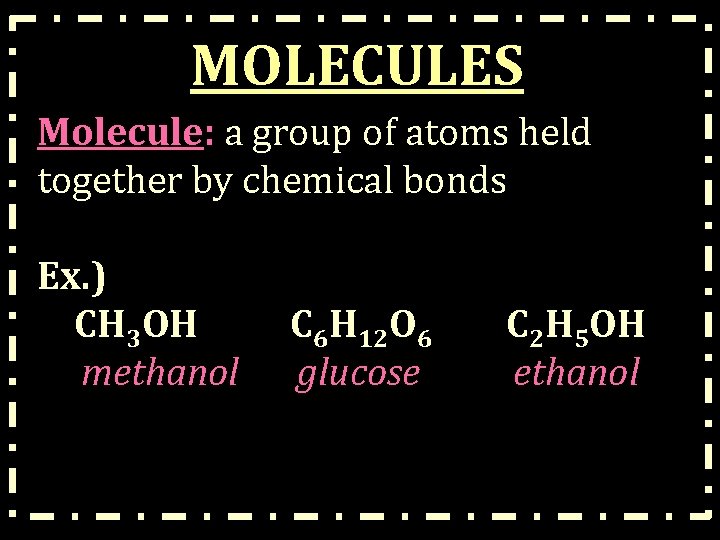
MOLECULES Molecule: a group of atoms held together by chemical bonds Ex. ) CH 3 OH methanol C 6 H 12 O 6 glucose C 2 H 5 OH ethanol

MOLECULES If I want to show more than one molecule in a chemical equation, put a coefficient before the chemical formula…. . 4 C 6 H 12 O 6 symbolizes 4 glucose molecules

How many Carbon atoms are in 1 molecule of glucose? 4 C 6 H 12 O 6 How many Carbon atoms are in 4 molecules of glucose?

How many atoms are in 1 molecule of glucose? 4 C 6 H 12 O 6 How many atoms are in 4 molecules of glucose?