Sub Science Matter Anything which has mass and

Sub- Science

• Matter : - Anything which has mass and occupies space is called matter. • Characteristics of matter: (1) Matter is made up of very thin particles. These particles have space between them and they attract each other. (2) It occupies space. (3) It has volume and mass.

Physical states of matter: Matter exist in these physical states, viz. , solid, liquid and gas. (A fourth state of matter called plasma occurs at Very high temperature and fifth state of matter is called as Bose-Einstein condensate (BEC)). 1. Solid : A substance which has definite shape and volume is called a solid. Ex. : - Stone, table, salts are solids.

2. Liquid: A substance which does not have definite shape but has a definite volume is called liquid. Ex: - water, milk, etc.

3. Gas: A substance which has neither a definite shape or nor definite volume is called a gas. Ex: - air, oxygen, carbon dioxide, etc.

• Characteristics of solids : (1) Solids have a definite shape and volume. (2) Most of the solids are rigid while some are elastics. (3) Solids are practically incompressible, i. e. the volume is not charged by external forces. (4) In solids, intermolecular forces are strong enough to keep the particles together, as a result solids are strong and have a high density.

• Characteristics of liquids : (1) Liquids have a definite volume. (2) Liquids do not have a definite shape. They take the shape of the container in which they are stored. (3) They have very low compressibility.

• Characteristics of gases : (1) Gases flow. (2) They spread in all directions. (3) Gases can be compressed or expanded easily.

• Melting point : The temperature at which a substance changes from solid to the liquid state is called the melting point.

• Freezing point : The temperature at which a substance changes from liquid to the solid state is called the freezing point.

• Evaporation : The process in which liquid gets converted into gaseous form at any temperature below its boiling point is called evaporation.

• Condensation : The process in which gas gets converted into liquid at constant temperature by giving out heat energy is called condensation.
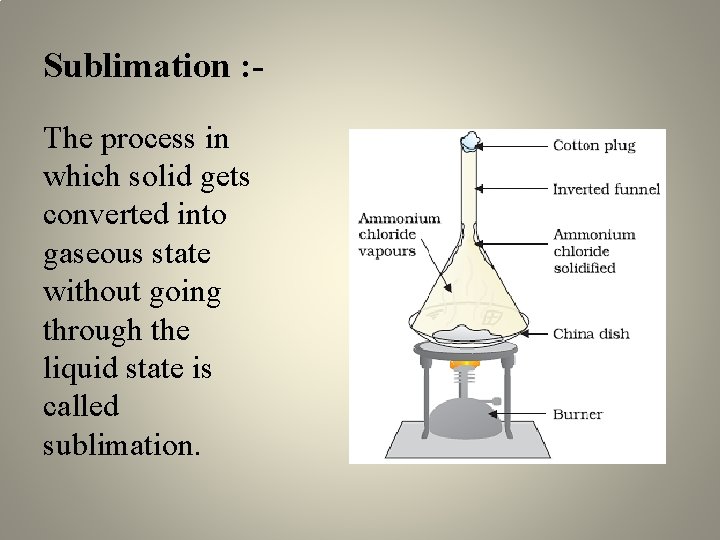
Sublimation : The process in which solid gets converted into gaseous state without going through the liquid state is called sublimation.

- Slides: 14