The Gas Laws Characteristics of Gases highly compressible

The Gas Laws

Characteristics of Gases • highly compressible. • occupy the full volume of their containers. • exert a uniform pressure on all inner surfaces of a container • diffuse (mix) easily and quickly • have very low densities.

Kinetic Molecular Theory – Gases consist of a large number of molecules in constant random motion. – Volume of individual molecules negligible compared to volume of container. – Intermolecular forces (forces between gas molecules) negligible. – Collision of gas particles are elastic so no kinetic energy is lost – As temperature increases the gas particles move faster, hence increased kinetic energy.
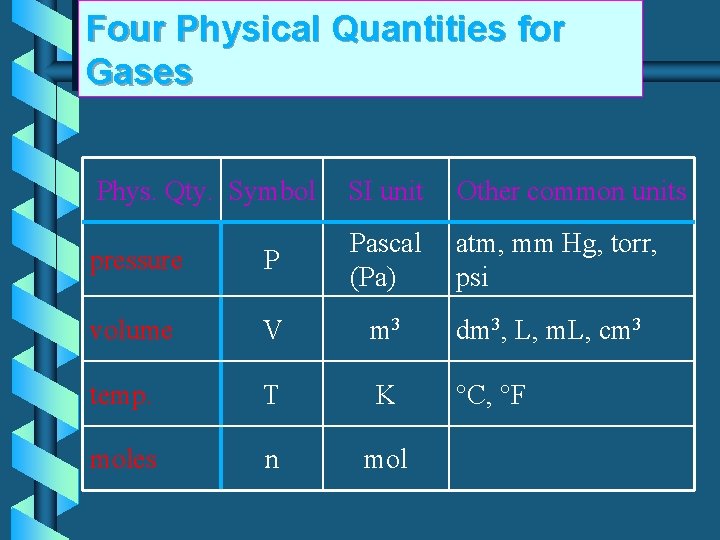
Four Physical Quantities for Gases Phys. Qty. Symbol SI unit Other common units pressure P Pascal (Pa) atm, mm Hg, torr, psi volume V m 3 dm 3, L, m. L, cm 3 temp. T K °C, °F moles n mol

Temperature Always use absolute temperature (Kelvin) when working with gases. ºF -459 ºC -273 K 0 32 212 0 100 273 373 K = ºC + 273

Kelvin Practice What is the approximate temperature for absolute zero in degrees Celsius and kelvin? Absolute zero is – 273 C or 0 K Calculate the missing temperatures 0 C 273 K = _______ 373 K 100 C = _______ – 173 C 100 K = _______ 243 K – 30 C = _______ 300 K = _______ C 27 403 K = _______ 130 C 298 K 25 C = _______ 0 K – 273 C = _______

Pressure (P ) is defined as the force exerted per unit area. The atmospheric pressure is measured using a barometer. Which shoes create the most pressure?

Pressure b KEY UNITS AT SEA LEVEL 101. 325 k. Pa (kilopascal) 1 atm 760 mm Hg 760 torr 14. 7 psi • 1 atm = 760 mm. Hg = 760 torr = 101325 Pa.
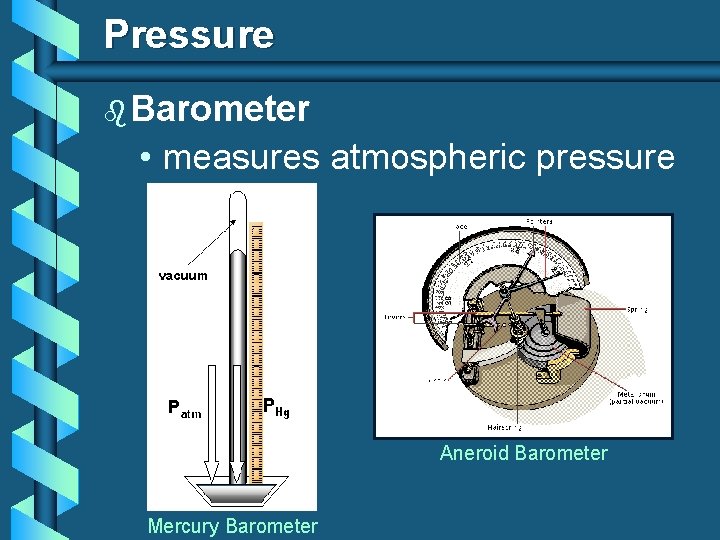
Pressure b Barometer • measures atmospheric pressure Aneroid Barometer Mercury Barometer

STP Standard Temperature & Pressure 273 K 101. 325 k. Pa

STP SLC Standard Laboratory Conditions 25°C or 298 K 101. 325 k. Pa

The Gas Laws -BOYLES -CHARLE -GAY-LUSSAC

Boyle’s Law The pressure and volume of a gas are inversely related at constant mass & temp. PV = k P V
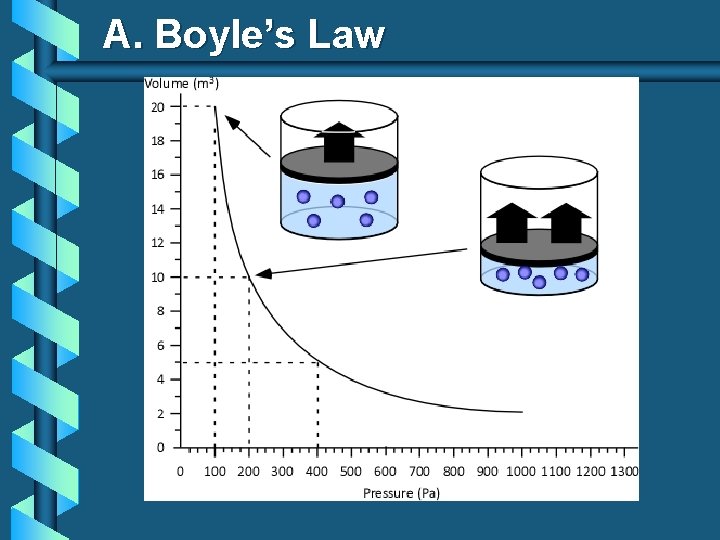
A. Boyle’s Law

Practice A sample of chlorine gas occupies a volume of 946 m. L at a pressure of 726 mm. Hg. What is the pressure of the gas (in mm. Hg) if the volume is reduced at constant temperature to 154 m. L?

A sample of chlorine gas occupies a volume of 946 m. L at a pressure of 726 mm. Hg. What is the pressure of the gas (in mm. Hg) if the volume is reduced at constant temperature to 154 m. L? P 1 x V 1 = P 2 x V 2 P 1 = 726 mm. Hg P 2 = ? V 1 = 946 m. L V 2 = 154 m. L P 1 x V 1 726 mm. Hg x 946 m. L = P 2 = = 4460 mm. Hg 154 m. L V 2

Charles’ Law The volume and absolute temperature (K) of a gas are directly related at constant mass & pressure V T
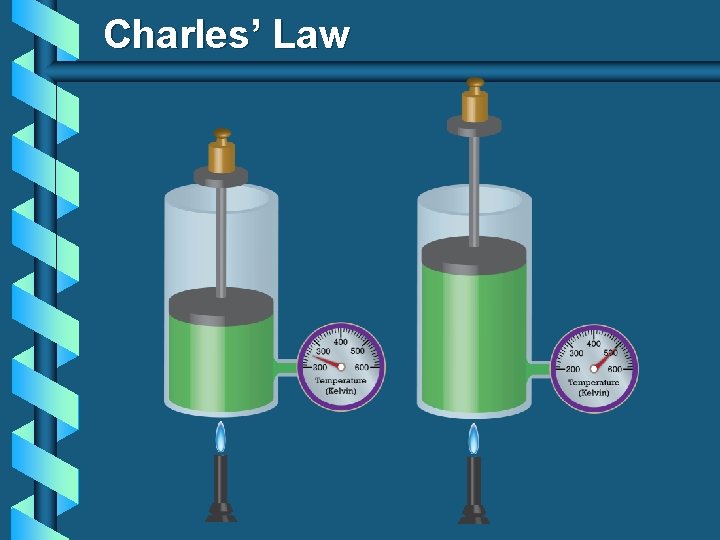
Charles’ Law

Practice 2. A sample of gas occupies 3. 5 L at 300 K. What volume will it occupy at 200 K? 3. If a 1 L balloon is heated from 22°C to 100°C, what will its new volume be?

2. A sample of gas occupies 3. 5 L at 300 K. What volume will it occupy at 200 K? V 1 = 3. 5 L, T 1 = 300 K, V 2 = ? , T 2 = 200 K 3. 5 L / 300 K = V 2 / 200 K V 2 = (3. 5 L/300 K) x (200 K) = 2. 3 L 3. If a 1 L balloon is heated from 22°C to 100°C, what will its new volume be? V 1 = 1 L, T 1 = 22°C = 295 K V 2 = ? , T 2 = 100 °C = 373 K V 1/T 1 = V 2/T 2, 1 L / 295 K = V 2 / 373 K V 2 = (1 L/295 K) x (373 K) = 1. 26 L For more lessons, visit www. chalkbored. com

A sample of carbon monoxide gas occupies 3. 20 L at 125 0 C. At what temperature will the gas occupy a volume of 1. 54 L if the pressure remains constant? V 1/T 1 = V 2/T 2 V 1 = 3. 20 L V 2 = 1. 54 L T 1 = 398. 15 K T 2 = ? V 2 x T 1 = T 2 = V 1 1. 54 L x 398. 15 K 3. 20 L = 192 K

Gay-Lussac’s Law The pressure and absolute temperature (K) of a gas are directly related at constant mass & volume P T
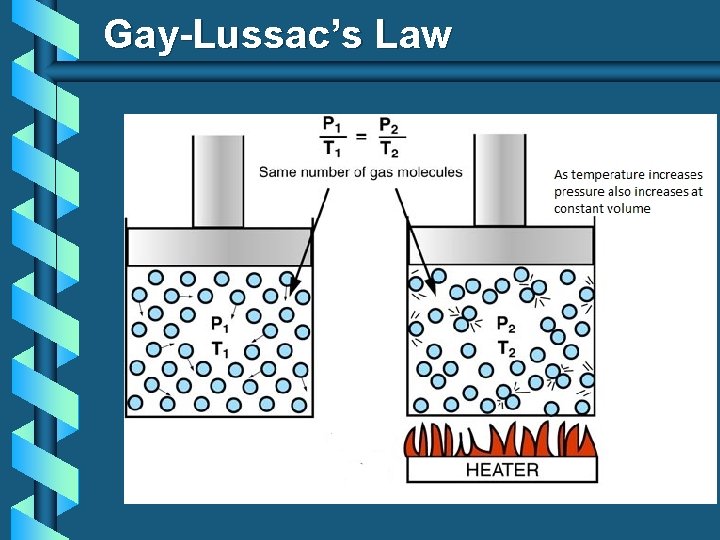
Gay-Lussac’s Law
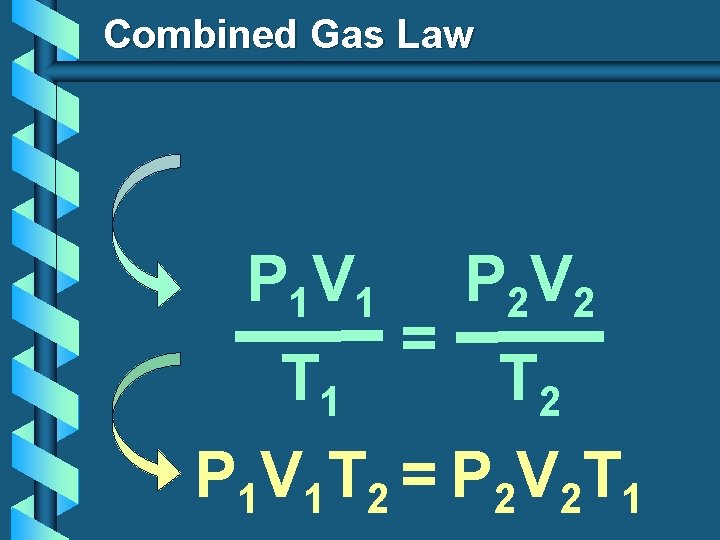
Combined Gas Law P 1 V 1 T 1 = P 2 V 2 T 2 P 1 V 1 T 2 = P 2 V 2 T 1
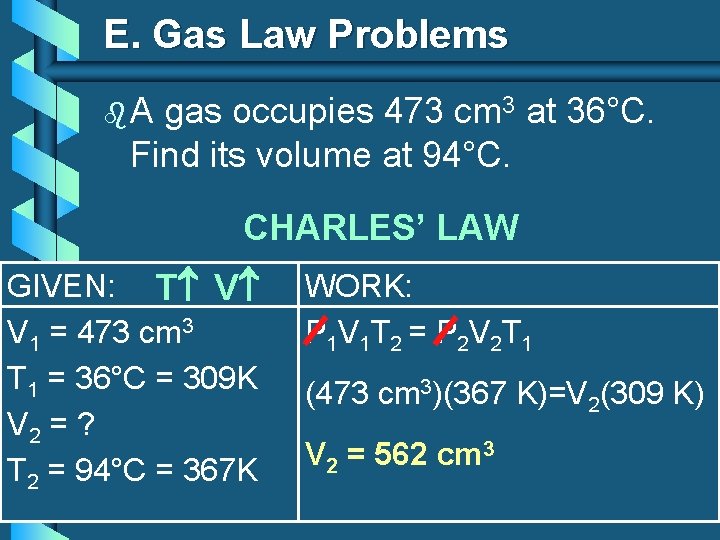
E. Gas Law Problems b. A gas occupies 473 cm 3 at 36°C. Find its volume at 94°C. CHARLES’ LAW GIVEN: T V V 1 = 473 cm 3 T 1 = 36°C = 309 K V 2 = ? T 2 = 94°C = 367 K WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (473 cm 3)(367 K)=V 2(309 K) V 2 = 562 cm 3

E. Gas Law Problems b. A gas occupies 100. m. L at 150. k. Pa. Find its volume at 200. k. Pa. BOYLE’S LAW GIVEN: P V V 1 = 100. m. L P 1 = 150. k. Pa V 2 = ? P 2 = 200. k. Pa WORK: P 1 V 1 T 2 = P 2 V 2 T 1 (150. k. Pa)(100. m. L)=(200. k. Pa)V 2 = 75. 0 m. L

Practice b. A gas occupies 7. 84 cm 3 at 71. 8 k. Pa & 25°C. Find its volume at STP.

E. Gas Law Problems b. A gas occupies 7. 84 cm 3 at 71. 8 k. Pa & 25°C. Find its volume at STP. COMBINED GAS LAW GIVEN: P T V WORK: V 1 = 7. 84 cm 3 P 1 V 1 T 2 = P 2 V 2 T 1 P 1 = 71. 8 k. Pa (71. 8 k. Pa)(7. 84 cm 3)(273 K) T 1 = 25°C = 298 K =(101. 325 k. Pa) V 2 (298 K) V 2 = ? P 2 = 101. 325 k. Pa V 2 = 5. 09 cm 3 T 2 = 273 K

E. Gas Law Problems b. A gas’ pressure is 765 torr at 23°C. At what temperature will the pressure be 560. torr? GAY-LUSSAC’S LAW GIVEN: P T WORK: P 1 = 765 torr P 1 V 1 T 2 = P 2 V 2 T 1 = 23°C = 296 K (765 torr)T 2 = (560. torr)(309 K) P 2 = 560. torr T 2 = 226 K = -47°C T 2 = ?

Avogadro’s Principle b Equal volumes of all gases contain equal numbers of moles at constant temp & pressure. V n

11. 5 The Ideal Gas Equation The gas laws can be combined into a general equation that describes the physical behavior of all gases. Boyle’s law Charles’s law rearrangement Avogadro’s law PV = n. RT R is the proportionality constant, called the gas constant.

B. Ideal Gas Law PV=n. RT UNIVERSAL GAS CONSTANT R = 8. 3145 J/mol·K R=0. 0821 L atm/mol K

b. R = 0. 0821 liter·atm/mol·K R = 8. 3145 J/mol·K R = 8. 2057 m 3·atm/mol·K R = 62. 3637 L·Torr/mol·K or L·mm. Hg/mol·K

B. Ideal Gas Law b Calculate the pressure in atmospheres of 0. 412 mol of He at 16°C & occupying 3. 25 L. IDEAL GAS LAW GIVEN: WORK: P = ? atm PV = n. RT n = 0. 412 mol P(3. 25)=(0. 412)(0. 0821)(289) mol L atm/mol K T = 16°C = 289 K K L V = 3. 25 L P = 3. 01 atm R= 0. 0821 L atm/mol K

B. Ideal Gas Law b Find the volume of 85 g of O 2 at 25°C and 104. 5 k. Pa. IDEAL GAS LAW GIVEN: WORK: V=? 85 g 1 mol = 2. 7 mol n = 85 g = 2. 7 mol 32. 00 g T = 25°C = 298 K PV = n. RT P = 104. 5 k. Pa (104. 5)V=(2. 7) (8. 315) (298) k. Pa mol dm 3 k. Pa/mol K R = 8. 315 V = 64 dm 3 k. Pa/mol K
- Slides: 35