Matter Properties of Matter Matter anything that has


















- Slides: 21

Matter & Properties of Matter

Matter anything that has mass and takes-up space the quantity of matter present mixtures and substances of the same kind of different kinds

Matter. Mass. All matter can be divided into two categories: Homogeneous. Heterogeneous-

Mixtures definitions • CAN be separated by physical means (heating, cooling, sorting, filtering) • Homogeneous & Heterogeneous

Mixtures Mixture- 2 categories of mixtures:

More on Mixtures Homogeneous: Example: Heterogeneous: Examples:

Mixtures definitions • is visibly the same throughout § Ex. salt water, Kool-Aid, milk • is visibly different throughout; different samples may not be exactly alike • ex. Salad dressing, chicken noodle soup

§ Pure Substance§ Two Categories: § Element: § Example: § Compound: § Examples:

§ CAN NOT be separated by physical means, ex: heating, cooling, sorting, filtering § Elements & Compounds § contains one kind of matter § Ex: hydrogen, oxygen, copper § a substance made of two or more elements that cannot be separated by physical means. § Ex: water, carbon dioxide, salt, and sugar

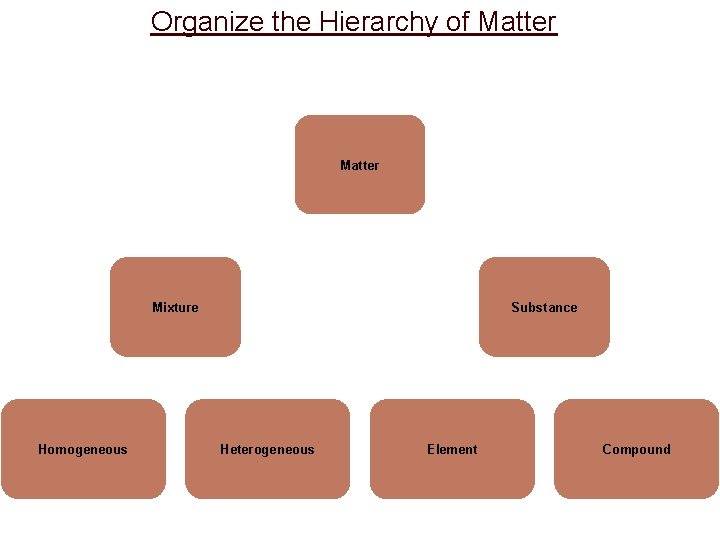
Organize the Hierarchy of Matter Mixture Homogeneous Substance Heterogeneous Element Compound

Substance Element: contains one kind of matter Example: hydrogen, oxygen, copper Compound: a substance made of two or more elements that cannot be separated by physical means. Examples: water, carbon dioxide, salt, and sugar

Questions Answer A = True and B = False 1. Matter is anything that has mass and takes up space 2. Mass is the quantity of matter present 3. A mixture cannot be broken down by physical means 4. A substance cannot be broken down by physical means

Questions A. Substance B. Matter C. Mixture 5 6 Homogeneous 7 Heterogeneous Element Compound

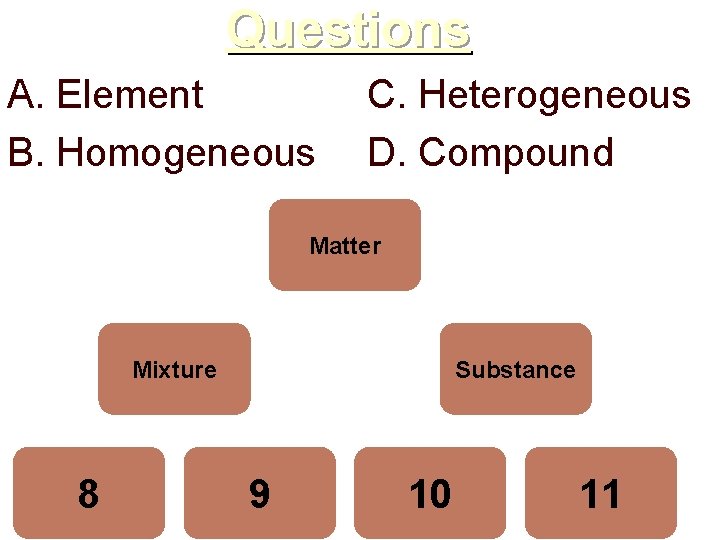
Questions A. Element B. Homogeneous C. Heterogeneous D. Compound Matter Mixture 8 Substance 9 10 11

States of Matter What determines the state of the matter? Depends on the amount of energy of the particles Kinetic energy (Ek): energy of motion How fast are the particles moving? Depends on temperature

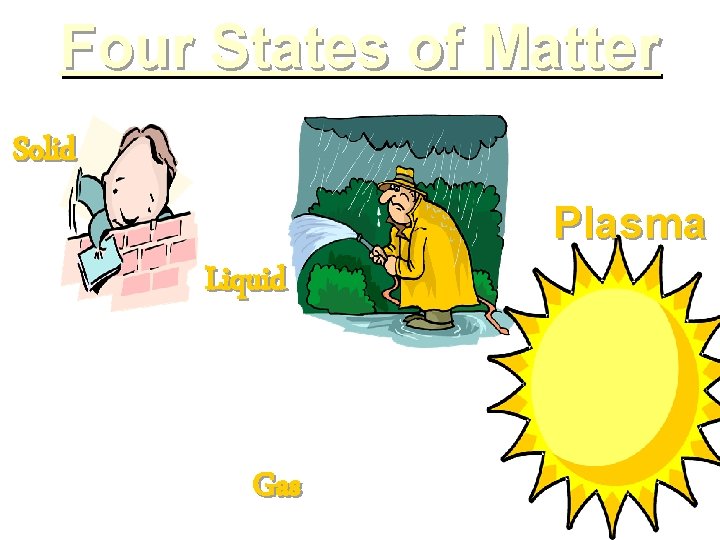
Four States of Matter Solid Plasma Liquid Gas

States of Matter Solid: E k? molecular behavior? Liquid: Ek ? molecules can slip out of their fixed position

States of Matter • definite volume, definite shape LOW Ek molecules vibrate in position definite volume, NO definite shape (can change shape easily) Moderate amount of Ek molecules can slip out of their fixed position

States of Matter Gas: NO definite volume, NO definite shape HIGH Ek molecules can move freely Plasma: Most common state of matter in the universe, it is rarely found on Earth VERY HIGH Ek

States of Matter Gas: Ek? Molecule behavior? Plasma: Ek?

Solid, Liquid, Gas (a) Particles in solid (b) Particles in liquid (c) Particles in gas