Properties of Matter Matter anything that has mass






















- Slides: 23

Properties of Matter

Matter – anything that has mass and volume Pretty much everything!

How Can We Describe Matter? Properties – the characteristics of a substance What are the properties of matter? l. Mass, volume l. Color, size, shape, etc.

What’s the Matter? l Matter is grouped into three phases l Solids l Liquids l Gases

Solids l Particles are close together and can vibrate but cannot move apart because of their strong attractions for each other l Have a definite shape and volume

Liquids l Particles are farther apart and can slide past each other l Have a definite volume, but no definite shape; they take the shape of the container they are in

Gases l Particles are even farther apart and are free to move anywhere in a container l Have no definite volume or shape

Let’s Compare Particles in Matter

Your Task l Make a tri-fold brochure comparing solids, liquids and gases. It must include: l. Definitions l. Drawings of the particle arrangements l. Examples – at least two!

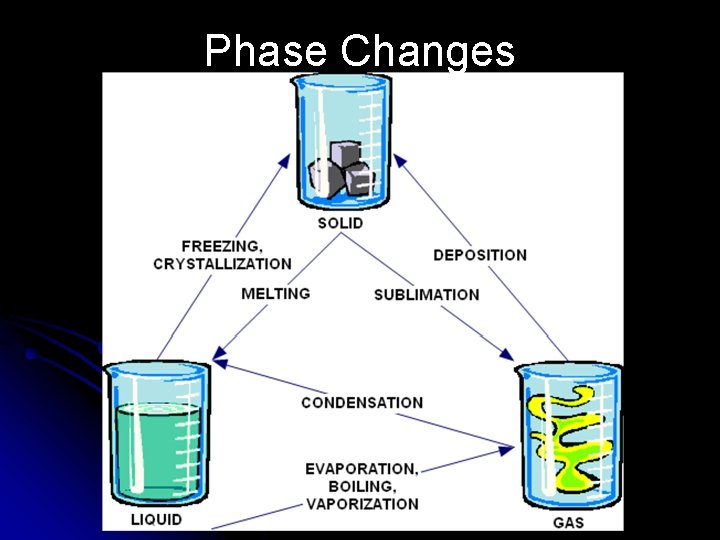
Phase Changes

How Phases Change l Solid to Liquid l Melting – the change of a substance from a solid to a liquid when heat is added l Liquid to Solid l Freezing – the change of a substance from a liquid to a solid when heat is removed

How Phases Change l Liquid to Gas l Vaporization – the change of a substance from a liquid to a gas when heat is added l Gas to Liquid l Condensation – the change of a substance from a gas to a liquid when heat is removed

3 Minute Chat l Discuss the following, explaining your answers: l What are pure substances? l What are mixtures? l How are they alike? l How are they different?

Elements l. Element – pure substance that cannot be separated by physical or chemical means - atoms l. Any one element on the periodic table! l. Ex.

Compounds l Compound – substance made of two or more elements that are joined by chemical bonds - molecules l Can only be separated by chemical means, not physical l H 2 O l Na. Cl l Two or more elements

Mixtures l. Mixture – a combination of two or more pure substances that are not chemically combined l. Can physically separate

Stuff Lab Is the “stuff” an element, a compound, or a mixture? l If stuff is a mixture, how can you separate the components? l Physical methods l

How to Fold Filter Paper

Homogeneous Mixtures l Two or more substances spread out evenly without settling - the same throughout l Appears clear l Also called solutions l Iced tea l Salt water

Heterogeneous Mixtures l. A mixture in which different parts are easily distinguished l Sandwich pencil (lead, wood, eraser)

Heterogeneous Mixtures l Colloid – heterogeneous mixture that never settles l Milk l Can tell by Tyndall Effect l Scattering of light by the particles in the mixture

Heterogeneous Mixtures l Suspensions – contain a liquid with visible particles that settle out when it stands – also have Tyndall Effect l Orange juice l Different from a colloid or solution because particles will settle out

Whiteboard Check What type of mixture (homogeneous or heterogeneous) is each of the following: l Chocolate chip cookie l Coffee l Apple juice l Lucky Charms® cereal l Trail mix l