BASIC CHEMISTRY Matter anything that occupies space Elements

BASIC CHEMISTRY �Matter: anything that occupies space �Elements: substances that cannot be broken down chemically into simpler parts (Ex. Hydrogen, oxygen, carbon, iron) �Over 100 have been identified in nature �Less than 30 are important to living things � Atom: the simplest particle of an element that retains all of the properties of that element � The basic building blocks of all matter
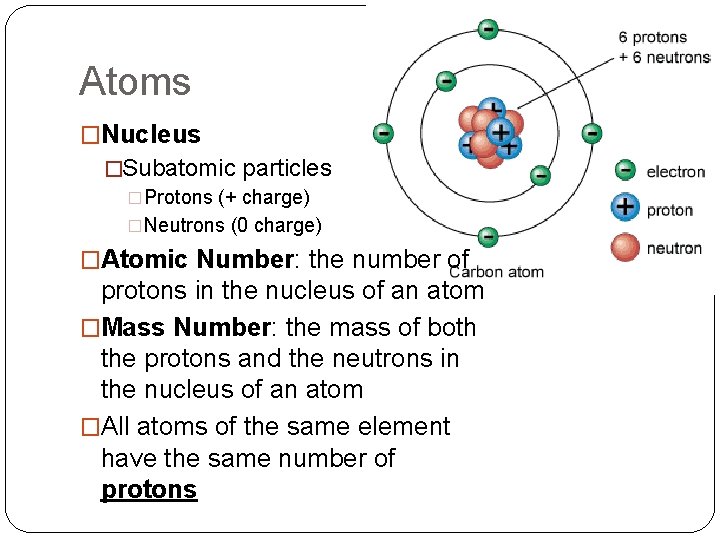
Atoms �Nucleus �Subatomic particles �Protons (+ charge) �Neutrons (0 charge) �Atomic Number: the number of protons in the nucleus of an atom �Mass Number: the mass of both the protons and the neutrons in the nucleus of an atom �All atoms of the same element have the same number of protons

Isotopes �All atoms of an element do not have the same number of neutrons �Isotopes: atoms of the same element that have a different number of neutrons

Electrons �Electron: negatively charged, energetic particles orbiting the nucleus of an atom �The number of protons = the number of electrons �Outside electrons have more energy and are responsible for bonding atoms to each other to form COMPOUNDS

Compounds �Compound: 2 or more atoms or elements bonded together in fixed proportions �Chemical formula: shows the proportions of each element �Examples: �H₂O = water (2 hydrogen, 1 oxygen) �O₂ = oxygen gas �C₂O = Carbon Dioxide
Chemical Bonds �Most elements are not stable in nature �They want to have a balanced number of electrons in their orbit to become stable �Molecule: the simplest part of a substance that retains all of the properties of that substance and can exist in a free state
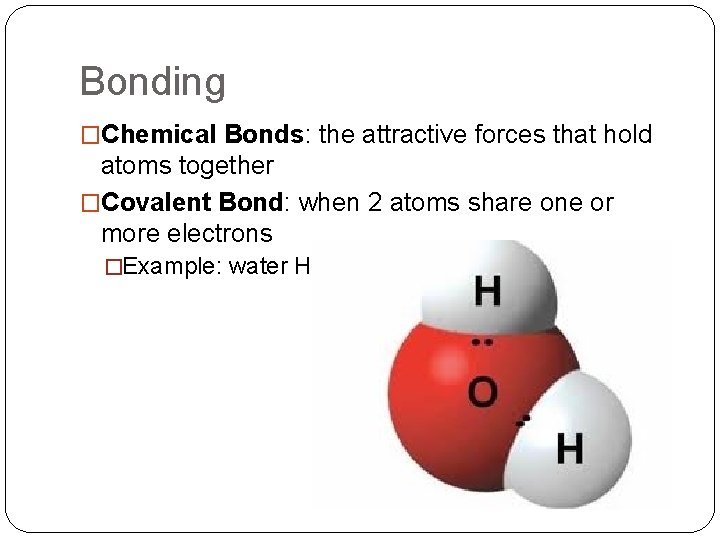
Bonding �Chemical Bonds: the attractive forces that hold atoms together �Covalent Bond: when 2 atoms share one or more electrons �Example: water H₂O

Bonding �Ionic bond: bonding of 2 atoms because of the electrical attraction to each other �+ and – charges attract �Ex. Sodium has a positive charge and Chlorine has a negative charge = sodium chloride (table salt)
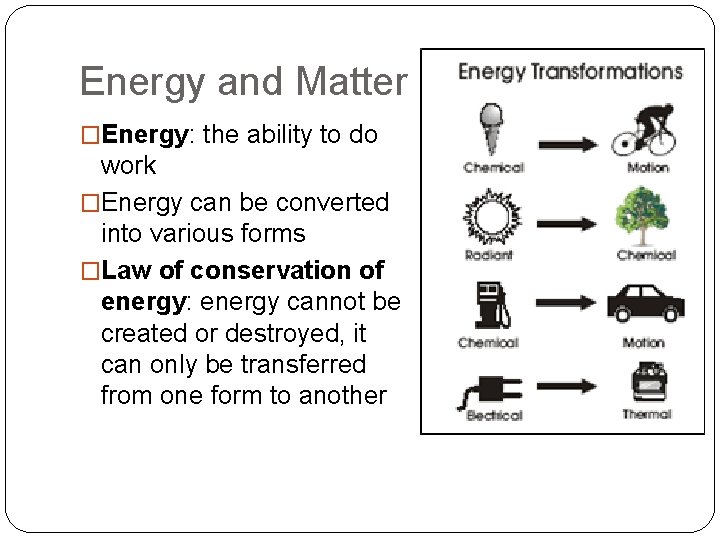
Energy and Matter �Energy: the ability to do work �Energy can be converted into various forms �Law of conservation of energy: energy cannot be created or destroyed, it can only be transferred from one form to another
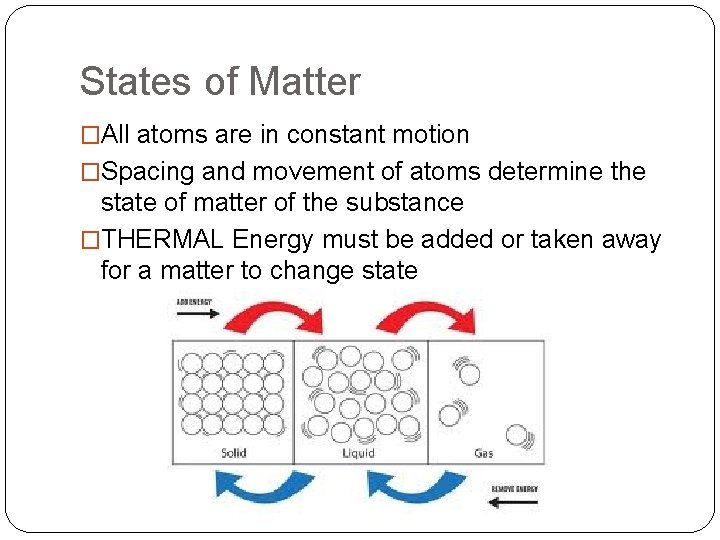
States of Matter �All atoms are in constant motion �Spacing and movement of atoms determine the state of matter of the substance �THERMAL Energy must be added or taken away for a matter to change state

States of Matter � Solid �Atoms are tightly packed �Little movement �Maintains a fixed volume and shape � Liquid �Has a fixed volume �Atoms move more freely than in a solid �Can flow and conform to different shapes � Gas �Atoms have little or no attraction to each other �Atoms fill the space they occupy � **Water is the only exception to this rule**
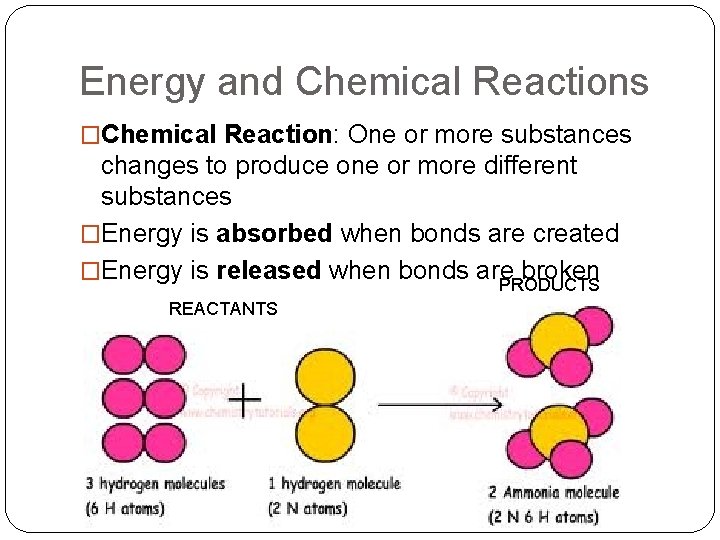
Energy and Chemical Reactions �Chemical Reaction: One or more substances changes to produce one or more different substances �Energy is absorbed when bonds are created �Energy is released when bonds are broken PRODUCTS REACTANTS

Chemical Reactions �Metabolism: all of the chemical reactions that occur in an organism to maintain its internal function and create energy �Breaking down food �Making new cells and DNA �Growing �Making hormones �Circulating blood
- Slides: 13