BASIC CHEMISTRY DEFINITIONS 1 Matter anything that occupies

BASIC CHEMISTRY DEFINITIONS

1) Matter anything that occupies space and has mass. (also inertia- resisitance to a change in motion) Total Shuttle Mass 2, 029, 203 KG (4, 474, 574 LB) = 2, 237. 287 tons

2) Element • - a pure substance that consists entirely of one kind of atom. They are represented on the periodic table of elements with 1 -2 letters such a H for hydrogen, and Na for Sodium.


3) Atom It means “unable to cut…” It is the basic unit of matter (simplest part of an element that retains the properties of that element. )

Current Atomic Theory • The atom consists of a small, dense positively-charged nucleus surrounded by mostly empty space. (Most of the atom’s volume is empty space. ) • If the nucleus was the size of an orange, the whole atom would measure about 24 city blocks.

• If the nucleus was the size of a small paper clip, the whole atom would be the size of a football stadium. • Even though the atom’s nucleus is relatively small, it makes up over 99. 9% of the atom’s mass.

4) nucleus (of an atom, not of a cell) – the center of the atom that contains the protons and the neutrons or subatomic particles

5) protons positive particle in the nucleus 6) neutron the neutral (no charge) particle in the nucleus 7) electrons the negative particle that moves around the nucleus, (Electrons are of negligible mass compared to the protons and neutrons. )
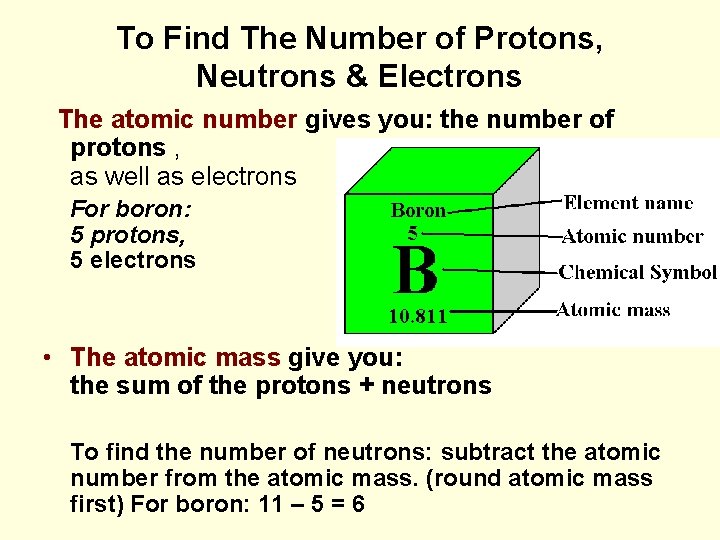
To Find The Number of Protons, Neutrons & Electrons The atomic number gives you: the number of protons , as well as electrons For boron: 5 protons, 5 electrons • The atomic mass give you: the sum of the protons + neutrons To find the number of neutrons: subtract the atomic number from the atomic mass. (round atomic mass first) For boron: 11 – 5 = 6

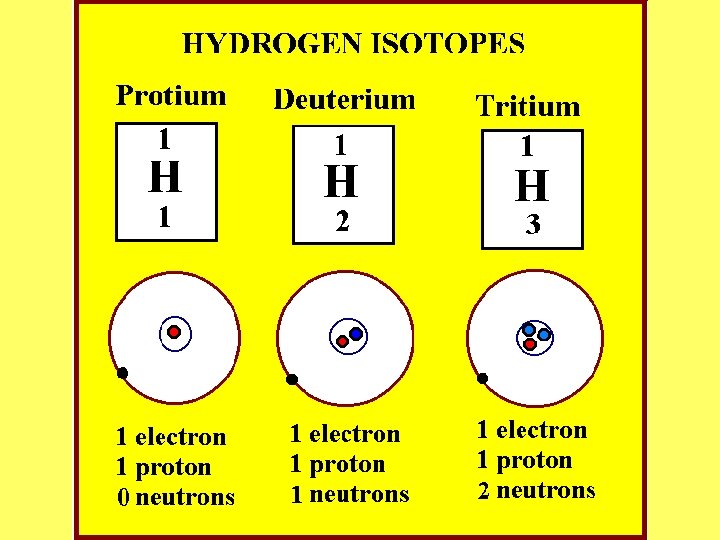
Isotope Atoms (elements) with the same atomic number that have different numbers of neutrons. (for example: Carbon 12, Carbon 13, & Carbon 14, or Hydrogen 1, 2, & 3). Some are radioactive. C-12 C-14

8) compound a pure substance that is made up of the atoms of two or more elements, chemically combined in definite proportions. It may be represented by a chemical formula, such as: H 2 O or Na. Cl, or CO 2.

9) Chemical reaction The process by which compounds are formed or broken down. Most elements tend to undergo chemical reactions to make their atoms more stable. Bonds are broken and formed, and atoms are rearranged. Substances formed have different properties than the original substances.

• Outer shell electrons called valance electrons are involved in chemical bonding. • Atoms are more stable what they have 8 (sometimes 2) valance electrons in their outer shell. – This is called the octet rule.

10) Covalent bond Two atoms share one or more pairs of electrons. formed when electrons are shared between atoms in a compound. The structure formed is called a molecule and is the smallest part of a covalent compound. (A water molecule is covalently bonded. )

11) Molecule - simplest part of a substance (covalently-bonded compound) that retains the properties of the substance. (Made up of two or more different elements chemically combined. )

12) Ionic bond a chemical bond between atoms in which one or more electrons are transferred.

13) ions The atoms involved in an ionic bond become charged during the chemical reaction. They are called ions (+ ions lost, while – ions gain electron/s) Negative ions are attracted and bond to positive ions. Na. Cl (Salt) = Na+ and Cl-

14) States of matter Solids, liquids, gases- states of matter determined by the rate at which atoms or molecules move. • Solids- tightly packed with fixed volume & shape, • Liquids – not as tightly, linked with fixed volume, but not shape. • Gases- particle with little or no attraction-no fixed volume or shape.

15) Reactants and products Reactants – left side of the chemical equation, while the right side = Products.

16) mixture matter composed of two or more elements, or compounds, that are physically mixed together, but not chemically combined. Salt and pepper stirred together is a mixture.

17) solution (homogenous) • a mixture of two or more substances in which the molecules of the substances are evenly distributed. (salt dissolved in water) Solutions can be mixtures of liquids, solids, or gases.

solution, solute & solvent A solution is a mixture in which a solute is dissolved in a solvent. It has very small particle size. (homogenous mixture. )

Salt water solute -substance dissolved to form a solution (salt - Na. Cl), solvent – substance in which a solute is dissolved (water – H 2 O)

COLLOIDS: • Particles that are intermediate in size between those in a solution or a suspension from heterogeneous mixtures called colloidal dispersions or simply colloids. Particles of a colloid are small enough to stay suspended by the constant movement of surrounding molecules. • Colloidal particles make up a dispersed phase, while the solvent-like phase is called the dispersing medium. Examples: Gels, emulsions, foams, aerosols, fog, smog, etc. Cytoplasm is said to be a colloidal dispersion.

SUSPENSIONS: • A mixture of water and undissolved materials. The particles larger than those in solutions or colloids, but are small enough so that they don’t settle out, or are suspended for a time by the movement of the water. If the water stops moving, they tend to settle-out.

18) acid Solution with higher concentration of H+ ions (also hydronium ions) than pure water. Strong acids tend to have p. H values that are low & range from 0 -3. (lower OH- hydroxide ions)
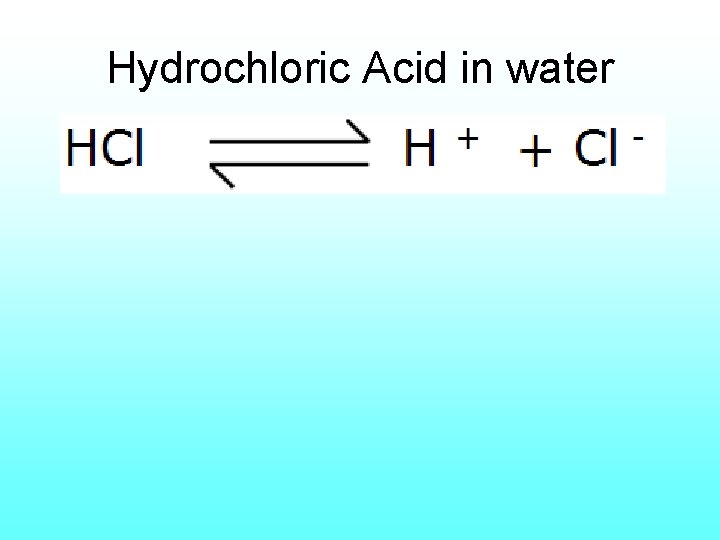
Hydrochloric Acid in water

• Acids tend to be sour, and in concentrated forms, highly corrosive to some materials. • Your stomach contains hydrochloric acids which makes vomiting irritating to tissues. • Sulfuric acids is found in car batteries and used in many manufacturing processes. • Penny tarnish can be removed with the acids in catsup.
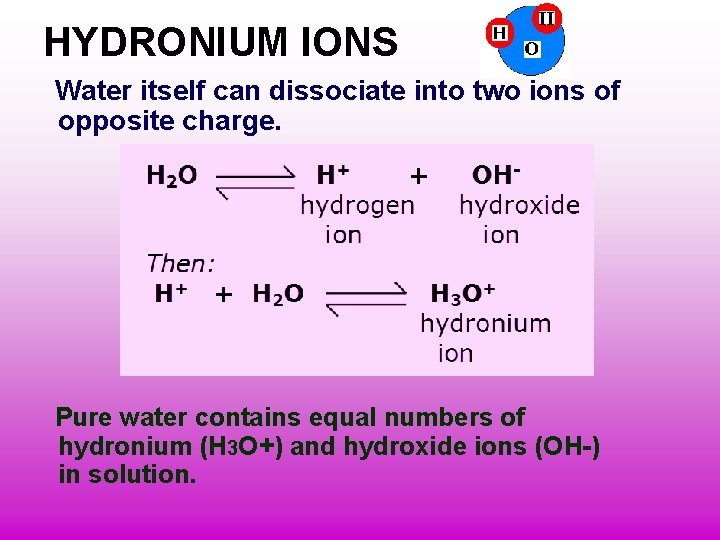
HYDRONIUM IONS Water itself can dissociate into two ions of opposite charge. Pure water contains equal numbers of hydronium (H 3 O+) and hydroxide ions (OH-) in solution.

19) base A solution with lower concentration of H+ ions (hydronium H 3 O+) than water (higher OHhydroxide ions) These solutions are said to be alkaline. Strong bases tend to have p. H values from 11 – 14.
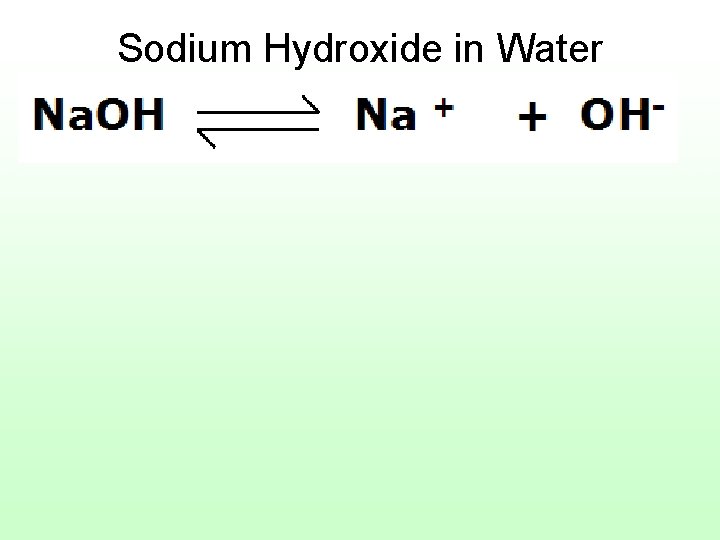
Sodium Hydroxide in Water

Bases tend to taste bitter. Basic solutions are called alkaline. Bases feel slippery. They can react with the oils in the skin to form soap. Soap can be made by reacting fat with a base.

Early soap-making with lye and animal fat. It is called saponification.

Bases can be caustic. Sodium hydroxide is also called “caustic soda” and can cause chemical burns.

20) p. H scale a scale to measure the concentration of H+ ions (hydrogen ions or hydronium ions) in solution.

p. H Numbers

Acid – Base indicators • Chemical Indicators: substances such as litmus paper can be used to detect the presence of an acid or base. They can also approximate p. H number.

An Electrolyte Tester Electrolytes have ions in solution. These allow the electric current to flow. This can be demonstrated by an electrolyte tester. HCl dissolved in water released ions into the water that can conduct an electric current.

Acid, base, or salt solutions are electrolytes ELECTROLYTE: any substance containing free ions that make the substance electrically conductive. (The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible. )

21) buffers weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in p. H.
- Slides: 42