Matter Anything that has mass and takes up














- Slides: 22


Matter • Anything that has mass and takes up space • The “stuff” everything is made of • Composed of small particles called atoms • The different atoms that exist combine in different ways to make up all the matter in the universe 2

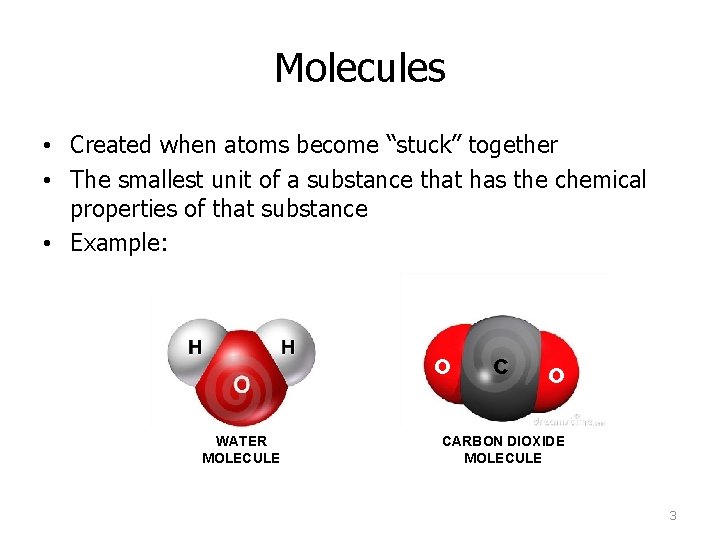
Molecules • Created when atoms become “stuck” together • The smallest unit of a substance that has the chemical properties of that substance • Example: o WATER MOLECULE c o CARBON DIOXIDE MOLECULE 3

Elements • Substances containing only one type of atom – Cl 2 – N 2 – H 2 Remember: Diatomic Elements Br I N Cl H O F • Found on the periodic table • Cannot be broken down into simpler substances by chemical means 4

Compounds • Substances containing 2 or more different types of atoms combined in a definite ratio • Example: – H 2 O (2 atoms hydrogen; 1 atom oxygen) – Na. Cl (1 atom sodium; 1 atom chlorine) – CO 2 (1 atom carbon; 2 atoms oxygen) • Can be broken down by chemical means 5

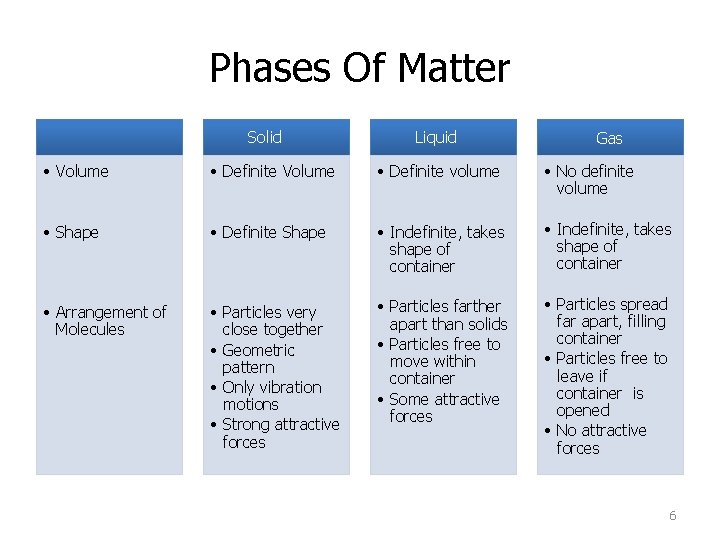
Phases Of Matter Solid Liquid Gas • Volume • Definite volume • No definite volume • Shape • Definite Shape • Indefinite, takes shape of container • Arrangement of Molecules • Particles very close together • Geometric pattern • Only vibration motions • Strong attractive forces • Particles farther apart than solids • Particles free to move within container • Some attractive forces • Particles spread far apart, filling container • Particles free to leave if container is opened • No attractive forces 6

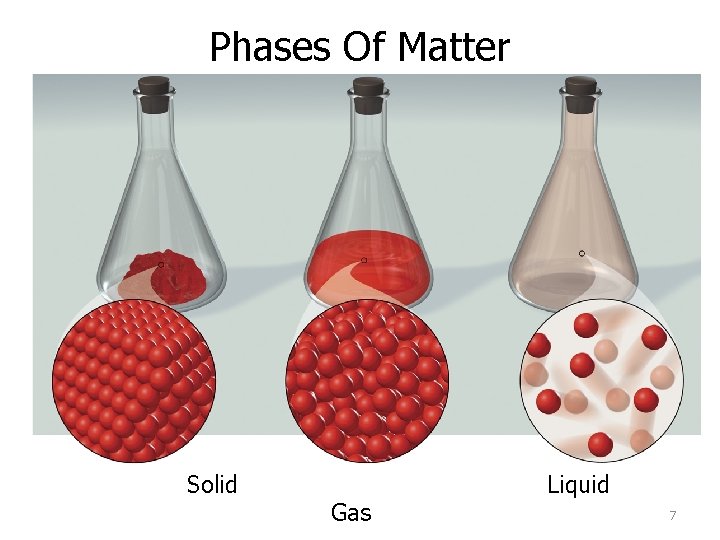
Phases Of Matter Solid Gas Liquid 7

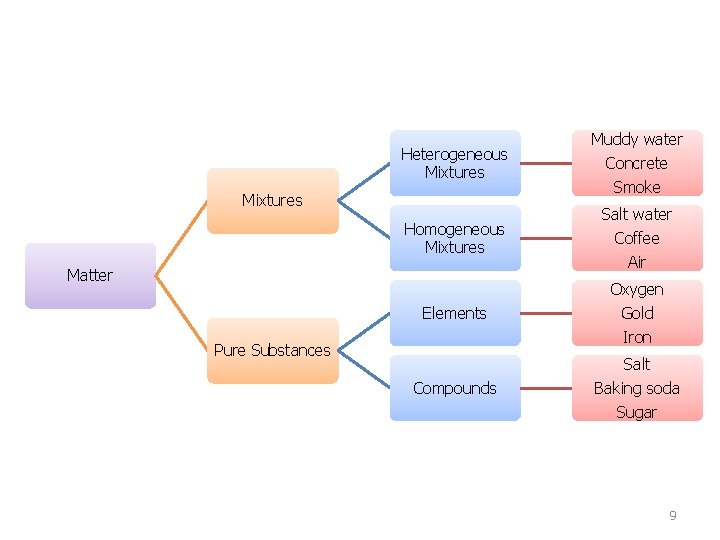
Mixture • Contains two or more components not chemically combined 1. Homogeneous • • • Same throughout; uniformly mixed Also known as solutions Ex. Air (gas), milk (liquid), brass (solid) 2. Heterogeneous • • Regions differ from one area to another; not uniformly mixed Ex. Chocolate chip cookie, Italian salad dressing 8

Heterogeneous Mixtures Homogeneous Mixtures Matter Muddy water Concrete Smoke Salt water Coffee Air Oxygen Elements Gold Iron Pure Substances Salt Compounds Baking soda Sugar 9

Separating Mixtures • By physical means such as: – Decanting • Separates a liquid from insoluble solid sediment • Involves carefully pouring the liquid from the solid without disturbing the solid 10

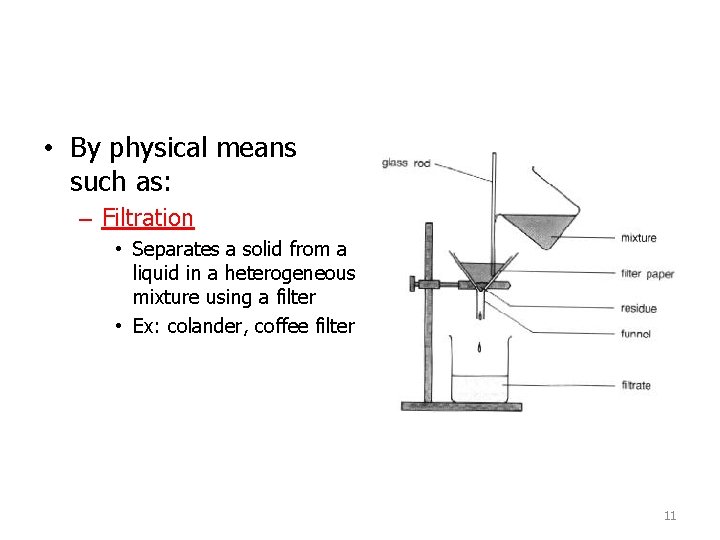
• By physical means such as: – Filtration • Separates a solid from a liquid in a heterogeneous mixture using a filter • Ex: colander, coffee filter 11

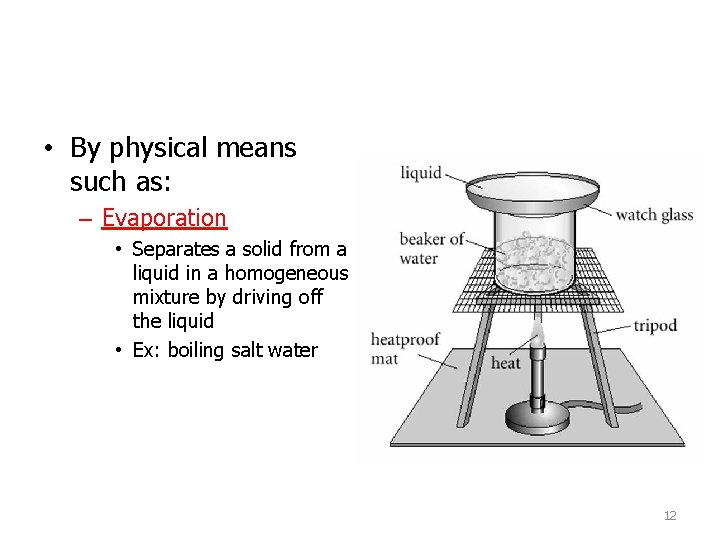
• By physical means such as: – Evaporation • Separates a solid from a liquid in a homogeneous mixture by driving off the liquid • Ex: boiling salt water 12

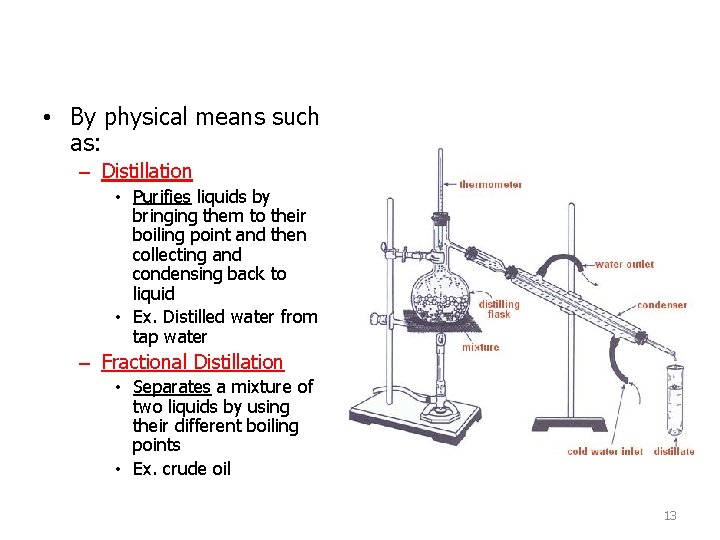
• By physical means such as: – Distillation • Purifies liquids by bringing them to their boiling point and then collecting and condensing back to liquid • Ex. Distilled water from tap water – Fractional Distillation • Separates a mixture of two liquids by using their different boiling points • Ex. crude oil 13

• By physical means such as: – Chromatography • Separates pigments of different densities • Ex: Extracting pigment from a leaf and separating the chlorophyll, carotene, etc. from each other 14

Separating Compounds • Involves a chemical reaction which breaks the bonds between atoms • Causes a chemical change (a change in matter from one substance to another) • Ex. H 2 O Electricity H 2(g) + O 2(g) (l) Decomposes 15

Try These!!! 16

Properties • Used to describe substances (matter that has a uniform and definite composition) Physical Properties • Properties that can change without the substance becoming a new substance Chemical Properties • Properties that cause a substance to become a new substance 17

Intensive Physical Properties (depend on the type of matter) • • State (phase) of matter, color, odor, density, solubility, hardness, melting point, boiling point Malleability Capable of being hammered into sheets – • • Ductility Capable of being drawn into wire – • • Aluminum foil Copper Conductivity – – Ability of electrons to flow Heat and electrical 18

Extensive Physical Properties (depend on the amount of matter) • Volume – Space occupied by an object • Mass – Amount of matter an object contains 19

Chemical Properties • Rusting • Burning • Leaves turning color • Digestion 20

Physical & Chemical Changes Physical Changes • Change in form or phase of a substance but not its chemical composition • Ex. – Phase changes Chemical Changes • • Melting, freezing, evaporation, condensation, sublimation – Cutting hair – Breaking a window – Crushing a piece of chalk Change in chemical composition due to a reaction Ex. – Silver tarnishes by reacting with substances in the air – Making scrambled eggs – Burning a candle Evidence of a chemical change may include: – – • Transfer of energy Change in color Production of gas Formation of a precipitate (solid that forms and settles out of a liquid mixture) Referred to as chemical reactions 21

Chemical Reactions • When at least 1 substance changes into a new substance Z (products) X + Y (reactants) • Reactant- Substances present at the start of a reaction • Product- Substances present at the end of a reaction 22